Membranas celulares: estructura, funciones y transporte
- 1. Membranas
- 4. Concentración de iones libres y potenciales de equilibrio de las células músculo esqueléticas de mamífero. Los potenciales de equilibrio fueron calculados para 37 ºC asumiendo un potencial de -90 mV para el potencial de reposo de la membrana de las células musculares. El radio iónico relativo se indica en la parte superior.
- 5. Lipid composition of the plasma membrane and organelle membranes of a rat hepatocyte. The functional specialization of each membrane type is reflected in its unique lipid composition. Cholesterol is prominent in plasma membranes but barely detectable in mitochondrial membranes. Cardiolipin is a major component of the inner mitochondrial membrane but not of the plasma membrane. Phosphatidylserine, phosphatidylinositol, and phosphatidylglycerol are relatively minor components (yellow) of most membranes but serve critical functions; phosphatidylinositol and its derivatives, for example, are important in signal transductions triggered by hormones. Sphingolipids, phosphatidylcholine, and phosphatidylethanolamine are present in most membranes, but in varying proportions. Glycolipids, which are major components of the chloroplast membranes of plants, are virtually absent from animal cells.
- 7. Distribución asimétrica de lípidos en la membrana.
- 14. Distribución asimétrica de fosfolípidos y glicolípidos
- 24. Phospholipids can be “flipped” across a bilayer membrane by the action of flippase proteins. When, by normal diffusion through the bilayer, the lipid encounters a flippase, it can be moved quickly to the other face of the bilayer.
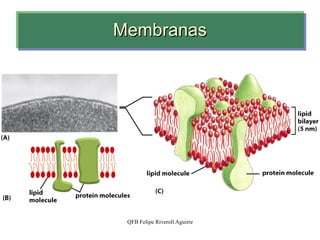
- 26. Modelo de mosaico fluido
- 29. Diagram of how various classes of proteins associate with the lipid bilayer . Integral (transmembrane) proteins span the bilayer. Lipid-anchored proteins are tethered to one leaflet by a long covalently attached hydrocarbon chain. Peripheral proteins associate with the membrane primarily by specific noncovalent interactions with integral proteins or membrane lipids. Farther from the membrane are membraneassociated proteins including the cytoskeleton, extracellular matrix in animal cells, and cell wall in plant and bacterial cells (not depicted). Carbohydrate chains are attached to many extracellular proteins and to the exoplasmic domains of many transmembrane proteins.
- 30. Proteínas de membrana ancladas a lípidos
- 32. Maneras en que se asocian las proteínas de membrana con la bicapa lipídica : (1)Una simple α -hélice, (2) como múltiples α -hélices, o (3) como una lámina- β enrrollada ( β-barril ), (4) algunas están ancladas a la superficie de la cara citosólica por una α -hélice particionada dentro de la monocapa citosólicade la bicapa lipídica a través de la cara hidrofóbica de la hélice, (5) otras están ancladas la bicapa solamente por un enlace covalente a una molécula lipídica de la membrana, (6) o por un enlace a un oligosacárido o fosfatidilinositol en la monocapa nocitosólica. (7 y 8) Finalmente, muchas proteínas están ancladas a la membrana por interacciones no covalentes, con otras proteínas membranales
- 33. Peripheral, integral, and amphitropic proteins. Membrane proteins can be operationally distinguished by the conditions required to release them from the membrane. Most peripheral proteins are released by changes in pH or ionic strength, removal of Ca 2+ by a chelating agent, or addition of urea or carbonate. Integral proteins are extractable with detergents, which disrupt the hydrophobic interactions with the lipid bilayer and form micelle-like clusters around individual protein molecules. Integral proteins covalently attached to a membrane lipid, such as a glycosyl phosphatidylinositol (GPI; see Figure 11-14), can be released by treatment with phospholipase C. Amphitropic proteins are sometimes associated with membranes and sometimes not, depending on some type of regulatory process, such as reversible palmitoylation.
- 38. ...Transporte
- 45. Ley de Van´t Hoff
- 49. Tonicidad
- 55. 1 2 3 4 Passive and active transport protein function. The protein binds its specific substrate and then undergoes a conformational change allowing the molecule or ion to be released on the other side of the membrane. Cotransporters have specific binding sites for each transported species.
- 58. Bomba Na/K
