Notechapter2
- 2. Diffusion of bromine gas Experiment diffusion of bromine in air The diffusion of bromine progresses Air colourless ______________________ spreads through the first cylinder
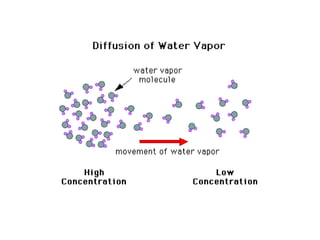
- 3. ___________________________ The reddish-brown vapor moves upward, slowly filling the ___________________ Reddish-brown vapour spreads through the __________________
- 4. Kinetic Theory of Matter The _________________ and _________________ of ______________________ in
- 8. ________ More than in _______ but less than in _______ _________ _________ contents of the particles Low _________ _________ _________ GAS LIQUIDS SOLIDS Properties
- 10. CHANGES IN THE STATE OF MATTER ___________________ ____________________ EVAPORATION or BOILING _____________________ _____________________
- 11. INTER-CONVERSION OF THE STATES OF MATTER
- 12. MELTING When a _________ is heated The particles in the solid __________________ _____________and _______________________ The particles vibrate faster as the __________ _________until the energy they gained is able to ____________________ that hold them at their _______________________ At this point the __________ becomes _________________ Process _______________ HEAT SOLID LIQUID The temperature at this point: _______________
- 14. FREEZING When a __________ is _____________ The particles in the liquid ______________ __________and ______________________ As the _____________ continues to ________, the particles continue to _________________ until they do not enough energy to ___________________ At this point the _____________ changes into a ________________ Process ________________ The temperature at this point: ______________________________
- 16. CHANGE IN THE STATE OF MATTER IN NAPHTHALENE HEATING CURVE OF NAPHTALENE Melting point -------- A Solid B C Start to melt Melt completely D
- 17. At point ____, naphthalene exists as ___________ When _______ is heated, ______________ is absorbed that cause particles to _______________ and _____________ ( This is why temperature ______________ from point _____ to point _____) At point ___, solid naphthalene begins to __________ During the ____________, the temperature of naphthalene does not rise even though heating continues because heat energy ____________ by the particles is used to _____________ the ____________________ so that _______________ can turn into ___________
- 18. This constant temperature: ____________________ of Naphthalene Both ______________ and ___________ are present At point _____, ________________ naphthalene has ____________ From point _____ to point ______, the particles in liquid naphthalene absorb heat energy and move faster ____________________
