Carbohydrate structure
- 1. CARBOHYDRATE STRUCTURE and PROPERTIES Whilhelmina Annie Mensah 1
- 2. • I would feel more optimistic about a bright future for man, if he spent less time proving that he can outwit Nature and more time tasting her sweetness and respecting her seniority. —E. B. White, “Coon Tree,” 1977 2
- 3. A. Biological and medical importance Functions B. Chemical nature Ketose and hexoses C. Structure Fisher , Haworth and Comformations D. Classification of carbohydrates Monosaccharides Hexoses of physiologic importance Important sugar derivatives Oligosaccharides: Disaccharides Polysaccharides Homopolysaccharides and heteropolysaccharides E. Properties of monosaccharides Lecture objectives 3
- 4. Key Words Saccharide: sugar Chiral /Asymmetrical : a carbon with four groups bounded to it. Anomeric or carbonyl : carbon 1 Hydroxyl group: OH group Reduction : addition of hydrogen, removal of oxygen Oxidation : addition of oxygen, removal of hydrogen, 4
- 5. 5 Carbohydrates are the most abundant biomolecules in nature Present in humans, animal tissues, plants and in micro- organisms. Also present in tissue fluids, blood, milk, secretions and excretions of animals. A. Medical And Biological Importance
- 6. A. Medical And Biological Importance – Functions Serve as energy stores and fuels, and metabolic intermediates, glucose and glycogen Components of several animal structure and plant structures e.g. cartilage, tendons, cellluose A part of DNA and RNA Part of drugs e.g. the antibiotic streptomycin etc. 6
- 7. Carbohydrates ( saccharides - SUGAR) are polyhydroxy alcohols with a aldehyde or keto functional group Empirical formula is (CH2O)n; some also contain nitrogen, phosphorus, or sulfur B. Chemical Nature Of Carbohydrates 7
- 8. C. STRUCTURE The structure of some carbohydrates exist in three (3) forms 8
- 9. Relating Fisher projection to Haworth projection(Ring) – Groups on the right in a Fischer projection point downwards in a Haworth projection – Groups on the left in a Fischer projection point upwards in a Haworth projection C. STRUCTURE 9
- 10. Ring form conformations Pyranose : 6 member ring furanose: 5 member ring D. STRUCTURE 10
- 11. D. Classification Of Carbohydrates 1. Monosaccharides 2. Oligosaccharides 2a. Disaccharides 3. Polysaccharides 11
- 12. D. Classification Of Carbohydrates 1. Monosaccharide (simple sugar)- cannot be hydrolyzed into simpler carbohydrates, they include Trioses Tetroses Pentoses Hexoses Or Heptoses 12
- 13. D. Classification Of Carbohydrates 1. Monosaccharide (simple sugar) Important monosaccharides, examples 13
- 14. D. Classification Of Carbohydrates 1. Monosaccharide (simple sugars) Hexoses of physiologic importance Sugar Source Clinical significance D –glucose •Fruit Juices, •cane sugar •Hydrolysis of starch, cane sugar and lactose Present in the urine (glycosuria) in diabetes mellitus owing to raised blood glucose (hyperglycemia) D-Fructose Fruit juices , honey Hereditary fructose intolerance leads to fructose accumulation and hypoglycemia D-Galactose Hydrolysis of lactose Failure to metabolize leads to galactosemia and cataract 14
- 15. Biologically Important Sugar (Glucose) Derivatives 1. Sugar Acids 2. Sugar Alcohols 3. Deoxy Sugars 4. Amino Sugars 5. Glycosides. D. Classification Of Carbohydrates 1. Monosaccharide (simple sugars) 15
- 16. 1. Sugar Acids Oxidation of aldo group of sugars produces aldonic acids. Ketoses are not easily oxidized. Examples • Glucuronic acid or uronic acid :oxidation of terminal alcohol group (–OH sixth carbon atom of glucose • Uronic acids are components of mucopolysaccharides. Oxidation D-Glucose D-Glucoronic acid Sugar (Glucose) Derivatives D. Classification Of Carbohydrates (1. Monosaccharides) 16
