Renal Pathology Lectures_Ppt Series (4 in 1)
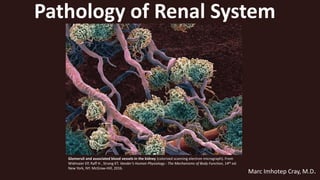
- 1. Glomeruli and associated blood vessels in the kidney (colorized scanning electron micrograph). From Widmaier EP, Raff H , Strang KT. Vander’s Human Physiology : The Mechanisms of Body Function, 14th ed. New York, NY: McGraw-Hill, 2016. Pathology of Renal System Marc Imhotep Cray, M.D.
- 2. Marc Imhotep Cray, MD Overall Goal 2 The overall goal of this lecture series is to provide the learner with a basic scientific understanding of the patterns, causes, mechanisms and effects of the most clinically common renal disorders; including: Acute renal failure (AKI) and chronic renal failure (CKD) Glomerular diseases Urinary Tract Infections Tubulointerstitial diseases Obstructive uropathy, hydronephrosis & urolithiasis Vascular diseases Renal neoplasms Congenital and cystic diseases of kidney Volume regulation & electrolyte disorders Acid-base disorders Covered elsewhere within the sequence.
- 3. Marc Imhotep Cray, MD Learning objectives (abridged*) 3 1.Describe the causes, mechanisms and effects of acute kidney injury. 2.Describe the causes, mechanisms and effects of chronic kidney disease. 3. Classify glomerular diseases based on acute vs chronic, histopathologic features and clinical presentation. 4. Explain the pathogenesis (ie, immune-mediated Injury) and pathophysiology of the most common glomerulonephritidies. 5. Describe the clinicopathologic features of the nephrotic vs nephritic syndrome *See Renal Pathology Learning Objectives, unabridged version.pdf
- 4. Marc Imhotep Cray, MD Learning objectives cont’d. 4 6. Describe the characteristic light microscopy (LM), immunofluorescence (IF), and electron microscopy (EM) findings glomerular diseases. 7. Describe the renal features of systemic diseases (eg., HTN, DM, SLE, amyloidosis, vasculitides) that involve the kidney. 8. Describe the major causes and pathologic mechanisms of tubulointerstitial diseases. 9. Explain in the classification, etiology and pathogenesis of urinary tract infections.
- 5. Marc Imhotep Cray, MD Learning objectives cont’d. 5 10. Describe the gross and microscopic changes in acute pyelonephritis (PN) and chronic PN. 11. List the causes of renal papillary necrosis. Explain the causes and complications of obstructive uropathy, and hydronephrosis. 12. Describe the composition of the most common forms, favored sites for formation and complications of urolithiasis. 13. Describe the gross and microscopic features and typical pathologic and clinical findings of commonest benign and malignant tumors of the kidney. 14. List the congenital and cystic anomalies of the kidney.
- 6. Marc Imhotep Cray, MD Topics Discussion Outline 6 Scope of Problem Anatomy, Histology and Cell Biology Functions of kidney and lower urinary tract Overview of Renal Pathology Clinical Manifestations of Renal Diseases Glomerular Diseases Tubulointerstitial Diseases Vascular Diseases Obstructive uropathy, hydronephrosis & urolithiasis Renal Neoplasia Congenital and Cystic Diseases of Kidney
- 7. Marc Imhotep Cray, MD Scope of Problem 7 Centers for Disease Control and Prevention estimates in U.S. more than 10% of people 20 years and older (or more than 20 million individuals) have chronic kidney disease many more suffer from acute kidney injury annually thus, Clinicians of all specialties will encounter patients w renal disorders behooves all to be aware of various risk factors and causes of kidney disease (eg. hypertension and DM) Important b/c w early detection and appropriate management may be able to prevent or at least slow rate of progression to kidney failure or other complications NB: Diagnosing kidney disease is particularly challenging, as patients are typically asymptomatic until relatively advanced kidney failure is present.
