04 Solutions
•Als PPT, PDF herunterladen•
0 gefällt mir•767 views
Melden
Teilen
Melden
Teilen
Empfohlen
Weitere ähnliche Inhalte
Was ist angesagt?
Was ist angesagt? (10)
Ähnlich wie 04 Solutions
Ähnlich wie 04 Solutions (20)
Periodic Properties Of Elements In The Periodic Table

Periodic Properties Of Elements In The Periodic Table
Periodic Properties Of Elements In The Periodic Table

Periodic Properties Of Elements In The Periodic Table
Periodic Properties Of Elements In The Periodic Table

Periodic Properties Of Elements In The Periodic Table
Periodic Properties Of Elements In The Periodic Table

Periodic Properties Of Elements In The Periodic Table
Periodic Properties Of Elements In The Periodic Table

Periodic Properties Of Elements In The Periodic Table
Periodic Properties Of Elements In The Periodic Table

Periodic Properties Of Elements In The Periodic Table
Periodic Properties Of Elements In The Periodic Table

Periodic Properties Of Elements In The Periodic Table
Periodic Properties Of Elements In The Periodic Table

Periodic Properties Of Elements In The Periodic Table
Periodic Properties Of Elements In The Periodic Table

Periodic Properties Of Elements In The Periodic Table
Periodic Properties Of Elements In The Periodic Table

Periodic Properties Of Elements In The Periodic Table
pdfslide.net_redox-reactions-and-electrochemistry-redox-reactions-galvanic-ce...

pdfslide.net_redox-reactions-and-electrochemistry-redox-reactions-galvanic-ce...
Kürzlich hochgeladen
Kürzlich hochgeladen (20)
Call Girls Jp Nagar Just Call 👗 7737669865 👗 Top Class Call Girl Service Bang...

Call Girls Jp Nagar Just Call 👗 7737669865 👗 Top Class Call Girl Service Bang...
Value Proposition canvas- Customer needs and pains

Value Proposition canvas- Customer needs and pains
Nelamangala Call Girls: 🍓 7737669865 🍓 High Profile Model Escorts | Bangalore...

Nelamangala Call Girls: 🍓 7737669865 🍓 High Profile Model Escorts | Bangalore...
Uneak White's Personal Brand Exploration Presentation

Uneak White's Personal Brand Exploration Presentation
Call Girls Service In Old Town Dubai ((0551707352)) Old Town Dubai Call Girl ...

Call Girls Service In Old Town Dubai ((0551707352)) Old Town Dubai Call Girl ...
The Abortion pills for sale in Qatar@Doha [+27737758557] []Deira Dubai Kuwait![The Abortion pills for sale in Qatar@Doha [+27737758557] []Deira Dubai Kuwait](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![The Abortion pills for sale in Qatar@Doha [+27737758557] []Deira Dubai Kuwait](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
The Abortion pills for sale in Qatar@Doha [+27737758557] []Deira Dubai Kuwait
Call Now ☎️🔝 9332606886🔝 Call Girls ❤ Service In Bhilwara Female Escorts Serv...

Call Now ☎️🔝 9332606886🔝 Call Girls ❤ Service In Bhilwara Female Escorts Serv...
Falcon's Invoice Discounting: Your Path to Prosperity

Falcon's Invoice Discounting: Your Path to Prosperity
Call Girls Ludhiana Just Call 98765-12871 Top Class Call Girl Service Available

Call Girls Ludhiana Just Call 98765-12871 Top Class Call Girl Service Available
Call Girls Electronic City Just Call 👗 7737669865 👗 Top Class Call Girl Servi...

Call Girls Electronic City Just Call 👗 7737669865 👗 Top Class Call Girl Servi...
JAYNAGAR CALL GIRL IN 98274*61493 ❤CALL GIRLS IN ESCORT SERVICE❤CALL GIRL

JAYNAGAR CALL GIRL IN 98274*61493 ❤CALL GIRLS IN ESCORT SERVICE❤CALL GIRL
Call Girls In Noida 959961⊹3876 Independent Escort Service Noida

Call Girls In Noida 959961⊹3876 Independent Escort Service Noida
Russian Call Girls In Rajiv Chowk Gurgaon ❤️8448577510 ⊹Best Escorts Service ...

