Melden
Teilen
Empfohlen
Rayegan thermo i-cengel-chapter 5-2

This document discusses mass and energy analysis of control volumes for steady-flow engineering devices. It covers topics like nozzles, diffusers, turbines, compressors, throttling valves, mixing chambers, heat exchangers, and pipe/duct flow. Examples are provided for analyzing the energy and mass balances of these systems using the first law of thermodynamics. The document concludes with assigning homework problems involving these thermodynamic concepts.
Rayegan thermo i-cengel-chapter 3-p1

This document discusses thermodynamic properties of pure substances. It introduces pure substances and their phases of solid, liquid, and gas. Phase change processes are described, including saturated liquid, vapor, and superheated regions. T-V, P-V, and P-T diagrams are presented to illustrate the relationships between these properties. Key concepts covered include saturation temperature/pressure, latent heats of fusion and vaporization, the p-v-T surface, and how substances' behaviors differ when expanding or contracting during phase changes. Objectives are listed for understanding pure substance thermodynamics and properties.
Rayegan thermo i-cengel-chapter 5-1

This document provides an overview of chapter 5 of a thermodynamics textbook, which covers mass and energy analysis of control volumes. The key objectives are to develop the conservation of mass and first law of thermodynamics as applied to control volumes. It defines concepts like mass flow rate, volume flow rate, enthalpy, and flow work. Examples are provided to demonstrate applying conservation of mass to problems involving steady and unsteady flow systems as well as compressible and incompressible fluids. The chapter also discusses applying the energy balance to steady flow systems like nozzles, compressors, and heat exchangers.
Rayegan thermo i-cengel-chapter 4-p2

This document discusses energy analysis of closed systems, including specific heats at constant volume and pressure, internal energy, enthalpy, and specific heats of ideal gases and incompressible substances like solids and liquids. It provides equations relating these concepts and explains how to calculate changes in internal energy and enthalpy using data tables, functional forms of specific heats, or average specific heat values. Examples are also included.
Thermo chapter 1

- Thermodynamics deals with energy and its transformations. Key concepts introduced include systems, properties, state, equilibrium, processes and cycles.
- Important units and dimensions in the metric (SI) and English systems are discussed. The concept of dimensional homogeneity is emphasized.
- Temperature scales, pressure, atmospheric pressure, and their measurement using instruments such as thermometers, manometers and barometers are described.
- Variations in pressure with depth and its transmission in fluids, as governed by Pascal's law, are explained.
Thermo chapter 2-p2

The document discusses key concepts of the first law of thermodynamics including:
- Energy can change forms but is conserved, neither created nor destroyed.
- The energy balance of a system equals energy entering minus exiting by heat, work, and mass flow.
- Energy transfer occurs through heat, work, and for open systems, mass flow.
- Energy efficiency compares useful energy transferred to total energy supplied.
It provides examples of efficiency calculations for devices like heaters, generators, and pumps. The homework problems reinforce applying the first law to analyze energy transfers and changes within thermodynamic systems.
SSL7 Energy Analysis of Closed Systems

Moving boundary work
Boundary work for an isothermal process
Boundary work for a constant-pressure process
Boundary work for a polytropic process
Energy balance for closed systems
Energy balance for a constant-pressure expansion or compression process
Specific heats
Constant-pressure specific heat, cp
Constant-volume specific heat, cv
Internal energy, enthalpy and specific heats of ideal gases
Energy balance for a constant-pressure expansion or compression process
Internal energy, enthalpy and specific heats of incompressible substances (Solids and liquids)
Bab 1 Thermodynamic of Engineering Approach

This document provides an introduction to basic thermodynamics concepts. It defines thermodynamics as the science of energy and discusses the first and second laws of thermodynamics. The first law states that energy is conserved and can change forms, while the second law says that the quality of energy decreases in actual processes. The document introduces systems, properties, processes, cycles and other foundational topics, providing objectives and definitions for understanding thermodynamics.
Empfohlen
Rayegan thermo i-cengel-chapter 5-2

This document discusses mass and energy analysis of control volumes for steady-flow engineering devices. It covers topics like nozzles, diffusers, turbines, compressors, throttling valves, mixing chambers, heat exchangers, and pipe/duct flow. Examples are provided for analyzing the energy and mass balances of these systems using the first law of thermodynamics. The document concludes with assigning homework problems involving these thermodynamic concepts.
Rayegan thermo i-cengel-chapter 3-p1

This document discusses thermodynamic properties of pure substances. It introduces pure substances and their phases of solid, liquid, and gas. Phase change processes are described, including saturated liquid, vapor, and superheated regions. T-V, P-V, and P-T diagrams are presented to illustrate the relationships between these properties. Key concepts covered include saturation temperature/pressure, latent heats of fusion and vaporization, the p-v-T surface, and how substances' behaviors differ when expanding or contracting during phase changes. Objectives are listed for understanding pure substance thermodynamics and properties.
Rayegan thermo i-cengel-chapter 5-1

