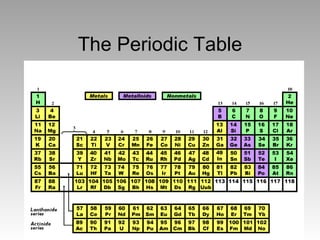
The Periodic Table
•Als PPT, PDF herunterladen•
224 gefällt mir•125,846 views
organizing the elements, periodic table, Mendeleev, periodic law, metals, nonmetals, metalloinds, periodic trends
Melden
Teilen
Melden
Teilen
Weitere ähnliche Inhalte
Was ist angesagt?
Was ist angesagt? (20)
Atomic number, Mass number, Relative atomic mass and Atomic mass unit

Atomic number, Mass number, Relative atomic mass and Atomic mass unit
Grade 8 Chemistry Structure of Matter : Atoms, Molecules and Ions

Grade 8 Chemistry Structure of Matter : Atoms, Molecules and Ions
Andere mochten auch
Andere mochten auch (6)
Periodic Table of SEO Success Factors & Guide to SEO by SearchEngineLand

Periodic Table of SEO Success Factors & Guide to SEO by SearchEngineLand
Ähnlich wie The Periodic Table
Ähnlich wie The Periodic Table (20)
Periodic Classification of Elements and Periodicity

Periodic Classification of Elements and Periodicity
Mehr von Currituck County High School
Mehr von Currituck County High School (20)
Kürzlich hochgeladen
Making communications land - Are they received and understood as intended? we...

Making communications land - Are they received and understood as intended? we...Association for Project Management
Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...

Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...christianmathematics
Kürzlich hochgeladen (20)
UGC NET Paper 1 Mathematical Reasoning & Aptitude.pdf

UGC NET Paper 1 Mathematical Reasoning & Aptitude.pdf
Making communications land - Are they received and understood as intended? we...

Making communications land - Are they received and understood as intended? we...
Fostering Friendships - Enhancing Social Bonds in the Classroom

Fostering Friendships - Enhancing Social Bonds in the Classroom
Mixin Classes in Odoo 17 How to Extend Models Using Mixin Classes

Mixin Classes in Odoo 17 How to Extend Models Using Mixin Classes
On National Teacher Day, meet the 2024-25 Kenan Fellows

On National Teacher Day, meet the 2024-25 Kenan Fellows
Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...

Explore beautiful and ugly buildings. Mathematics helps us create beautiful d...
ICT role in 21st century education and it's challenges.

ICT role in 21st century education and it's challenges.
Unit-IV; Professional Sales Representative (PSR).pptx

Unit-IV; Professional Sales Representative (PSR).pptx
Kodo Millet PPT made by Ghanshyam bairwa college of Agriculture kumher bhara...

Kodo Millet PPT made by Ghanshyam bairwa college of Agriculture kumher bhara...
This PowerPoint helps students to consider the concept of infinity.

This PowerPoint helps students to consider the concept of infinity.
Python Notes for mca i year students osmania university.docx

Python Notes for mca i year students osmania university.docx
The Periodic Table
- 5. Henry Moseley 1887 - 1915 In 1913, through his work with X-rays, he determined the actual nuclear charge (atomic number) of the elements*. He rearranged the elements in order of increasing atomic number. *“There is in the atom a fundamental quantity which increases by regular steps as we pass from each element to the next. This quantity can only be the charge on the central positive nucleus.”
- 22. Trends in the periodic table: Ionization Energy Atomic Radius Electron Affinity Electronegativity
- 25. Atomic Radius