- 8. Marc Imhotep Cray, MD Lecture 1: Scope of Problem Anatomy, Histology and Cell Biology Functions of kidney and lower urinary tract Overview of Renal Pathology Clinical Manifestations of Renal Diseases 8
- 9. Marc Imhotep Cray, MD Anatomy, Histology and Cell Biology 9 Basic structural and functional unit of kidney function is nephron A structure consisting of a tuft of capillaries termed glomerulus and a tubule Each human kidney has approx. 1 million nephrons Each nephron is composed of an initial filtering component (renal corpuscle [glomerulus& Bowman's capsule]) and a tubule specialized for reabsorption and secretion (renal tubule) NB: Understanding the complex functional organization of glomerulus is crucial for understanding both normal renal function and characteristics of different glomerular diseases.
- 10. Marc Imhotep Cray, MD Schematic of Gross Anatomy 10 Mohan H. Textbook of Pathology, 7th Ed. New Delhi: Jaypee Brothers Medical Publishers, 2015. Cross-section of kidney showing gross structures. Cross-section of kidney showing arterial blood supply Renal blood flow: renal artery segmental artery interlobar artery arcuate artery interlobular artery afferent arteriole glomerulus efferent arteriole vasa recta/ peritubular capillaries venous outflow
- 11. Marc Imhotep Cray, MD Kidney and lower urinary tract 11 Kidney (upper urinary tract) & lower urinary tract are functionally connected abnormalities in one component can affect function of other components of system Bladder, urethra, ureters, & renal pelvis (lower urinary tract) are lined by transitional epithelium (urothelium) subject to similar types of disease processes Widmaier EP, Raff H , Strang KT. Vander’s Human Physiology : The Mechanisms of Body Function, 14th ed. New York, NY: McGraw-Hill, 2016.
- 12. 12 Normal Kidney, Gross Klatt EC. Robbins and Cotran Atlas of Pathology, 3rd Ed. Philadelphia: Saunders, 2015.
- 13. 13 Normal Kidney, Gross cross section Klatt EC. Robbins and Cotran Atlas of Pathology, 3rd Ed. Philadelphia: Saunders, 2015.
- 14. 14 Normal Kidney, CT image Klatt EC. Robbins and Cotran Atlas of Pathology, 3rd Ed. Philadelphia: Saunders, 2015.
- 15. 15 Normal Kidney, microscopic Klatt EC. Robbins and Cotran Atlas of Pathology, 3rd Ed. Philadelphia: Saunders, 2015.
- 16. 16 Normal Kidney, microscopic Klatt EC. Robbins and Cotran Atlas of Pathology, 3rd Ed. Philadelphia: Saunders, 2015.
- 17. 17 Normal Kidney, microscopic HP Klatt EC. Robbins and Cotran Atlas of Pathology, 3rd Ed. Philadelphia: Saunders, 2015.
- 18. Marc Imhotep Cray, MD Structure & histology of renal corpuscle 18 Kelly CR, Landman J (Eds.). The Netter Collection of Medical Illustrations, 2Ed. Vol 5- Urinary System. Philadelphia: Saunders- Elsevier, 2012;21. Four layers make up filtering apparatus of glomerulus each of these can be affected in renal disease with effects on filtration: • basement membrane • blood vessel lining (endothelial cells) • podocytes (epithelial cells) • mesangium (smooth muscle-like cells with phagocytes) See: Histology of the glomerular filtration barrier.pdf
- 19. Marc Imhotep Cray, MD Glomerulus and glomerular capillary 19 Hammer GD & McPhee SJ (Eds.). Pathophysiology of Disease: An Introduction to Clinical Medicine, 7th Ed. New York, NY: McGraw-Hill Education, 2014; 457. A glomerulus consists of an afferent and an efferent arteriole and an intervening tuft of capillaries lined by endothelial cells and covered by epithelial cells that form a continuous layer with those of Bowman capsule and renal tubule. Space between capillaries in glomerulus is called mesangium. Material comprising a basement membrane is located between capillary endothelial cells and epithelial cells.
