Molecular geometry cheat sheet
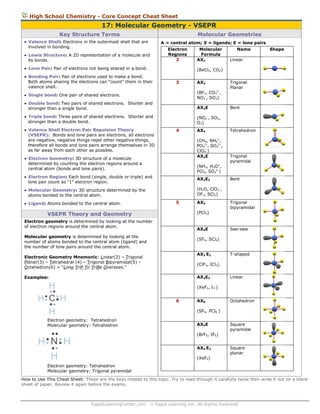
- 1. High School Chemistry - Core Concept Cheat Sheet 17: Molecular Geometry - VSEPR Key Structure Terms Molecular Geometries Valence Shell: Electrons in the outermost shell that are involved in bonding. A = central atom; X = ligands; E = lone pairs Electron Regions 2 Lewis Structure: A 2D representation of a molecule and its bonds. Lone Pair: Pair of electrons not being shared in a bond. Molecular Formula AX2 Name Shape Linear (BeCl2, CO2) Bonding Pair: Pair of electrons used to make a bond. Both atoms sharing the electrons can “count” them in their valence shell. 3 AX3 Single bond: One pair of shared electrons. (BF3, CO32-, NO3-, SO3) Double bond: Two pairs of shared electrons. Shorter and stronger than a single bond. AX2E Triple bond: Three pairs of shared electrons. Shorter and stronger than a double bond. Trigonal Planar (NO2-, SO2, O3) Valence Shell Electron Pair Repulsion Theory (VSEPR): Bonds and lone pairs are electrons, all electrons are negative, negative things repel other negative things, therefore all bonds and lone pairs arrange themselves in 3D as far away from each other as possible. 4 AX4 (CH4, NH4+, PO43-, SO42-, ClO4-) AX3E Electron Geometry: 3D structure of a molecule determined by counting the electron regions around a central atom (bonds and lone pairs). (NH3, H3O+, PCl3, SO32-) Electron Region: Each bond (single, double or triple) and lone pair count as “1” electron region. AX2E2 Molecular Geometry: 3D structure determined by the atoms bonded to the central atom. Bent Tetrahedron Trigonal pyramidal (H2O, ClO2-, OF2, SCl2) 5 Ligand: Atoms bonded to the central atom. AX5 (PCl5) VSEPR Theory and Geometry Electron geometry is determined by looking at the number of electron regions around the central atom. AX4E Molecular geometry is determined by looking at the number of atoms bonded to the central atom (ligand) and the number of lone pairs around the central atom. Trigonal bipyramidal See-saw (SF4, SCl4) AX3 E2 Electronic Geometry Mnemonic: Linear(2) – Trigonal Planar(3) – Tetrahedral (4) – Trigonal Bipyramidal(5) – Octahedron(6) = “Long TriP To TriBe Overseas.” T-shaped (ClF3, ICl3) AX2E3 Examples: H H C H H Bent Linear (XeF2, I3-) 6 AX6 Octahedron (SF6, PCl6-) Electron geometry: Tetrahedron Molecular geometry: Tetrahedron AX5E (BrF5, IF5) H N H AX4 E2 H (XeF4) Square pyramidal Square planar Electron geometry: Tetrahedron Molecular geometry: Trigonal pyramidal How to Use This Cheat Sheet: These are the keys related to this topic. Try to read through it carefully twice then write it out on a blank sheet of paper. Review it again before the exams. RapidLearningCenter.com Rapid Learning Inc. All Rights Reserved
