5_Pressure_Measurement.pdf
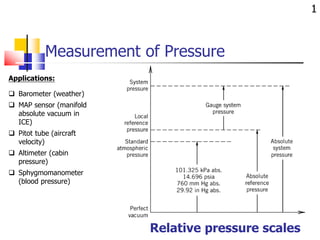
- 1. Measurement of Pressure 1 Relative pressure scales Applications: Barometer (weather) MAP sensor (manifold absolute vacuum in ICE) Pitot tube (aircraft velocity) Altimeter (cabin pressure) Sphygmomanometer (blood pressure)
- 2. Fortin Barometer In the Fortin barometer, the level of mercury in the glass at the bottom of the barometer cistern is adjusted to a scale zero, known as the zeroing (Ivory pointer) point, each time a reading is to be taken. The level of mercury in the column is then read against the scale, using a vernier adjustment for extra accuracy. Air is evacuated from the top of the tube of mercury and the lower end is fixed in the cistern containing the reservoir of mercury. The Fortin barometer is simple to use as it has a clear easy-to-read linear scale. These barometers can be mounted on a wall or suitable pillar. 2
- 3. Manometer Manometer is the simplest device for measuring static (differential) pressure. It contains water/mercury or other suitable fluid(s) inside the tube. When a pressure line is connected to one column of manometer, the fluid in the column is forced down and the fluid in the other column rises. By measuring the difference in height of the fluid in the two columns, the pressure of the inlet can be expressed in some height of the manometric fluid. 3
- 4. Manometer Inclined tube manometer U-tube manometer 4
- 5. Manometer If a manometer contains multiple fluids, then the pressure is determined by equating pressures at the same vertical height of continuous columns of each fluid. 5
- 6. Bourdon Tube Pressure Gauge ‘Aneroid’ (meaning “without fluid”) gauges are based on an elastic metallic pressure-sensing element that flexes elastically under the effect of a pressure difference across the element. This fact distinguished these gauges from the hydrostatic gauges. They are also called ‘mechanical’ gauges. However, aneroid gauges can be used to measure the pressure of a liquid as well as a gas. The pressure sensing element may be a Bourdon tube, a diaphragm, a capsule, or a set of bellows, which will change shape in response to the pressure of the region in question. The deflection of the pressure sensing element may be read by a linkage connected to a needle, or it may be read by a secondary transducer. The common secondary transducers in modern vacuum gauges measure a change in resistance/capacitance/inductance due to the mechanical deflection. 6
- 7. Elastic Elements used as Pressure Sensors 7
- 8. Used for static calibration Deadweight pressure tester 8
- 9. Diaphragm Pressure Transducer with Strain Gauge 9
- 11. Electric Resistance Pressure Gauge (Coil) The sensing element consists of a loosely wound coil of relatively fine wire which compresses when subjected to a high pressure. The change in length and cross-section of the wire affect its electrical resistance when pressure applied at a rate determined by the bulk modulus of the material. 11
- 12. Electric Resistance Pressure Gauge (Others) 12
- 13. Electric Resistance Pressure Gauge (Others) 13
- 15. LVDT Pressure Transducer (With Bourdon Tube) LVDT can be combined with a Bourdon tube LVDT converts displacements into an electrical signal The signal can be displayed on an electrical device calibrated in terms of pressure 15
- 16. LVDT Pressure Transducer (Others) LVDT can be combined with bellows LVDT can also be used with diaphragms LVDT with bellows LVDT with diapharms 16
- 17. 17 McLeod Gauge (10-4 to 10-6 torr, i.e., 10 mPa to 0.1 mPa) One variation of the McLeod gauge is shown in Figure (a), showing its sensing position. In this sensing position, the gauge is connected directly to the low-pressure source. The measuring position is shown in Figure (b).
- 18. McLeod Gauge (10-4 to 10-6 torr, i.e., 10 mPa to 0.1 mPa) 18 The McLeod gauge is a pressure-measuring instrument and laboratory reference standard used to establish gas pressures in the sub-atmospheric range of 1 mm Hg abs down to 0.1 mm Hg abs. The glass tubing is arranged so that a sample of the gas at an unknown low pressure 𝒑𝟏 can be trapped by inverting the gauge from the sensing position (a) to that of the measuring position (b). In this way, the gas trapped within the capillary is isothermally compressed by a rising column of mercury. At the equilibrium and measuring position, the capillary pressure, 𝒑𝟐, is related to the unknown gas pressure 𝒑𝟏 by Boyle’s law as 𝒑𝟐 = 𝒑𝟏 ∀𝟏 ∀𝟐 where ∀𝟏 is the gas volume of the gauge in sensing position which is a constant for a gauge at any pressure, and ∀𝟐 is the capillary volume found in the measuring position.