- 20. Marc Imhotep Cray, MD Detailed structure of glomerulus and glomerular filtration membrane composed of endothelial cell, basement membrane, and podocyte. 20 Hammer GD & McPhee SJ (Eds.). Pathophysiology of Disease: An Introduction to Clinical Medicine, 7th Ed. New York, NY: McGraw-Hill Education, 2014; 457. Glomerular capillaries have unique features not found in most peripheral capillaries. First, glomerular capillary endothelium is fenestrated. However, because endothelial cells have a coat of negatively charged glycoproteins and glycosaminoglycans, they normally exclude plasma proteins such as albumin. On other side of glomerular basement membrane are epithelial cells. Termed “podocytes” because of their numerous extensions or foot processes, these cells are connected to one another by modified desmosomes. Mesangium is an extension of GBM.
- 21. 21 (Shown on left) Anatomy of a normal glomerular capillary. Note fenestrated endothelium (EN), glomerular basement membrane (GBM), and epithelium with its foot processes (EP). Mesangium is composed of mesangial cells (MC) surrounded by extracellular matrix (MM) in direct contact with endothelium. Ultrafiltration occurs across glomerular wall and through channels in mesangial matrix into urinary space (US). (Shown on right) Typical localization of immune deposits and other pathologic changes. (1) Uniform subepithelial deposits as in membranous nephropathy. (2) Large, irregular subepithelial deposits or “humps” seen in acute postinfectious glomerulonephritis. (3) Subendothelial deposits as in diffuse proliferative lupus glomerulonephritis. (4) Mesangial deposits characteristic of immunoglobulin A nephropathy. (5) Antibody binding to glomerular basement membrane (as in Goodpasture syndrome) does not produce visible deposits, but a smooth linear pattern is seen on immunofluorescence. (6) Effacement of epithelial foot processes is common in all forms of glomerular injury with proteinuria. Hammer GD & McPhee SJ (Eds.). Pathophysiology of Disease: An Introduction to Clinical Medicine, 7th Ed. New York, NY: McGraw-Hill Education, 2014; 462.
- 22. 22 Normal glomerular capillary electron micrograph Rubin R, Strayer D (eds.) Rubin’s Pathology. Clinicopathologic Foundations of Medicine, 6th ed. Baltimore: Wolters Kluwer Health, 2012. This EM depicts a single capillary loop and adjacent mesangium. Capillary wall portion of lumen (L) is lined by a thin layer of fenestrated endothelial cytoplasm (high magnification next slide) that extends out from endothelial cell body (E) Endothelial cell body is in direct contact with mesangium, which includes the mesangial cell (M) and adjacent matrix Outer aspect of basement membrane (B) is covered by foot processes (F) from podocyte (P) that line urinary space (U)
- 23. 23 Electron micrograph of glomerular filter Molecules that pass from capillary lumen (CL) to urinary space (US) traverse fenestrations (F) of endothelial cell (E) trilaminar basement membrane (BM) (lamina rara interna [LRI], lamina densa [LD] and lamina rara externa [LRE]) and slit pore diaphragm (D) that connects podocyte foot processes (FP) Rubin R, Strayer D (eds.) Rubin’s Pathology. Clinicopathologic Foundations of Medicine, 6th ed. Baltimore: Wolters Kluwer Health, 2012. REMEMBER: Endothelial cells have a coat of negatively charged glycoproteins and glycosaminoglycans, they normally exclude plasma proteins such as albumin.
- 24. 24 Photo: Glomerulus in a human kidney scanning electron micrograph. From: Widmaier EP, Raff H & Strang KT. Vander’s Human Physiology: The Mechanisms Of Body Function, 13th Ed. New York, NY: McGraw-Hill Companies, 2014; 490. [Labeling mine] Foot processes Cell body of podocyte Filtration slits Bowman’s capsule podocytes with foot processes and filtration slits. Note the filtration slits between adjacent foot processes. The podocytes and their foot processes encircle the glomerular capillaries.