Russian Call Girls In Rajiv Chowk Gurgaon ❤️8448577510 ⊹Best Escorts Service ...
Cheap Rate Call Girls In Noida Sector 62 Metro 959961乂3876

Cheap Rate Call Girls In Noida Sector 62 Metro 959961乂3876
Mysore Call Girls 8617370543 WhatsApp Number 24x7 Best Services

Mysore Call Girls 8617370543 WhatsApp Number 24x7 Best Services
Chandigarh Escorts Service 📞8868886958📞 Just📲 Call Nihal Chandigarh Call Girl...

Chandigarh Escorts Service 📞8868886958📞 Just📲 Call Nihal Chandigarh Call Girl...
04 Solutions
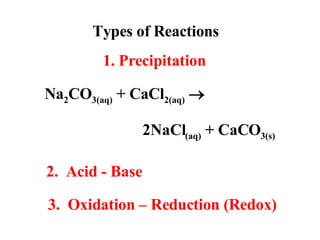
- 1. Types of Reactions Na 2 CO 3(aq) + CaCl 2(aq) 2NaCl (aq) + CaCO 3(s) 1. Precipitation 2. Acid - Base 3. Oxidation – Reduction (Redox)
- 2. Acids & Bases (Bronsted) Acid = Proton (H + ) donor Base = Proton acceptor Acid Properties: sour (vinegar and citrus fruit) turns litmus red
- 3. Strong Acids HCl, HNO 3 , H 2 SO 4 HCl (aq) H + (aq) + Cl - (aq) or ….. HCl + H 2 O (l) H 3 O + (aq) + Cl - (aq) There is no HCl present in solution proton hydronium ion monoprotic diprotic
- 4. Weak Acids HCN (aq) H + (aq) + CN - (aq) HCN, H + and CN - are present in solution H 2 O (l) H + (aq) + OH - (aq) All aqueous solutions will contain protons and hydroxide ions. Acetic acid = H(C 2 H 3 O 2 ) Phosphoric Acid = H 3 PO 4
- 5. Base Properties: bitter slippery turns litmus blue Strong bases: NaOH; KOH Weak bases: NH 3 , CO 3 2-
- 6. Neutralization Reactions Acid + Base salt + H 2 O HNO 3(aq) + KOH (aq) KNO 3(aq) + H 2 O (l) HCl (aq) + Na 2 CO 3(aq) KNO 3(aq) + CO 2(g) + H 2 O (l)
- 7. Oxidation Reduction Reactions (Redox) Transfer of electrons (between atoms in compounds) oxidation = loss of electrons reduction = gain of electrons oxidizing agent – gets reduced by accepting electrons reducing agent – gets oxidized by donating electrons
- 8. Oxidation State - Rules 1. 0 in elemental form (e.g. H 2 , O 2 , Zn (s) ) 2. Ion charge = oxidation state for ions (of elements ) 3. O = -2 (except in peroxides, e.g. H 2 O 2 ) 4. H = +1 (except as hydride in ionic cpd) 5. F = -1 (other halides = -1 if ionic cpd) 6. Sum of ox #s = charge of ion or 0 for neutral cpd
- 9. Oxidation-Reduction Reactions Any combustion reaction cpd of (CHO) + O 2 CO 2 + H 2 O example methane combustion …. Reaction of acid with metals …. 2HCl (aq) + Zn (s) ZnCl 2(aq) + H 2(g) __Al (s) + __Fe 2 O 3(s) __Fe (s) + __Al 2 O 3(s) + heat
- 11. Blood Alcohol Levels Legal limit = 0.1% by mass/(weight) What is approximate Molarity? ethanol ~ 46.0 g mol -1 How much dilution of 3.2% beer results in this? Why is this misleading?
- 12. Breath Analyzer ___C 2 H 6 O + ___ K 2 Cr 2 O 7 + ___ H 2 SO 4 ___C 2 H 4 O 2 + ___ Cr 2 (SO 4 ) 3 + ___ K 2 SO 4 + ___ H 2 O
- 13. Getting Mg from seawater 1. CaCO 3(s) CaO (s) + CO 2(g) 2. CaO (s) + H 2 O (l) + Ca 2+ (aq) + 2OH - (aq) 3. Mg 2+ (aq) + 2OH - (aq) MgOH 2(s) 4. MgOH 2(s) + HCl (aq) MgCl 2(aq) + 2H 2 O (l) 5. MgCl 2(l) + e - Mg (l) + Cl 2(g)