Post translational modification by Prof Viyatprajna Acharya
•Als PPTX, PDF herunterladen•
0 gefällt mir•188 views
This presentation is targeted for MBBS, MD, BDS students depicting the process of protein synthesis and its biomedical significance
Melden
Teilen
Melden
Teilen
Empfohlen
Weitere ähnliche Inhalte
Was ist angesagt?
Was ist angesagt? (18)
A quick revision of Carbohydrate metabolism with case- based discussions and ...

A quick revision of Carbohydrate metabolism with case- based discussions and ...
Ähnlich wie Post translational modification by Prof Viyatprajna Acharya
Ähnlich wie Post translational modification by Prof Viyatprajna Acharya (20)
Post translational modification/PTM/protein modifications

Post translational modification/PTM/protein modifications
Co and post translationational modification of proteins

Co and post translationational modification of proteins
INTRODUCTION TO BIOTRANSFORMATION OF DRUG (METABOLISM OF PHENYTOIN AND CODEINE)

INTRODUCTION TO BIOTRANSFORMATION OF DRUG (METABOLISM OF PHENYTOIN AND CODEINE)
Amino acids and Protein chemistry by Dr. Anurag Yadav

Amino acids and Protein chemistry by Dr. Anurag Yadav
Functional role and activation of protein synthesis in insects

Functional role and activation of protein synthesis in insects
Mehr von Prof Viyatprajna Acharya
This presentation targets MBBS, MD and BDS students and tells about the checkpoints of genetic expression. Regulation of gene expression by Prof Viyatprajna Acharya, KIMS, Bhubaneswar

Regulation of gene expression by Prof Viyatprajna Acharya, KIMS, BhubaneswarProf Viyatprajna Acharya
Mehr von Prof Viyatprajna Acharya (20)
Talk on Cancer awareness on World cancer day-at JCI Talcher meet

Talk on Cancer awareness on World cancer day-at JCI Talcher meet
Cancer-an overview by Prof Viyatprajna Acharya, KIMS, Bhubaneswar

Cancer-an overview by Prof Viyatprajna Acharya, KIMS, Bhubaneswar
Regulation of gene expression by Prof Viyatprajna Acharya, KIMS, Bhubaneswar

Regulation of gene expression by Prof Viyatprajna Acharya, KIMS, Bhubaneswar
Genetic code and Translation by Prof Viyatprajna Acharya, KIMS Bhubaneswar

Genetic code and Translation by Prof Viyatprajna Acharya, KIMS Bhubaneswar
Mutation by Prof V P Acharya, Biochemistry, KIMS Bhubaneswar

Mutation by Prof V P Acharya, Biochemistry, KIMS Bhubaneswar
Kürzlich hochgeladen
Kürzlich hochgeladen (20)
Call Girls Kochi Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Kochi Just Call 8250077686 Top Class Call Girl Service Available
Call Girls Service Jaipur {9521753030} ❤️VVIP RIDDHI Call Girl in Jaipur Raja...

Call Girls Service Jaipur {9521753030} ❤️VVIP RIDDHI Call Girl in Jaipur Raja...
Call Girls Agra Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Agra Just Call 8250077686 Top Class Call Girl Service Available
Call Girls Bangalore Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Bangalore Just Call 8250077686 Top Class Call Girl Service Available
Top Rated Bangalore Call Girls Mg Road ⟟ 9332606886 ⟟ Call Me For Genuine S...

Top Rated Bangalore Call Girls Mg Road ⟟ 9332606886 ⟟ Call Me For Genuine S...
Top Rated Hyderabad Call Girls Erragadda ⟟ 6297143586 ⟟ Call Me For Genuine ...

Top Rated Hyderabad Call Girls Erragadda ⟟ 6297143586 ⟟ Call Me For Genuine ...
Night 7k to 12k Chennai City Center Call Girls 👉👉 7427069034⭐⭐ 100% Genuine E...

Night 7k to 12k Chennai City Center Call Girls 👉👉 7427069034⭐⭐ 100% Genuine E...
Best Rate (Guwahati ) Call Girls Guwahati ⟟ 8617370543 ⟟ High Class Call Girl...

Best Rate (Guwahati ) Call Girls Guwahati ⟟ 8617370543 ⟟ High Class Call Girl...
Call Girls Bareilly Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Bareilly Just Call 8250077686 Top Class Call Girl Service Available
College Call Girls in Haridwar 9667172968 Short 4000 Night 10000 Best call gi...

College Call Girls in Haridwar 9667172968 Short 4000 Night 10000 Best call gi...
Call Girls Dehradun Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Dehradun Just Call 9907093804 Top Class Call Girl Service Available
Call Girls Nagpur Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Nagpur Just Call 9907093804 Top Class Call Girl Service Available
Premium Bangalore Call Girls Jigani Dail 6378878445 Escort Service For Hot Ma...

Premium Bangalore Call Girls Jigani Dail 6378878445 Escort Service For Hot Ma...
Top Quality Call Girl Service Kalyanpur 6378878445 Available Call Girls Any Time

Top Quality Call Girl Service Kalyanpur 6378878445 Available Call Girls Any Time
(👑VVIP ISHAAN ) Russian Call Girls Service Navi Mumbai🖕9920874524🖕Independent...

