Polynuclear hydrocarbons module9
•
3 gefällt mir•229 views
Module 9 - Phenanthrene- Synthesis - Haworth synthesis -Reactions- Structural elucidation of phenanthrene
Melden
Teilen
Melden
Teilen
Downloaden Sie, um offline zu lesen
Empfohlen
Empfohlen
Weitere ähnliche Inhalte
Was ist angesagt?
Was ist angesagt? (20)
Unit iii heterocyclic compounds as per PCI Syllabus of POC-III

Unit iii heterocyclic compounds as per PCI Syllabus of POC-III
Synthesis and reactions of Seven membered heterocycle-Azepines

Synthesis and reactions of Seven membered heterocycle-Azepines
Imidazole - Synthesis of Imidazole - Reactions of Imidazole - Medicinal uses ...

Imidazole - Synthesis of Imidazole - Reactions of Imidazole - Medicinal uses ...
Phenols: methods of preparation, chemical reaction

Phenols: methods of preparation, chemical reaction
Heterocyclic compounds classification nomenclature

Heterocyclic compounds classification nomenclature
Heterocyclic compounds - pyrrole - synthesis of pyrrole - characteristic rea...

Heterocyclic compounds - pyrrole - synthesis of pyrrole - characteristic rea...
Quinoline and isoquinoline- heterocyclic chemistry- pharmacy

Quinoline and isoquinoline- heterocyclic chemistry- pharmacy
Ähnlich wie Polynuclear hydrocarbons module9
Ähnlich wie Polynuclear hydrocarbons module9 (20)
Sandrogreco Stereoselective Mannich Reaction Of Camphor Titanium Enolate

Sandrogreco Stereoselective Mannich Reaction Of Camphor Titanium Enolate
naphthalene for organic chemistry to understand industrial chemistry

naphthalene for organic chemistry to understand industrial chemistry
PEGylated magnetic nanoparticles (PEG@Fe3O4) as cost effectivealternative for...

PEGylated magnetic nanoparticles (PEG@Fe3O4) as cost effectivealternative for...
PEGylated magnetic nanoparticles (PEG@Fe3O4) as cost effectivealternative for...

PEGylated magnetic nanoparticles (PEG@Fe3O4) as cost effectivealternative for...
vdocuments.net_heterocyclic-chemistry-58bb82e5b406c.ppt

vdocuments.net_heterocyclic-chemistry-58bb82e5b406c.ppt
Polynuclear Hydrocarbons Preparations and Reactions

Polynuclear Hydrocarbons Preparations and Reactions
Mehr von syed mohamed
Mehr von syed mohamed (15)
Kürzlich hochgeladen
+971581248768 Mtp-Kit (500MG) Prices » Dubai [(+971581248768**)] Abortion Pills For Sale In Dubai, UAE, Mifepristone and Misoprostol Tablets Available In Dubai, UAE CONTACT DR.Maya Whatsapp +971581248768 We Have Abortion Pills / Cytotec Tablets /Mifegest Kit Available in Dubai, Sharjah, Abudhabi, Ajman, Alain, Fujairah, Ras Al Khaimah, Umm Al Quwain, UAE, Buy cytotec in Dubai +971581248768''''Abortion Pills near me DUBAI | ABU DHABI|UAE. Price of Misoprostol, Cytotec” +971581248768' Dr.DEEM ''BUY ABORTION PILLS MIFEGEST KIT, MISOPROTONE, CYTOTEC PILLS IN DUBAI, ABU DHABI,UAE'' Contact me now via What's App…… abortion Pills Cytotec also available Oman Qatar Doha Saudi Arabia Bahrain Above all, Cytotec Abortion Pills are Available In Dubai / UAE, you will be very happy to do abortion in Dubai we are providing cytotec 200mg abortion pill in Dubai, UAE. Medication abortion offers an alternative to Surgical Abortion for women in the early weeks of pregnancy. We only offer abortion pills from 1 week-6 Months. We then advise you to use surgery if its beyond 6 months. Our Abu Dhabi, Ajman, Al Ain, Dubai, Fujairah, Ras Al Khaimah (RAK), Sharjah, Umm Al Quwain (UAQ) United Arab Emirates Abortion Clinic provides the safest and most advanced techniques for providing non-surgical, medical and surgical abortion methods for early through late second trimester, including the Abortion By Pill Procedure (RU 486, Mifeprex, Mifepristone, early options French Abortion Pill), Tamoxifen, Methotrexate and Cytotec (Misoprostol). The Abu Dhabi, United Arab Emirates Abortion Clinic performs Same Day Abortion Procedure using medications that are taken on the first day of the office visit and will cause the abortion to occur generally within 4 to 6 hours (as early as 30 minutes) for patients who are 3 to 12 weeks pregnant. When Mifepristone and Misoprostol are used, 50% of patients complete in 4 to 6 hours; 75% to 80% in 12 hours; and 90% in 24 hours. We use a regimen that allows for completion without the need for surgery 99% of the time. All advanced second trimester and late term pregnancies at our Tampa clinic (17 to 24 weeks or greater) can be completed within 24 hours or less 99% of the time without the need surgery. The procedure is completed with minimal to no complications. Our Women's Health Center located in Abu Dhabi, United Arab Emirates, uses the latest medications for medical abortions (RU-486, Mifeprex, Mifegyne, Mifepristone, early options French abortion pill), Methotrexate and Cytotec (Misoprostol). The safety standards of our Abu Dhabi, United Arab Emirates Abortion Doctors remain unparalleled. They consistently maintain the lowest complication rates throughout the nation. Our Physicians and staff are always available to answer questions and care for women in one of the most difficult times in their lives. The decision to have an abortion at the Abortion Clinic in Abu Dhabi, (United Arab Emirates)+971581248768+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...

