Marcus theory
- 1. Organic Pedagogical Electronic Network Marcus Theory Elizabeth Greenhalgh, Amanda Bischoff, and Matthew Sigman University of Utah
- 2. Describing Electron Transfer Reactions Marcus, R. A. The Nobel Prize in Chemistry 1992 1992, 69-92 Anslyn, E. V.; Dougherty, D.A (2006) Modern Physical Organic Chemistry. University Science Books Marcus, R. A. The Journal of Chemical Physics 1956, 24(5), 966-978 Transition State Theory (TST) Overview: • Developed by Henry Eyring in the 1930s • Describes reaction rates focusing on geometry of the transition state at the top of the energy barrier • Effective for describing bond breakage/bond formation The Problem: Reactions that do not involve bond breakage/formation, i.e. electron transfer reactions, involve little nuclear movement in the transition state (rate of electron transfer is faster than rate of molecular vibrations); thus, TST fails and this necessitates a different model. Background: Morse potentials Morse potentials describe potential energy as a function of bond distance. Marcus theory focuses on a parabolic approximation of the boxed portion. Frank-Condon principle In an electronic transition between two reactants, the solvent molecules do not have time to rearrange; thus, the atomic configuration and total energy of the system remain the same; only the electronic state changes. Quantum Tunneling Some small particles (electrons, hydrogen atoms, etc.) can tunnel through energy barriers rather than going over them. This complicates free energy calculations.
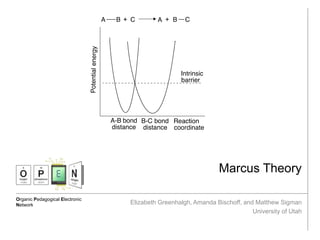
- 3. Marcus Theory and Implications Marcus, R. A. The Nobel Prize in Chemistry 1992 1992, 69-92 Anslyn, E. V.; Dougherty, D.A (2006) Modern Physical Organic Chemistry. University Science Books Why does Marcus theory work when TST doesn’t? TST focuses on orientation of reactants, while Marcus theory accounts for the many configurations of the solvent surrounding the inner coordination sphere; Marcus theory finds the potential curves for different orientations of the solvent molecules, and the point of intersection is the point at which electron transfer occurs. Marcus Theory Overview of Marcus Theory: • Developed by Rudolph A. Marcus in the 1950s • Uses Morse potentials to describe reaction coordinates • This theory won the Nobel Prize in chemistry in 1992 Marcus Theory and Exergonicity After aligning the Morse potentials along the intrinsic barrier, the product potential is adjusted to compensate for the free energy change of the reaction. The intersection of the curves defines the energy of the transition state. A tunneling term is also included. As the reaction becomes more exergonic, the energy of the transition state lowers; but, as it becomes still more exergonic, the energy of the transition state rises! The region beyond the zero point is called the “Marcus inverted region.”
- 4. Marcus Theory to Predict Hydrogen Atom Transfer Mayer, J. Understanding Hydrogen Atom Transfer: From Bond Strengths to Marcus Theory, Acc. Chem. Res. 2011 44 (1), 36-46. A H + A A + H A kAH/A (3) The simplest form of the Marcus equation for electron transfer (eq 1) predicts the reaction barrier (∆G‡) from the reaction driving force (∆G°) and intrinsic barrier (λ) which is the energy required to reorganize the reactants and surrounding solvent without electron transfer. This equation, with a few assumptions, can be rearranged to the Marcus cross relation (eq 2). The kinetic information is primarily in the rate constants for the respective hydrogen-atom self exchange reactions (such as eq 3). Tests of the Marcus cross relation for HAT: log/log plot of observed versus calculated HAT rate constant for a number of metal complexes reacting with various substrates. The diagonal line illustrates kobs = kcalc. The estimated errors on kcalc are typically ±1 log unit; they are larger for MeCN reactions of Ru(O)bpy2py2+ because the BDFE is only available in H2O, and smaller in three cases where KAH/B was measured directly.
- 5. Hydrogen Atom Transfer Continued Schematic free energy surface for the reaction shown above through H bonding intermediates. HAT from FeII(H2bip) to the stable nitroxyl radical TEMPO is very unusual in that it becomes faster at lower temperatures, with ∆H‡ = -2.7 ± 0.4 kcal mol-1. The figure below shows the cross relation quantitatively predicts the cross rate constants and the negative temperature dependence. The most temperature dependent parameter is keq, as the reaction is more favorable at lower temperatures causing the Marcus cross relation to have a temperature dependence as well. Mayer, J. Understanding Hydrogen Atom Transfer: From Bond Strengths to Marcus Theory, Acc. Chem. Res. 2011 44 (1), 36-46.
- 6. Problems 1. What does Marcus theory describe that transition state theory doesn’t? a. Quantum tunneling b. Electron transfer reactions c. Transition state energies d. All of the above 2. Which pair of potential curves would you expect to result in the reaction with the fastest rate?
- 7. Problems 3. Does the box on the adjacent diagram represent the intrinsic barrier, the transition state, or both? What is the difference between the two? 4. Marcus cross relation relates what values? a. Resonance b. Induction c. Reaction rates d. All of the above 5. Marcus theory is based on the quantum mechanical principle called_______. 6. True or False: Marcus cross relation can tell us information about temperature.
- 8. This work is licensed under a Creative Commons Attribution- ShareAlike 4.0 International License. Contributed by: Elizabeth Greenhalgh, Amanda Bischoff, and Matthew Sigman University of Utah, 2015