- 19. McLeod Gauge (10-4 to 10-6 torr, i.e., 10 mPa to 0.1 mPa) 19 But ∀𝟐= 𝑨𝒚, where 𝑨 is the known cross-sectional area of the capillary and 𝑦 is the vertical length of the capillary occupied by the gas. With 𝛾 as the specific weight of the mercury, the difference in pressures is related by 𝒑𝟐 − 𝒑𝟏 = 𝜸𝒚 such that the unknown gas pressure is just a function of 𝑦, i.e., 𝒑𝟏 = 𝜸𝑨𝒚𝟐 /(∀𝟏 − 𝑨𝒚) In practice, a commercial McLeod gauge has the capillary etched and calibrated to indicate either pressure, 𝒑𝟏, or its equivalent head, 𝒑𝟏/𝒈, directly. The technique is very slow and unsuited to continual monitoring, but is capable of good accuracy.
- 20. Pirani Thermal-conductivity Gauge (10-3 to 10 torr) 20 A Pirani gauge consist of a metal wire open to the pressure being measured. The wire is heated by a current flowing through it and cooled by the gas surrounding it. If the gas pressure is reduced, the cooling effect will decrease, hence the equilibrium temperature of the wire will increase. The resistance of the wire is a function of its temperature: by measuring the voltage across the wire and the current flowing through it, the resistance (and so the gas pressure) can be determined. At low pressures the effective thermal conductivity of gases decreases with pressure. The Pirani gauge is a device which measures the pressure through the change in thermal conductance of the gas.
- 21. Ionization Gauge (10-10 to 10-3 torr) 21 Heated cathode emits electrons into the vacuum, where they collide with gas molecules to create ions. These positively charged ions are accelerated toward a collector where they create a current in a conventional ion gauge detector circuit. The amount of current formed is proportional to the gas density or pressure.
- 22. For moderate pressure • Manometers • Elastic elements (diaphragm, bellows, capsules, tubes, spiral helix, etc.) For high pressure (>1000 atm) • Electrical resistance/capacitance/inductive (LVDT)/magnetic pressure transducers • Piezoelectric/optical/resonant pressure transducer For very low pressure (< 0.001 atm i.e., 1/760 atm which is 1 torr ) • McLeod gauge • Pirani gauge • Ionisation gauge 22 Selection of Pressure Measuring Instrument
- 23. Selection of Pressure Measuring Instrument One must understand the pressure range, accuracy and the response time of the instrument to be used. Parameters such as hysteresis and stability should be obtained from the manufacturer’s specification. Other aspects such as cost, maintenance and lifetime expectancy should also be considered. The liquid in the tube must not interact with the pressure gas (causing contamination / evaporation / condensation). 23
- 24. Pressure in a Moving Fluid Streamline flow over a bluff body 24
- 25. Pitot Tube Used for Static Pressure 25
- 26. Fluid Pressure and Velocity Measurement Pitot static pressure probe 26
- 27. Fluid Pressure and Velocity Measurement Total pressure: Dynamic pressure: Velocity of fluid: 27 Note that 𝜌 in the equation is the density of the flowing fluid, not the manometric fluid. [Manometric fluid is used to calculate the differential pressure in the manometer]
- 28. Fluid Pressure and Velocity Measurement Velocity of fluid: 28 The manometer connected to the Pitot tube of an aircraft records the stagnation pressure as 0.77 m of mercury. At the given altitude, the air density is measured as 1.13 kg/m3 and the static pressure is measured as 90 kPa. What is the aircraft velocity in km/h? 𝑉 ∞ = 2𝑔 𝑝𝑡−𝑝𝑥 𝛾 = 2 𝑝𝑡−𝑝𝑥 𝜌 = 2 0.77×13.6×9810−90000 1.13 = 150 𝑚/𝑠 = 540 𝑘𝑚/ℎ Note that in case of closed channel (e.g., pipe) flow, the volumetric flow-rate can also be measured using the pitot-tube if the cross-section of the channel is known.