(👑VVIP ISHAAN ) Russian Call Girls Service Navi Mumbai🖕9920874524🖕Independent...
Best Rate (Patna ) Call Girls Patna ⟟ 8617370543 ⟟ High Class Call Girl In 5 ...

Best Rate (Patna ) Call Girls Patna ⟟ 8617370543 ⟟ High Class Call Girl In 5 ...
VIP Service Call Girls Sindhi Colony 📳 7877925207 For 18+ VIP Call Girl At Th...

VIP Service Call Girls Sindhi Colony 📳 7877925207 For 18+ VIP Call Girl At Th...
Call Girls Ludhiana Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Ludhiana Just Call 9907093804 Top Class Call Girl Service Available
Best Rate (Hyderabad) Call Girls Jahanuma ⟟ 8250192130 ⟟ High Class Call Girl...

Best Rate (Hyderabad) Call Girls Jahanuma ⟟ 8250192130 ⟟ High Class Call Girl...
Call Girls Varanasi Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Varanasi Just Call 8250077686 Top Class Call Girl Service Available
Post translational modification by Prof Viyatprajna Acharya
- 1. POST-TRANSLATIONAL MODIFICATION Prof (Dr) V P AcharyaMD,PhD KIMS & PBH Hospital, Bhubaneswar
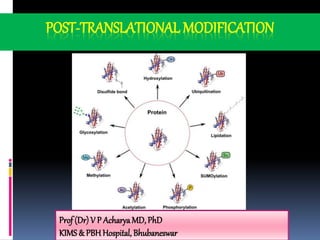
- 2. Post-translational modification of Polypeptide Protein folding Proteolytic cleavage Intein splicing Covalent modification Different structures Insulin, Zymogens Inteins removed & Exteins spliced a. Phosphorylation b. Hydroxylation c. Glycosylation d. Carboxylation e. Ubiquitylation
- 3. Amino terminal and carboxyl terminal modifications 1st residue- N- formylmethionine (bacteria) or Met (eukaryotes) Formyl group and amino terminal residues is mostly removed 50% eukaryotic proteins- NH3 group of amino terminal residue is N-acetylated Carboxyl terminal also may be modified 15-30 residues at the amino-terminal act as signal sequences to direct a protein to its target– later they are removed
- 4. Modifications of individualAA residues Phosphorylation- -OH groups of Ser,Thr ,Tyr are enzymatically phosphorylated by ATP– adds negative charge to the protein Casein binds to Ca+2 due to this negative charge due to phosphoserine groups Phosphorylation and dephosphorylation regulate many enzymes and proteins Carboxylation - γ- carboxylation of Prothrombin- requiresVitamin K Glycosylation - N-linked oligosaccharides (attached to Asn) and O- linked oligosaccharides (Ser/Thr residues) Isoprenylation - thioether bond formed between Cys and isoprene
- 5. Additionof prosthetic groups Prosthetic groups bound to proteins by covalent groups Hb / Cyt C- Heme group Biotin- Acetyl CoA-carboxylase
- 6. Proteolytic processing- Proinsulin, viral proteins, proteases Formation of disulfide cross-links- Insulin
- 7. Inhibitors ofTranslation A. Reversible inhibitor a.Tetracyclin– Binds to 30s ribosome ↓ Inhibit attachment of aminoacyl tRNA to the A site (bacteriostatic) b. Chloramphenicol– Inhibits peptidyl transferase- prevents elongation of peptide chain c. Erythromycin & Clindamycin– Prevent translocation by binding to 50s subunit of bacterial ribosome
- 8. B.Irreversibleinhibitor Streptomycin and Aminoglycosides—Bind to 30s ribosomal sub- unit Low conc.- Misreading of protein ↓ Useless bacterial proteins Pharma. Conc.- Inhibit IC synthesis ↓ Protein synthesis inhibited
- 9. C. Inhibitors in mammals 1. Puromycin– Structural analogue of tyrosinyl t- RNA 2. Cycloheximide– Inhibits peptidyl transferase 3. Diphtheria toxin—Inactivation of EF-2 by attachment of ADP to EF-2 4. Ricin– Inactivates 28s rRNA
- 10. KNOWLEDGE HAS NO COPYRIGHT
- 12. For more ppt onMedicalBiochemistrypleasevisit www.vpacharya.com
Hinweis der Redaktion
- Protein splicing is an intramolecular reaction of a particular protein in which an internal protein segment (called an intein) is removed from a precursor protein with a ligation of C-terminal and N-terminal external proteins (called exteins) on both sides. The splicing junction of the precursor protein is mainly a cysteine or a serine, which are amino acids containing a nucleophilic side chain. The protein splicing reactions which are known now do not require exogenous cofactors or energy sources such as adenosine triphosphate (ATP) or guanosine triphosphate (GTP). Normally, splicing is associated only with pre-mRNA splicing. This precursor protein contains three segments—an N-extein followed by the intein followed by a C-extein. After splicing has taken place, the resulting protein contains the N-extein linked to the C-extein; this splicing product is also termed an extein.