- 25. Marc Imhotep Cray, MD Functions of kidney and urinary tract 25 1. Excretion: of waste products and drugs this involves selective reabsorption and excretion of substances as they pass through nephron 2. Regulation: of body fluid volume and ionic composition kidneys have a major role in homeostasis and are also involved in maintaining acid–base balance Kidney participates in whole-body homeostasis via 4 main functions:
- 26. Marc Imhotep Cray, MD Functions of kidney cont’d. 26 3. Endocrine: kidneys are involved in synthesis of renin (which generates angiotensin I from angiotensinogen) thus has a role in blood pressure and sodium balance) Erythropoietin controls erythrocyte production; and Prostaglandins involved in regulation of renal function ADH retain water in body and constrict blood vessels 4. Metabolism: Vitamin D is metabolized to its active form kidney is a major site for the catabolism of low-molecular- weight proteins including several hormones such as insulin, PTH and calcitonin has ability to produce glucose through gluconeogenesis
- 27. Marc Imhotep Cray, MD Overview of Renal Pathology 27 Kidney consists of glomerular, vascular, tubular & interstitial anatomic compartments Renal diseases are best understood in relation to compartments affected & assoc. functional impairment Pathology of kidney can be organized into four anatomic categories= diseases of 1. glomeruli 2. tubules and 3. interstitium 4. vessels thus,
- 28. Marc Imhotep Cray, MD Overview (2) 28 Diseases that affect glomeruli most often have an immunologic etiology Glomerular function disrupted by diseases that alter glomerular structural arrangements seen w structural damage to basement membrane, endothelium, epithelium or mesangium whereas, Diseases that affect tubules and interstitium usually have an infectious or toxic (drugs) etiology Tubular function disrupted by metabolic insult to tubular cells hypoxia or toxins
- 29. Marc Imhotep Cray, MD Overview (3) 29 Early in disease process, predominantly affect one of four anatomic structures over time, however, entire kidney becomes diseased b/c of close structural & functional relationships in nephron tubules & interstitium usually affected together As both glomerular and tubular functions are highly dependent on adequate perfusion by blood if bld perfusion disrupted, both functions are impaired
- 30. Marc Imhotep Cray, MD Overview (4) 30 b/c of large physiologic reserve of kidneys, many diseases do not become clinically apparent until majority of organ is affected subtle abnormalities in laboratory findings* are only early indication of renal disease Recognition of patterns of abnormalities, pathologic findings, and clinical presentation are arguably more important to renal pathology than in any other organ system N.B. *Chronic kidney disease can be detected at an early stage by measuring GFR (nml 90–140 mL/ min men, and 80–125 mL/ min for women).
- 31. Marc Imhotep Cray, MD Overview(5)Types of renal failure syndromes 31 Partial renal failure syndromes affect only some elements of renal function There are four main types of partial renal failure: 1. Asymptomatic hematuria 2. Persistent proteinuria 3. Nephrotic syndrome 4. Nephritic syndrome Total renal failure syndromes all functions of nephron are impaired There are two types of total renal failure syndromes: 1. Acute renal failure (=AKI) sudden cessation of nephron function, affecting all nephrons at same time 2. Chronic renal failure (=CKD) nephrons are destroyed one by one over a long period
- 32. Marc Imhotep Cray, MD QUESTION: How does someone with a partial renal failure syndrome, such as a nephrotic syndrome, eventually develops total renal failure syndrome(CKD)? Steps in pathological progression follow… 32 In most glomerular diseases damage causing nephritic or nephrotic syndrome also eventually causes glomeruli to become completely scarred (hyalinized) leading to loss of individual nephrons ↓ Glomerular hyalinization is result of excessive production of mesangial matrix by mesangial cells over a long period of time ↓ Expanding mesangial matrix mass slowly but progressively crushes glomerular architecture out of existence until no blood flows through glomerular capillaries and no oxygenated blood passes into efferent arterioles and peritubular capillary systems ↓ Tubules are deprived of oxygenated blood, and tubular epithelial cells die irrevocably and become atrophic…cont’d. on next slide
- 33. Marc Imhotep Cray, MD Partial renal failure to complete renal failure explained cont’d. 33 Thus destruction of glomerulus leads to destruction of entire nephron unit ↓ As more and more nephrons are destroyed partial renal failure syndrome (nephritic or nephrotic) develops into total renal failure syndrome of chronic renal failure (CKD) ↓ This is associated with progressive shrinkage of kidney to form a small, scarred organ termed end-stage kidney ↓
- 34. 34Kibble J , Cannarozzi ML. Pathophysiology Flash Cards. New York: McGraw-Hill, 2013.
