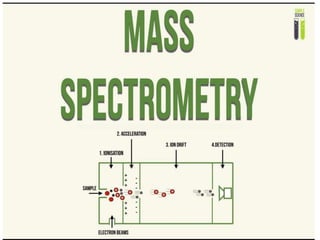
Mass spectrometry
- 2. Weight The most common definition of weight found in introductory physics textbooks defines weight as the force exerted on a body by gravity. An object's weight depends on its mass and the strength of the gravitational pull. The force with which an object near the Earth or another celestial body is attracted toward the center of the body by gravity.
- 3. Mass • Mass is both a property of a physical body and a measure of its resistance to acceleration (a change in its state of motion) when a net force is applied. An object's mass also determines the strength of its gravitational attraction to other bodies. The basic SI unit of mass is the kilogram (kg).
- 8. • Mass spectrometry is a microanalytical technique that can be used selectively to detect and determine the amount of a given analyte. • Mass spectrometry is also used to determine the elemental composition and some aspects of the molecular structure of an analyte. • The tools of mass spectrometry are mass spectrometers, and the data are mass spectra.
- 9. • The experimental measurement of the mass of gas-phase ions produced from molecules of an analyte. • Mass spectrometry concerns itself with the mass of the isotopes of the elements, not the atomic mass of the elements.
- 10. Atomic number
- 11. Atomic number The atomic number or proton number (symbol Z) of a chemical element is the number of protons found in the nucleus of every atom of that element. The atomic number uniquely identifies a chemical element. It is identical to the charge number of the nucleus. In an uncharged atom, the atomic number is also equal to the number of electrons.
- 14. Atomic mass • The atomic mass of an element is the weighted average of the naturally occurring stable isotopes that comprise the element.
- 15. Ionization • Ionization or ionisation, is the process by which an atom or a molecule acquires a negative or positive charge by gaining or losing electrons, often in conjunction with other chemical changes.
- 19. • Mass spectrometry does not directly determine mass; it determines the mass-to- charge ratio (m/z) of ions. • It is a fundamental requirement of mass spectrometry that the ions be in the gas phase before they can be separated according to their individual m/z values and detected.
- 20. Before 1970- ionization techniques • Analytes having significant vapor pressure were amenable to mass spectrometry because gas-phase ions could only be produced from gas-phase molecules by the techniques of electron ionization (EI) or chemical ionization (CI). • Nonvolatile and thermally labile molecules were not amenable.
- 21. After 1970- Desorption techniques • After 1970, the capabilities of mass spectrometry were expanded by the development of desorption/ionization (D/I)techniques, the generic process of generating gas-phase ions directly from a sample in the condensed phase. • The first viable and widely accepted technique2 for D/I was fast atom bombardment (FAB), which required nanomoles of analyte to produce an interpretable mass spectrum.
- 22. In 1980 • During the 1980s, electrospray ionization (ESI) and matrix-assisted laser desorption/ionization (MALDI) eclipsed FAB, in part because they required only picomoles of analyte for analysis. • ESI and MALDI are suitable for analysis of femtomole quantities of thermally labile and nonvolatile analytes
- 23. The Concept of Mass Spectrometry • Ions are charged particles and, as such, their position in space can be manipulated with the use of electric and magnetic fields. • When only individual ions are present, they can be grouped according to their unique properties (mass and the number of charges) and moved from one point to another. • In order to have individual ions free from any other forms of matter, it is necessary to analyze them in a vacuum.
- 24. • Mass spectrometry takes advantage of ions in the gas phase at low pressures to separate and detect them according to their • mass-to-charge ratio (m/z) – the mass of the ion on the atomic scale divided by the number of charges that the ion possesses.
- 25. • Only ions are detected in mass spectrometry. • Any particles that are not ionic (molecules or radicals ) are removed from the mass spectrometer by the continuous pumping that maintains the vacuum. • Both molecules and radicals are particles that have no charge. • Molecules are characterized by an even number of electrons . • Radicals by an odd number of electrons.