+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...?#DUbAI#??##{{(☎️+971_581248768%)**%*]'#abortion pills for sale in dubai@
Kürzlich hochgeladen (20)
Vip profile Call Girls In Lonavala 9748763073 For Genuine Sex Service At Just...

Vip profile Call Girls In Lonavala 9748763073 For Genuine Sex Service At Just...
+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...

+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...
Thyroid Physiology_Dr.E. Muralinath_ Associate Professor

Thyroid Physiology_Dr.E. Muralinath_ Associate Professor
9999266834 Call Girls In Noida Sector 22 (Delhi) Call Girl Service

9999266834 Call Girls In Noida Sector 22 (Delhi) Call Girl Service
Human & Veterinary Respiratory Physilogy_DR.E.Muralinath_Associate Professor....

Human & Veterinary Respiratory Physilogy_DR.E.Muralinath_Associate Professor....
pumpkin fruit fly, water melon fruit fly, cucumber fruit fly

pumpkin fruit fly, water melon fruit fly, cucumber fruit fly
FAIRSpectra - Enabling the FAIRification of Spectroscopy and Spectrometry

FAIRSpectra - Enabling the FAIRification of Spectroscopy and Spectrometry
FAIRSpectra - Enabling the FAIRification of Analytical Science

FAIRSpectra - Enabling the FAIRification of Analytical Science
300003-World Science Day For Peace And Development.pptx

300003-World Science Day For Peace And Development.pptx
Module for Grade 9 for Asynchronous/Distance learning

Module for Grade 9 for Asynchronous/Distance learning
development of diagnostic enzyme assay to detect leuser virus

development of diagnostic enzyme assay to detect leuser virus
High Profile 🔝 8250077686 📞 Call Girls Service in GTB Nagar🍑

High Profile 🔝 8250077686 📞 Call Girls Service in GTB Nagar🍑
Connaught Place, Delhi Call girls :8448380779 Model Escorts | 100% verified

Connaught Place, Delhi Call girls :8448380779 Model Escorts | 100% verified
Polynuclear hydrocarbons module9
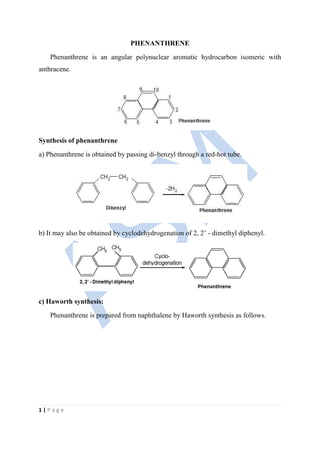
- 1. 1 | P a g e PHENANTHRENE Phenanthrene is an angular polynuclear aromatic hydrocarbon isomeric with anthracene. Synthesis of phenanthrene a) Phenanthrene is obtained by passing di-benzyl through a red-hot tube. b) It may also be obtained by cyclodehydrogenation of 2, 2’ - dimethyl diphenyl. c) Haworth synthesis: Phenanthrene is prepared from naphthalene by Haworth synthesis as follows.
- 2. 2 | P a g e Reactions of phenanthrene i) Pheanthrene can be reduced by sodium and isoamyl alcohol or copperoxide and chromic acid to 9, 10-dihydrophenanthrene. Its oxidation with sodium dichromate and sulphuric acid yield 9,10-phenanthraquinone. ii) It adds on bromine to give 9, 10-dibromophenanthrene and its ozonolysis yields diphenic acid.
- 3. 3 | P a g e iii) It also undergoes electrophilic substitution. It undergoes Friedel-Crafts reaction mainly at position 3. On nitration it gives 3-nitrophenanthrene and on bromination it gives 9-bromophenanthrene. Structure of phenanthrene 1) The molecular formula of phenanthrene is C14H10 . 2) It is isomeric with anthracene. 3) On oxidation it gives phenanthraquinone which on further oxidation gives diphenic acid. 4) Diphenic acid on decarboxylation gives diphenyl. 5) Hence phenanthrene must contain the following skeleton.
- 4. 4 | P a g e 6) On closing the middle ring and filling the hydrogen to satisfy the valencies of various carbon atoms phenanthrene gets the following structure. 7) Evidence in support of the above structure of phenanthrene: It gives five monosubstitution and twenty five disubstitution derivatives. 8) Resonance concept: Phenanthrene is a resonance hybride of the following five contributing structures. The existence of resonance in phenanthrene is proved by the following facts. a) Resonance energy of phenanthrene is 387 kJ/mole. b) Phenanthrene is a flat molecule.
- 5. 5 | P a g e c) X-ray diffraction studies show that all the (C-C) bonds in phenanthrene are not equivalent. C1-C2 has 3/5 double bond character. C2 - C3 has 2/5 double bond character and C9 - C10 has 4/5 double bond character. 9) Finally the sturcutre of phenanthrene is proved by Haworth synthesis: Prepared by Dr. A. Syed Mohamed HoD and Senior Assistant Professor, Research Dept. of Chemistry Sadakathullah Appa College (Autonomous) Tirunelveli, Tamilnadu, India. asm2032@gmail.com The author ackowledges Prof. H. Kassali Rahmathullah, Dr. T. Syed Ismail, Dr. M. Kamalutheen, Former Professor, Dept. of Chemistry, Sadakathullah Appa College for their valuable contribution.
