revision on chapter periodic table, chemical bonding and electrolysis with answers
•Als PPT, PDF herunterladen•
2 gefällt mir•1,670 views
The document contains information about the periodic table including: 1) It lists the elements hydrogen through argon and their symbols in the periodic table. 2) It explains that noble gases are chemically non-reactive because they have a stable electron configuration with a full outer shell. 3) Sodium undergoes reactions with oxygen, water, and chlorine that produce sodium oxide, sodium hydroxide, and sodium chloride, respectively.
Melden
Teilen
Melden
Teilen
Empfohlen
Empfohlen
Weitere ähnliche Inhalte
Was ist angesagt?
Was ist angesagt? (20)
F.Sc.2.Chemistry.Ch.2.Test Solved-Malik Xufyan ...

F.Sc.2.Chemistry.Ch.2.Test Solved-Malik Xufyan ...
Mcqs in inorganic and physical chemistry By Malik Xufyan

Mcqs in inorganic and physical chemistry By Malik Xufyan
F.sc. Part 1 Chemistry Chapterwise Test Solved by Malik Xufyan

F.sc. Part 1 Chemistry Chapterwise Test Solved by Malik Xufyan
Andere mochten auch
Andere mochten auch (20)
Fluoride as pollutant in air, water and its impacts on plants

Fluoride as pollutant in air, water and its impacts on plants
Chapter 9: Contact Process, Haber Process and Alloy

Chapter 9: Contact Process, Haber Process and Alloy
Ähnlich wie revision on chapter periodic table, chemical bonding and electrolysis with answers
Ähnlich wie revision on chapter periodic table, chemical bonding and electrolysis with answers (20)
Seminar revision on chapter electrchemistry, carbon compound and thermo che...

Seminar revision on chapter electrchemistry, carbon compound and thermo che...
Seminar revision on chapter electrchemistry, carbon compound and thermo che...

Seminar revision on chapter electrchemistry, carbon compound and thermo che...
Seminar april revision form 5== chapter electrchemistry, carbon compound and ...

Seminar april revision form 5== chapter electrchemistry, carbon compound and ...
Mehr von MRSMPC
Mehr von MRSMPC (20)
Redox part 3= rusting - reactivity series and diff between electrolytic cell...

Redox part 3= rusting - reactivity series and diff between electrolytic cell...
Revision on consumer, r te, thermo and carbon compound

Revision on consumer, r te, thermo and carbon compound
Revision on consumer, r te, thermo and carbon compound

Revision on consumer, r te, thermo and carbon compound
Revision for salt, electrochemistry, carbon compounds , thermochemistry

Revision for salt, electrochemistry, carbon compounds , thermochemistry
Soap and detergent, medicine , food additives consumer 2011-edited-2

Soap and detergent, medicine , food additives consumer 2011-edited-2
fats and natural rubber module 3 carbon compounds with answers

fats and natural rubber module 3 carbon compounds with answers
Quiz 2( chemical bond, electrchemistry, acid base)

Quiz 2( chemical bond, electrchemistry, acid base)
Quiz3with answers latest 1(salt, industry, rate of rex)

Quiz3with answers latest 1(salt, industry, rate of rex)
Quiz 2 (chemical bond, electrochemistry, acid base)

Quiz 2 (chemical bond, electrochemistry, acid base)
Kürzlich hochgeladen
Mehran University Newsletter is a Quarterly Publication from Public Relations OfficeMehran University Newsletter Vol-X, Issue-I, 2024

Mehran University Newsletter Vol-X, Issue-I, 2024Mehran University of Engineering & Technology, Jamshoro
Kürzlich hochgeladen (20)
This PowerPoint helps students to consider the concept of infinity.

This PowerPoint helps students to consider the concept of infinity.
Micro-Scholarship, What it is, How can it help me.pdf

Micro-Scholarship, What it is, How can it help me.pdf
General Principles of Intellectual Property: Concepts of Intellectual Proper...

General Principles of Intellectual Property: Concepts of Intellectual Proper...
Interdisciplinary_Insights_Data_Collection_Methods.pptx

Interdisciplinary_Insights_Data_Collection_Methods.pptx
Python Notes for mca i year students osmania university.docx

Python Notes for mca i year students osmania university.docx
ICT Role in 21st Century Education & its Challenges.pptx

ICT Role in 21st Century Education & its Challenges.pptx
Unit 3 Emotional Intelligence and Spiritual Intelligence.pdf

Unit 3 Emotional Intelligence and Spiritual Intelligence.pdf
Kodo Millet PPT made by Ghanshyam bairwa college of Agriculture kumher bhara...

Kodo Millet PPT made by Ghanshyam bairwa college of Agriculture kumher bhara...
Fostering Friendships - Enhancing Social Bonds in the Classroom

Fostering Friendships - Enhancing Social Bonds in the Classroom
revision on chapter periodic table, chemical bonding and electrolysis with answers
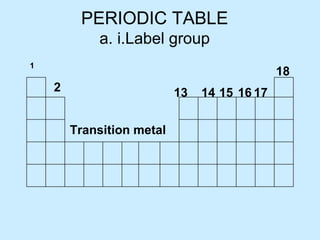
- 1. PERIODIC TABLE a. i.Label group 1 17 Transition metal 13 14 15 16 18 2
- 2. ii.Label period 1 to 5 Period 1 Period 2 Period 3 Period 4 Period 5 1 2 Transition metal 13 14 15 16 17 18
- 3. iii.Place proton number of elements 1 to 20 in the periodic table above 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20
- 4. iv.Place all the elements given in the periodic table above with their symbols Transition metal H He F Ne Na Cu Cl Br Ar K Mn Li
- 7. Sodium undergoes the following reactions. Write chemical equation: i) With oxygen: ii) With water: iii) With chlorine: 4Na + O 2 2Na 2 O 2Na + 2H 2 O 2NaOH + H 2 2Na + Cl 2 2NaCl
- 18. Draw electron arrangement for compounds formed between a) Sodium and oxygen ( given proton no. Na, 11: O, 8)
- 20. b) For water molecule
- 22. c) Carbon and chlorine
- 26. 1. MOLTEN LEAD(II)IODIDE WITH CARBON ELECTRODE
- 29. ANODE CATHODE OBSER A PURPLE GAS EVOLVED GREY AND SHINY SOLID DEPOSITED
- 30. 2. HYDROCHLORIC ACID 0.0001 moldm -3 WITH CARBON ELECTRODE ALL IONS PRESENT H + , Cl - & OH -
- 33. ANODE CATHODE OBS COLOURLESS GAS THAT REKINDLES GLOWING WOODEN SPLINTER COLOURLESS GAS THAT GIVES ‘POP’ SOUND -TESTED USING BURNING WOODEN SPLINTER
