carbon compound
•Als PPT, PDF herunterladen•
2 gefällt mir•2,837 views
carbon compound
Melden
Teilen
Melden
Teilen
Weitere ähnliche Inhalte
Was ist angesagt?
Was ist angesagt? (20)
Structures, Names and Physical Properties of Alcohol

Structures, Names and Physical Properties of Alcohol
Andere mochten auch
Andere mochten auch (20)
elimination reaction, addition reaction, oxydation reactions

elimination reaction, addition reaction, oxydation reactions
Présentation de la plate-forme d'éco-conception CORINE

Présentation de la plate-forme d'éco-conception CORINE
Animation obtention, conversion et séparation des aromatiques

Animation obtention, conversion et séparation des aromatiques
Zugriffszahlen –und dann? Das Projekt OA-Statistik

Zugriffszahlen –und dann? Das Projekt OA-Statistik
Nutzungsstatistiken für Repositorien - das Projekt OA-Statistik

Nutzungsstatistiken für Repositorien - das Projekt OA-Statistik
Ähnlich wie carbon compound
Ähnlich wie carbon compound (20)
CHAPTER 3 CHM 258 - Introduction to Organic Chemistry. Alkenes and cycloalkenes

CHAPTER 3 CHM 258 - Introduction to Organic Chemistry. Alkenes and cycloalkenes
Mehr von Miz Malinz
Mehr von Miz Malinz (20)
The Elephant Man (Chapter Summaries, Setting & Characters)

The Elephant Man (Chapter Summaries, Setting & Characters)
Kürzlich hochgeladen
Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...

Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...christianmathematics
Mattingly "AI & Prompt Design: The Basics of Prompt Design"

Mattingly "AI & Prompt Design: The Basics of Prompt Design"National Information Standards Organization (NISO)
Kürzlich hochgeladen (20)
Ecosystem Interactions Class Discussion Presentation in Blue Green Lined Styl...

Ecosystem Interactions Class Discussion Presentation in Blue Green Lined Styl...
A Critique of the Proposed National Education Policy Reform

A Critique of the Proposed National Education Policy Reform
Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...

Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...
Web & Social Media Analytics Previous Year Question Paper.pdf

Web & Social Media Analytics Previous Year Question Paper.pdf
IGNOU MSCCFT and PGDCFT Exam Question Pattern: MCFT003 Counselling and Family...

IGNOU MSCCFT and PGDCFT Exam Question Pattern: MCFT003 Counselling and Family...
Measures of Dispersion and Variability: Range, QD, AD and SD

Measures of Dispersion and Variability: Range, QD, AD and SD
Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...

Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...
Beyond the EU: DORA and NIS 2 Directive's Global Impact

Beyond the EU: DORA and NIS 2 Directive's Global Impact
social pharmacy d-pharm 1st year by Pragati K. Mahajan

social pharmacy d-pharm 1st year by Pragati K. Mahajan
Mattingly "AI & Prompt Design: The Basics of Prompt Design"

Mattingly "AI & Prompt Design: The Basics of Prompt Design"
carbon compound
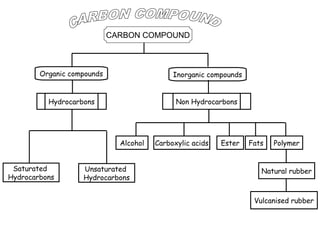
- 1. CARBON COMPOUND Organic compounds Inorganic compounds Hydrocarbons Non Hydrocarbons Alcohol Saturated Hydrocarbons Unsaturated Hydrocarbons Carboxylic acids Ester Fats Polymer Natural rubber Vulcanised rubber
- 2. CO2 + H2O C + CO + H2O Sufficient Insufficient Oxygen Oxygen Combustion ( CnH2n+2 ) H YDR OGE NA ALKANE TI O N Ethane Chemical Reaction CH3CH3 H2 / Ni,1 Making ethanol by Fermentation process HYDRATION ALKENE 80° c ( CnH2n ) H2O,300°C,60atm,H3PO4 ( Catalytic Hydration ) Ethene ALCOHOL Ethanol DEHYDRATION CH2= CH2 Ch re emi ac ca tio l n Addition CH3CH2OH Cold acidified Chlorination dilute KMnO4 Chloromethane ethanadiol ESTERIFICATION OXYDATION CARBOXYLIC ACID Ethanoic acid H2O(g) C2H4 ethene HBr C + CO + H2O CH3COOH Un Complete combustion Limited O2 Br2 OXYDATION Refluxing ethanol with an oxidising agent acidified K2Cr2O7 / KMnO4 solution Dichloromethane Trichloromethane Addition hydrogen halides Reaction between molecule of carboxylic acid and molecules of alcohol in a present of concentrated H2SO4 ( CnH2n+1COOH ) CO2 + H2O Addition of hydroxyl group ESTER CH3COOC2H5 Excess, O2 of water Halogenation ( Methane) CH4 ( CnH2n+1COOCmH2m+1 ) Ethyl ethanoate Ethanol vapour is passed over a heated catalyst (porcelain chips,porous pot or Al2O3 Substitution Tetrachloromethane ( CnH2n+1OH ) ESTERIFICATION 1,2-dibromoethane Addition halogen