- 35. Marc Imhotep Cray, MD Overview (5) Renal Function Tests (RFT) 35 To assess renal function, tests are available to give information regarding following parameters: Renal blood flow Glomerular filtration (next slide) Renal tubular function Urinary outflow unhindered by any obstruction NB: Urinalysis and measurement of serum creatinine are initial steps in evaluation of renal disorders. RFT divided into 4 groups: 1. Urinalysis 2. Concentration & dilution tests 3. Blood chemistry 4. Renal clearance tests
- 36. Marc Imhotep Cray, MD Overview (6) Glomerular Filtration 36 Filtration Barrier Composed of 1) fenestrated capillary endothelium, 2) fused glomerular basement membrane, and 3) podocyte foot processes epithelial layer o capillary endothelium serves as a size barrier, while basement membrane contains heparan sulfate leads to a negative charge barrier preventing protein (albumin) filtration (=primary barrier) Glomerular Filtration Rate and Filtration Fraction GFR can be estimated by clearance of creatinine (CCr) Effective renal plasma flow (RPF) can be estimated by clearance of para- aminohippuric acid (CPAH) Filtration fraction(FF) = fraction of RPF filtered across glomerular capillaries GFR/RPF=FF
- 37. Marc Imhotep Cray, MD GFR estimated by CCr 37 True picture of status of renal function can be obtained by measuring GFR achieved clinically by measuring CCr, using a calculation based on Cr content of a 24-hour collection of urine and a single bld Cr estimation GFR nml range: 90–140 mL/ min men & 80–125 mL/ min women Blood levels of urea and creatinine do not rise above nml until GFR (CCr) has fallen below 50% nml thus , for many years of a chronic progressive kidney disease pt. may be asymptomatic or show only minor Sx or Sn o important to detect these early stages and to investigate cause and monitor rate of progression
- 38. Marc Imhotep Cray, MD Overview (7) Urinalysis & Urine Microscopy 38 …Urine microscopy: Presence of casts indicates that hematuria/pyuria is of glomerular or renal tubular origin Bladder cancer and kidney stones→hematuria, no casts Acute cystitis → pyuria, no casts RBC casts → glomerulonephritis, malignant hypertension Understanding basic urinalysis (UA) and urine microscopy helps interpret pathology of kidney, for instance… (See Urinalysis & Urine Microscopy. Pdf notes)
- 39. Marc Imhotep Cray, MD Overview (9) Renal Biopsy 39 In addition to RFT, renal biopsy is performed on selected pts w kidney disease to confirm Dx Indications include: hematuria, proteinuria, renal failure, transplant kidney surveillance…more Renal biopsy tissue sample is fixed in alcoholic solution and examined morphologically supported by special stains… 1. Light microscopy (LM) o Periodic acid-Schiff (PAS) stain for highlighting GBM o Silver impregnation to outline glomerular & tubular BM 2. Immunofluorescence (IF) to localize antigens, complements and immunoglobulins type of immunologic injury is assessed 3. Electron microscopy (EM) to see ultrastructure of glomerular changes exact site of immune-complex deposition is visualized
- 40. Marc Imhotep Cray, MD Overview (10) Definitions of key terms 40 Nephrotic syndrome: A kidney disorder affecting glomerulus, leading to proteinuria >3.5 g/day, hypoalbuminemia, generalized edema, and hyperlipidemia Nephrosis: nephropathy without inflammation or neoplasia Nephritic syndrome: A kidney disorder of oliguria, hematuria, edema, and hypertension, resulting from glomerulonephritis (also sub-nephrotic proteinuria) Nephritis: nephropathy with inflammation
- 41. Marc Imhotep Cray, MD Definitions of key terms cont’d. 41 Primary glomerular disease: A condition of kidney that leads to nephrosis, such as membranous glomerulopathy, minimal change disease, and focal segmental glomerulosclerosis or nephritis, such as postinfectious glomerulonephritis, rapidly progressive glomerulonephritis and IgA nephropathy (Berger disease) Secondary glomerular disease: Conditions that are systemic, infectious, and (or) toxic & affect kidney, leading to nephrosis or nephritis Some of causes are DM, SLE, amyloidosis, penicillamine, chronic hepatitis B infection, HIV, SBE…