This document provides an overview of chapter 5 of a thermodynamics textbook, which covers mass and energy analysis of control volumes. The key objectives are to develop the conservation of mass and first law of thermodynamics as applied to control volumes. It defines concepts like mass flow rate, volume flow rate, enthalpy, and flow work. Examples are provided to demonstrate applying conservation of mass to problems involving steady and unsteady flow systems as well as compressible and incompressible fluids. The chapter also discusses applying the energy balance to steady flow systems like nozzles, compressors, and heat exchangers.
Rayegan thermo i-cengel-chapter 4-p2

This document discusses energy analysis of closed systems, including specific heats at constant volume and pressure, internal energy, enthalpy, and specific heats of ideal gases and incompressible substances like solids and liquids. It provides equations relating these concepts and explains how to calculate changes in internal energy and enthalpy using data tables, functional forms of specific heats, or average specific heat values. Examples are also included.
Thermo chapter 1

- Thermodynamics deals with energy and its transformations. Key concepts introduced include systems, properties, state, equilibrium, processes and cycles.
- Important units and dimensions in the metric (SI) and English systems are discussed. The concept of dimensional homogeneity is emphasized.
- Temperature scales, pressure, atmospheric pressure, and their measurement using instruments such as thermometers, manometers and barometers are described.
- Variations in pressure with depth and its transmission in fluids, as governed by Pascal's law, are explained.
Thermo chapter 2-p2

The document discusses key concepts of the first law of thermodynamics including:
- Energy can change forms but is conserved, neither created nor destroyed.
- The energy balance of a system equals energy entering minus exiting by heat, work, and mass flow.
- Energy transfer occurs through heat, work, and for open systems, mass flow.
- Energy efficiency compares useful energy transferred to total energy supplied.
It provides examples of efficiency calculations for devices like heaters, generators, and pumps. The homework problems reinforce applying the first law to analyze energy transfers and changes within thermodynamic systems.
SSL7 Energy Analysis of Closed Systems

Moving boundary work
Boundary work for an isothermal process
Boundary work for a constant-pressure process
Boundary work for a polytropic process
Energy balance for closed systems
Energy balance for a constant-pressure expansion or compression process
Specific heats
Constant-pressure specific heat, cp
Constant-volume specific heat, cv
Internal energy, enthalpy and specific heats of ideal gases
Energy balance for a constant-pressure expansion or compression process
Internal energy, enthalpy and specific heats of incompressible substances (Solids and liquids)
Bab 1 Thermodynamic of Engineering Approach

This document provides an introduction to basic thermodynamics concepts. It defines thermodynamics as the science of energy and discusses the first and second laws of thermodynamics. The first law states that energy is conserved and can change forms, while the second law says that the quality of energy decreases in actual processes. The document introduces systems, properties, processes, cycles and other foundational topics, providing objectives and definitions for understanding thermodynamics.
Hw#3 (2)

Homework assignment #3 in fluid mechanics is due November 24th and contains 12 problems from chapter 8 in the textbook ranging in topics from laminar and turbulent fluid flows to fluid properties and forces. The assignment spans two slides and students are instructed to see the next slide for additional details related to the homework.
Hw#6

This homework assignment contains 18 problems from chapter 6 that are due on April 17. The problems include 6.4C, 6.10C, 6.13C, 6.21, 6.23, 6.32C, 6.40, 6.50, 6.57, 6.80E, 6.81E, 6.90C, 6.96, 6.97, 6.99, 6.109, and 6.138.
Hw#5 (1)

Homework #5 from an unknown course is due on Tuesday, April 8. The homework consists of solving 13 problems from the textbook, including problems 5.8, 5.17, 5.24, 5.30, 5.42, 5.49, 5.56, 5.71E, 5.75, 5.90, 5.114, and 5.155.
Rayegan thermo i-cengel-chapter 3-p2

1) The document discusses properties of pure substances and how they are presented in tables. It focuses on water properties and steam tables.
2) It explains different phases like saturated liquid, saturated vapor, superheated vapor, and compressed liquid. It also discusses quality and using tables to find properties through interpolation.
3) The ideal gas equation of state is presented along with when it can be applied to water vapor. The compressibility factor is introduced as a measure of how gases deviate from ideal behavior.
Rayegan thermo i-cengel-chapter 4-p1

Thermodynamics I discusses energy analysis of closed systems. It examines moving boundary work from processes like in engines and compressors. The first law of thermodynamics states the principle of conservation of energy for closed systems. The general energy balance applied to closed systems relates the change in internal energy to heat and work. Specific heats at constant volume and pressure are defined and used to calculate changes in internal energy and enthalpy for ideal gases and incompressible substances.
Hw#4

This homework assignment contains 14 math problems from chapter 4 that are due on Thursday, March 27. The problems include 4.5E, 4.8, 4.24, 4.25, 4.34, 4.42, 4.46, 4.55, 4.59E, 4.70E, 4.74, 4.81, and 4.89 from the textbook.
Hw#3 

Homework #3 from an unknown course is due on Thursday, March 6. It consists of solving 13 problems from Chapter 3: problems 3.13C, 3.15C, 3.20C, 3.28, 3.37E, 3.42, 3.44, 3.57, 3.61, 3.78, 3.81, 3.89, and 3.93.
Thermo chapter 2-p1