- 17. 2. Sugar alcohols Reduction of aldose and keto groups of sugar produces polyhydroxy alcohols or polyols. Examples – Glycerol and inositol. – Glucose - sorbitol, – Galactose – galactitol – fructose are - sorbitol, Reduction Sugar (Glucose) Derivatives D. Classification Of Carbohydrates (1. Monosaccharides) 17
- 18. 3. Deoxy Sugars oxygen of a hydroxyl group is removed leaving hydrogen. Examples – Deoxyribose: in nucleic acids. – Fucose : in blood group substances. Removal of O from OH group Sugar (Glucose) Derivatives D. Classification Of Carbohydrates (1. Monosaccharides) 18
- 19. Substitution of OH with NH3 Sugar (Glucose) Derivatives 4. Aminosugars Those sugars in which an amino group is substituted for a hydroxyl group. Amino sugars are components of mucopolysaccharides, and antibiotics Examples D-glucosamine N-acetalglucosamine D. Classification Of Carbohydrates (1. Monosaccharides) 19
- 20. 2. Oligosaccharides : They consist of 2-10 monosaccharide units. Most important oligosaccharides are disaccharides 2a. Disaccharides : are condensation products of two monosaccharide units. E.g. are maltose =glucose + glucose sucrose = fructose + glucose lactose = galactose + glucose trehalose D. Classification Of Carbohydrates 20
- 21. 2a .Disaccharides : condensation products of two monosaccharide units. D. Classification Of Carbohydrates 21
- 22. Classification Of Carbohydrates 2a. Disaccharides Disaccharides of physiologic importance Sugar Source Clinical significance Maltose Digestion by amylase or hydrolysis of starch Lactose Milk, May occur in urine during pregnancy In lactase deficiency, Lactose intolerance causes malabsorption which leads to diarrhea and flatulence Sucrose Cane and beet sugar. Sorghum, Pineapple, carrot roots In sucrase deficiency, malabsorption leads to diarrhea and flatulence 22
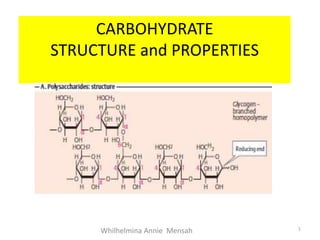
- 23. 3. Polysaccharides: Condensation products of more than ten monosaccharide units They are either linear α(1-4) linkage or branched α (1-6) linkage polymers. Examples starch, glycogen, cellulose, dextran and inulin and chitin. Proteoglycans and glycosaminoglycans. D. Classification Of Carbohydrates 23
- 24. D. Classification Of Carbohydrates 24 α (1-6) linkage α (1-4) linkage
- 25. 2b.Types of Polysaccharides are classified on the basis of the type of monosaccharide present. (a) Homopolysaccharides : They are entirely made up of one type of monosaccharides. On hydrolysis, they yield only one kind of monosaccharide E.g. starch, glycogen, cellulose, dextran and inulin and chitin. (b) Heteropolysaccharides : They are made up of more than one type of monosaccharides. On hydrolysis they yield more than one type of monosaccharides. Eg. Proteoglycans and glycosaminoglycans. D. Classification Of Carbohydrates 25
- 26. 2b. b.Heteropolysaccharides Types Proteoglycans (Mucopolysaccharides) : consist of protein with a repeating polysaccharide unit. Glycoproteins : Proteins which has less than 100 saccharide residues have been added. Glycosaminoglycans: a proteoglycan with the protein portion removed D.Classification Of Carbohydrates 26
- 27. 2b.b.Heteropolysaccharides Important proteoglycans or glycosaminoglycans (GAGs) Hyaluronic Acids: found on cell surfaces, synovial fluid and vitrous humour Classification Of Carbohydrates 27
- 28. 2b.b.Heteropolysaccharides Important proteoglycans or glycosaminoglycans (GAGs) Chrondroitin Sulfate: found in cartilage, tendon, bone Classification Of Carbohydrates 28
- 29. 2b.b. Heteropolysaccharides Important proteoglycans or glycosaminoglycans (GAGs) Heparin : Anti-coagulant present in blood Classification Of Carbohydrates 29
- 30. 1. Optical isomerism 2. Optical activity 3. Epimerism 4. Hemiacetal and hemiketal formation 5. L and D forms 6. Anomers 7. Mutarotaion 8. Formation of glycosidic bonds 9. Reducing properties E. Properties of Monosaccharides 30