- 26. • The mass component that makes up the dimensionless m/z unit is based on an atomic scale rather than the physical scale. • Mass physical scale is defined as one kilogram being the mass of one liter of water at a specific temperature and pressure. • The atomic mass scale is defined based on a fraction of a specific isotope of carbon; • i.e., 1 mass unit on an atomic scale is equal to 1/12 the mass of the most abundant naturally occurring stable isotope of carbon, 12C. This definition of mass. • Mass is represented by the symbol u, which is synonymous with dalton(Da).
- 27. • In the study of mass spectrometry, it is important to always keep in mind that the entity measured in the mass spectrometer is the mass-to-charge ratio of an ion, not the mass of the ion. • It is inappropriate to use a unit of mass when describing the mass-to-charge ratio of an ion. Ions have both mass and an m/z value.
- 28. M+ ions • The most common ionization process for gas- phase analysis, • EI, transfers energy to the neutral molecule (a species characterized as having an even number of electrons) in the vapor state, giving it sufficient energy to eject one of its own electrons, thereby leaving a residual positive charge on the now ionic species. This process produces a molecular ion with a positive charge and odd number of electrons, as represented by the M+
- 29. • This M + may have considerable excess energy that can be dissipated through fragmentation of certain chemical bonds. • Cleavage of various chemical bonds leads to the production of positive-charge fragment ions whose mass is equal to the sum of the atomic masses of the constituent atoms. • Not all of the molecular ions necessarily decompose into fragment ions. For compounds producing a relatively stable M +, such as those stabilized through resonance, like aromatic compounds, an intense molecular-ion peak will be recorded because the M+ tends to survive or resist fragmentation. • For compounds that do not produce stable molecular ions, like aliphatic alcohols, nearly all of them decompose into fragment ions. In these cases, the mass spectrum contains only a small peak representing the M+ Various combinations of the above-described processes are the basis of the chemical “fingerprint” in the form of a mass spectrum for a given compound.
- 30. Calutron mass spectrometers were used in the Manhattan Project for uranium enrichment.
- 31. Replica of J.J. Thomson's third mass spectrometer
- 32. Surface ionization source at the Argonne National Laboratory linear accelerator
- 34. ISOTOPES
- 35. • An element is specified by the number of protons in its nucleus. This equals the atomic number of the respective element, and thus determines its place within the periodic table of the elements. The atomic number is given as a subscript preceding the elemental symbol, e.g., 6C in case of carbon.
- 36. • Atoms with nuclei of the same atomic number differing in the number of neutrons are termed isotopes. • The mass number of an isotope is given as superscript preceding the elemental symbol, e.g., 12C.
- 37. Monoisotopic elements Elements do exist in only one naturally occurring stable isotope. • Fluorine (19F), • Sodium (23Na), • Phosphorus (31P) • Iodine (127I) • The monoisotopic elements are also referred to as A or X elements
- 38. Di-isotopic Elements • Several elements naturally exist in two isotopes. • The first group has been termed A+1 or X+1 elements, the latter ones have been termed A+2 or X+2 elements.
- 39. X+1 Elements • Hydrogen (1H, 2H = D), carbon (12C, 13C) and nitrogen (14N, 15N). • Deuterium (D) is of low abundance (0.0115 %) • And therefore, hydrogen is usually treated as monoisotopic or X element
- 40. X+2 Elements • Chlorine (35cl, 37cl) • Bromine (79br, 81br) are Commonly Known, • Copper (63cu, 65cu), • Gallium (69ga, 71ga), • Silver (107ag, 109ag), • Indium (113in, 115in) and • Antimony (121Sb, 123Sb)
- 41. X-1 element The elements lithium (6Li, 7Li), boron (10B, 11B) and vanadium (50V, 51V) come together with a lighter isotope of lower abundance than the heavier one and thus, they can be grouped together as X–1 elements.
- 42. Polyisotopic Elements The majority of elements belongs to the polyisotopic elements because they consist of three or more isotopes showing a wide variety of isotopic distributions.
- 43. The isotopic mass • It is the exact mass of an isotope. • It is very close to but not equal to the nominal mass of the isotope . • The only exception is the carbon isotope 12C which has an isotopic mass of 12.000000 u.
- 44. Monoisotopic Mass The exact mass of the most abundant isotope of an element is termed monoisotopic mass.