The document discusses various topics related to energy and energy transfer in thermodynamics:
1. It defines different forms of energy including internal, kinetic, potential, electrical, chemical, and nuclear energy. Internal energy is the sum of microscopic energies of a system.
2. It discusses heat and work as two mechanisms of energy transfer across boundaries of a system. Heat transfer is driven by temperature differences while work requires a force and displacement.
3. It describes different modes of heat transfer as conduction, convection, and radiation. Mechanical forms of work include shaft work, spring work, and electrical work.
4. The first law of thermodynamics and concept of energy balance within a system and
Hw#3

Homework #3 from an unknown course is due on Thursday, March 6. It consists of solving 13 problems from Chapter 3 of the textbook, ranging from problems 3.13C through 3.93. Students are to complete problems 3.13C, 3.15C, 3.20C, 3.28, 3.37E, 3.42, 3.44, 3.57, 3.61, 3.78, 3.81, 3.89, and 3.93.
Weitere ähnliche Inhalte
Mehr von Larry Howard
Hw#3 (2)

Homework assignment #3 in fluid mechanics is due November 24th and contains 12 problems from chapter 8 in the textbook ranging in topics from laminar and turbulent fluid flows to fluid properties and forces. The assignment spans two slides and students are instructed to see the next slide for additional details related to the homework.
Hw#6

This homework assignment contains 18 problems from chapter 6 that are due on April 17. The problems include 6.4C, 6.10C, 6.13C, 6.21, 6.23, 6.32C, 6.40, 6.50, 6.57, 6.80E, 6.81E, 6.90C, 6.96, 6.97, 6.99, 6.109, and 6.138.
Hw#5 (1)

Homework #5 from an unknown course is due on Tuesday, April 8. The homework consists of solving 13 problems from the textbook, including problems 5.8, 5.17, 5.24, 5.30, 5.42, 5.49, 5.56, 5.71E, 5.75, 5.90, 5.114, and 5.155.
Rayegan thermo i-cengel-chapter 3-p2

1) The document discusses properties of pure substances and how they are presented in tables. It focuses on water properties and steam tables.
2) It explains different phases like saturated liquid, saturated vapor, superheated vapor, and compressed liquid. It also discusses quality and using tables to find properties through interpolation.
3) The ideal gas equation of state is presented along with when it can be applied to water vapor. The compressibility factor is introduced as a measure of how gases deviate from ideal behavior.
Rayegan thermo i-cengel-chapter 4-p1

Thermodynamics I discusses energy analysis of closed systems. It examines moving boundary work from processes like in engines and compressors. The first law of thermodynamics states the principle of conservation of energy for closed systems. The general energy balance applied to closed systems relates the change in internal energy to heat and work. Specific heats at constant volume and pressure are defined and used to calculate changes in internal energy and enthalpy for ideal gases and incompressible substances.
Hw#4

This homework assignment contains 14 math problems from chapter 4 that are due on Thursday, March 27. The problems include 4.5E, 4.8, 4.24, 4.25, 4.34, 4.42, 4.46, 4.55, 4.59E, 4.70E, 4.74, 4.81, and 4.89 from the textbook.
Hw#3 

Homework #3 from an unknown course is due on Thursday, March 6. It consists of solving 13 problems from Chapter 3: problems 3.13C, 3.15C, 3.20C, 3.28, 3.37E, 3.42, 3.44, 3.57, 3.61, 3.78, 3.81, 3.89, and 3.93.
Thermo chapter 2-p1

The document discusses various topics related to energy and energy transfer in thermodynamics:
1. It defines different forms of energy including internal, kinetic, potential, electrical, chemical, and nuclear energy. Internal energy is the sum of microscopic energies of a system.
2. It discusses heat and work as two mechanisms of energy transfer across boundaries of a system. Heat transfer is driven by temperature differences while work requires a force and displacement.
3. It describes different modes of heat transfer as conduction, convection, and radiation. Mechanical forms of work include shaft work, spring work, and electrical work.
4. The first law of thermodynamics and concept of energy balance within a system and
Hw#3

Homework #3 from an unknown course is due on Thursday, March 6. It consists of solving 13 problems from Chapter 3 of the textbook, ranging from problems 3.13C through 3.93. Students are to complete problems 3.13C, 3.15C, 3.20C, 3.28, 3.37E, 3.42, 3.44, 3.57, 3.61, 3.78, 3.81, 3.89, and 3.93.
Mehr von Larry Howard (9)
Hw#2
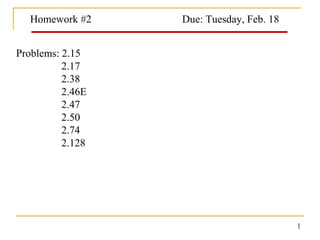
- 1. Homework #2 Due: Tuesday, Feb. 18 Problems: 2.15 2.17 2.38 2.46E 2.47 2.50 2.74 2.128 1