- 31. 1.Optical isomerism Enantiomers : They are mirror images. E. Properties of Monosaccharides 31
- 32. 2. Optical activity : the ability of a sugar to rotate plane polarized light Orientations – right - dextrorotatory – left -levorotatory. – ‘+’ sign or ‘d’ indicates dextrorotation – ‘–’ sign or 1 indicates levorotation of a sugar. Example D-glucose which is dextrorotatory is designated as D(+)glucose D-fructose, which is levorotatory is designated as D(–) fructose. Note: The letter ‘D’does not indicate whether a given sugar is dextro or levorotatory E. Properties of Monosaccharides 32
- 33. 3. Epimers Isomers differing as a result of variations in configuration of the OH and H on carbon atoms 2, 3, and 4 of glucose. E. Properties of Monosaccharides 33
- 34. E. Properties of Monosaccharides 4. Hemiacetal or Hemiketal formation Hemiacetal formation • The linkage between aldehyde group and alcohol is called as ‘hemiacetal’ linkage. Forming 6 member pyranose ring Hemiacet al formationHemiacetal formation 34
- 35. E. Properties of Monosaccharides 4. Hemiacetal or Hemiketal formation Hemiketal formation • The linkage between keto group and alcohol is called as ‘ketal linkage. Forming 5 member furanose ring Hemiketal formation 35
- 36. PROPERTIES and STRUCTURE 5. L and D forms D sugars :compounds that have the last asymmetrical carbon in the same orientation as D-glyceraldehyde L sugars: Related to L-glyceraldehyde E. Properties of Monosaccharides 36
- 37. 6. Anomers (α & β-form ) Monosaccharides that differ in configuration of OH groups on carbonyl carbon (C-1) or anomeric carbon. α-form - Right β-form - Left E. Properties of Monosaccharides 37
- 38. 7. Mutarotation Monosaccharides containing asymmetric carbon atom rotate plane polarized light. The change in optical rotation when either form of glucose is allowed to stand in solution is mutarotation Example : exchange between α and β forms E. Properties of Monosaccharides 38
- 39. 8. Glycosidic bond/ linkage (Glycosides) Types O- glycosidic When hydroxyl group on anomeric carbon of a sugar reacts with an alcohol ( or another sugar). They exist in disaccharides, oligosaccharides and polysaccharides N- glycosidic : hydroxyl group on anomeric carbon of sugar reacts with an amine. They are present in nucleotides, RNA and DNA. Properties of Monosaccharides 39
- 40. – α bond : if the anomeric hydroxyl is in is below the Hydrogen – β bond : if the anomeric hydroxyl is above the hydrogen 7. Types of Glycosidic bond/ linkage Types Properties of Monosaccharides 40
- 41. 9.Reducing properties Glucose and other sugars capable of reducing ferric or cupric ion in solution are called reducing sugars. They reduce and colour Benedicts and Fehlings solution with the aldehyde group of the sugar E. Properties of Monosaccharides 41
- 42. A.Biological and medical importance Functions : energy source, storage, structural, part of DNA & RNA, component of drugs B. Chemical nature Ketose and hexoses Empirical formular - ( CH2O)n C. Structure Fisher projection Haworth projection (Ring forms – pyranose and furanose) Comformations (boat and chair) Lecture Summary 42
- 43. D. Classification of carbohydrates Monosaccharides Hexoses of physiologic importance : glucose , fructose , galactose Important sugar derivatives : sugar acids, sugar alcohols, deoxy sugars, amino sugars and glycosides Oligosaccharides Disaccharides of physiologic importance: lactose , sucrose, maltose Polysaccharides Homopolysaccharides : chitin, cellulose, glycogen, starch heteropolysaccharides : Proteoglycans, glycoproteins, GAGS Lecture Summary 43
- 44. E. Properties of monosaccharides Optical isomerism Optical activity Epimerism Hemiacetal and hemiketal formation L and D forms Anomers Mutarotaion Formation of glycosidic bonds Lecture Summary 44
- 45. 45
- 46. 46
- 47. 47
