3.2 carbs, lipids & proteins notes
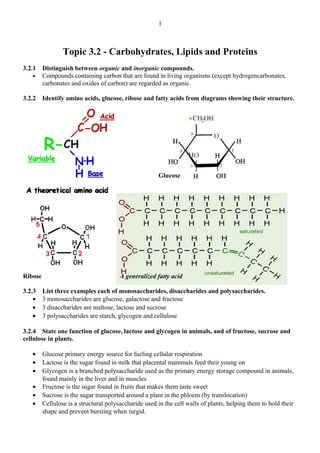
- 1. Topic 3.2 - Carbohydrates, Lipids and Proteins 3.2.1 Distinguish between organic and inorganic compounds. • Compounds containing carbon that are found in living organisms (except hydrogencarbonates, carbonates and oxides of carbon) are regarded as organic. 3.2.2 Identify amino acids, glucose, ribose and fatty acids from diagrams showing their structure. Glucose Ribose A generalized fatty acid 3.2.3 List three examples each of monosaccharides, disaccharides and polysaccharides. • 3 monosaccharides are glucose, galactose and fructose • 3 disaccharides are maltose, lactose and sucrose • 3 polysaccharides are starch, glycogen and cellulose 3.2.4 State one function of glucose, lactose and glycogen in animals, and of fructose, sucrose and cellulose in plants. • Glucose primary energy source for fueling cellular respiration • Lactose is the sugar found in milk that placental mammals feed their young on • Glycogen is a branched polysaccharide used as the primary energy storage compound in animals, found mainly in the liver and in muscles • Fructose is the sugar found in fruits that makes them taste sweet • Sucrose is the sugar transported around a plant in the phloem (by translocation) • Cellulose is a structural polysaccharide used in the cell walls of plants, helping them to hold their shape and prevent bursting when turgid. 1
- 2. 3.2.5 Outline the role of condensation and hydrolysis in the relationships between monosaccharides, disaccharides, and polysaccharides; fatty acids, glycerol and glycerides; amino acids, dipeptides and polypeptides. (Can use words or equations) • For monosaccharides, fatty acids, and amino acids to become disaccharides, glycerol, and dipeptides, a condensation reaction needs to occur. When these monomers covalently bond, a water molecule is released; this is a condensation reaction. When many monomers join together through condensation reactions, polymers result. • In a hydrolysis reaction, the addition of a water molecule breaks down the covalent bonds and polymers break down into monomers. Condensation synthesis • monomers linked together (=anabolised) to form polymers • through the release of H2O • with energy supplied Hydrolysis • polymers broken down (= catabolised) into monomers (as in digestion) • with H2O used as a source of -H and a -OH group • catalysed by enzymes CARBOHYDRATES Condensation synthesis monosaccharide + monosaccharide disaccharide + H2O polysaccharide + nH2O • two monosaccharide monomers are linked to form a disaccharide • releasing one H2O molecule • repeated condensation synthesis reactions produce polysaccharides Hydrolysis polysaccharide = nH2O disaccharide + H2O monosaccharide + monosaccharide •a polysaccharide is broken down into separate disaccharides, then down to monosaccharides • with H2O molecules used as a sources of -H and a -OH groups • catalysed by enzymes 2
- 3. DIPEPTIDES Condensation synthesis amino acid + amino acid dipeptide + H2O polypeptide + nH2O • two amino acid monomers are linked to form a dipeptide • releasing one H2O molecule • repeated condensation synthesis reactions produce polypeptides (=proteins) Hydrolysis polypeptide + nH2O dipeptide + H2O amino acid + amino acid • a polypeptide is broken down into separate amino acid molecules • with H2O molecules used as a sources of -H and a -OH groups • catalysed by enzymes LIPIDS Condensation synthesis 3 fatty acids + glycerol triglyceride + 3H2O • three fatty acids are linked to a glycerol molecule • releasing three H2O molecules Hydrolysis triglyceride + 3H2O 3 fatty acids + glycerol • a triglyceride is broken down into one glycerol and three fatty acid molecules • with three H2O molecules used as a sources of -H and a -OH groups • catalysed by enzymes 3
- 4. 3.2.6 State three functions of lipids. Triglycerides • Concentrated long-term energy storage ,which can release fuel for cellular respiration as needed • Great insulators (blubber!). Phospholipids • structural framework of cellular membranes Steroids • Hormones = messenger molecules 3.2.7 Compare the use of carbohydrates and lipids in energy storage. • The use of carbohydrates in energy storage is through its sugar polymers, glycogen in animals and starch in plants. • These sugars are released when the demand for sugar increases. • Animals use lipids, mainly fats, for long-term energy storage. • Lipids contain twice the amount of energy per unit mass or volume as do carbohydrates. 4