- 42. Marc Imhotep Cray, MD Definitions of key terms cont’d. 42 Azotemia is a biochemical abnormality that refers to an elevation of blood urea nitrogen (BUN) and creatinine (Cr) levels, and is related to a ↓ glomerular filtration rate (GFR) Uremia is when azotemia becomes assoc. w a constellation of clinical signs (Sn) and symptoms (Sx) and biochemical abnormalities
- 43. Marc Imhotep Cray, MD Definitions of key terms cont’d. 43 Glomerulosclerosis versus glomerulonephritis: In glomerulosclerosis (sclerosis means hardening) there are sclerotic, scarred areas that lose ability to filter, secondary to capillary collapse In glomerulonephritis (-itis means inflammation), there is ongoing glomerular inflammation (=hypercellularity and leukocytosis) Crescentic: Used to describe appearance when inflammatory cells fill Bowman’s space leading to a crescent appearance NB: always indicative of a rapidly progressive glomerulonephritis (RPGN)=bad disease
- 44. Marc Imhotep Cray, MD Definitions of key terms cont’d. 44 Glomerulopathy: can include processes that are inflammatory or noninflammatory, however b/c term glomerulitis exists for inflammatory conditions, glomerulopathy sometimes carries a noninflammatory implication Regarding location of glomerular lesions: Subepithelial: between podocyte and GBM (on epithelial side of GBM) Subendothelial: between endothelium and GBM (on endothelial side of GBM)
- 45. Marc Imhotep Cray, MD Clinical Manifestations of Renal Diseases 45 Following is a discussion of clinical manifestations and syndromes of renal diseases with their defining features two most common syndromes assoc. w glomerular diseases, nephrotic and nephritic, are given most attention Azotemia and uremia (defined above) Asymptomatic hematuria or proteinuria Acute kidney injury Chronic kidney disease End-stage renal disease (ESRD) Renal tubular defects Urinary tract obstruction and renal tumors Nephrolithiasis (renal stones) Nephrotic syndrome Nephritic syndrome
- 46. Marc Imhotep Cray, MD Clinical Manifestations (2) 46 Azotemia =↑of BUN & Cr levels reflects a ↓ (GFR) GFR may be ↓ as a consequence of intrinsic renal disease or extrarenal causes Azotemia has three classifications, depending on causative origin o Prerenal azotemia is encountered when there is hypoperfusion of kidneys usually due to reduced extracellular fluid volume usually reversible if hypoperfusion is corrected in time o Primary renal azotemia (ARF [AKI]) typically leading to uremia intrinsic disease of kidney resultant of renal parenchymal damage • Causes include renal failure, glomerulonephritis, acute tubular necrosis, or any other kind of renal disease o Postrenal azotemia results when urine outflow is obstructed relief of obstruction is followed by correction of azotemia
- 47. Marc Imhotep Cray, MD Clinical Manifestations (3) 47 When azotemia gives rise to clinical manifestations and systemic biochemical abnormalities it becomes uremia= failure of renal excretory function + a host of metabolic & endocrine alterations In addition, uremia is characterized by secondary GI (e.g., uremic gastroenteritis), neuromuscular (e.g., peripheral neuropathy), and cardiovascular (e.g., uremic pericarditis) involvement
- 48. 48 Renal Failure and Uremia Manifestations Buja LM, Krueger GR. Netter’s Illustrated Human Pathology 2nd Ed. Philadelphia: Saunders-Elsevier, 2014. Inability to make urine & excrete nitrogenous wastes: Consequences (MAD HUNGER): Metabolic Acidosis Dyslipidemia (especially ↑triglycerides) Hyperkalemia Uremia—clinical syndrome marked by ↑ BUN: Nausea and anorexia Pericarditis Asterixis Encephalopathy Platelet dysfunction Na+/H2O retention (HF, pulmonary edema, hypertension) Growth retardation and developmental delay Erythropoietin failure (anemia) Renal osteodystrophy
- 49. Marc Imhotep Cray, MD Clinical Manifestations (5) 49 Asymptomatic hematuria or proteinuria, or a combination of these two represents a manifestation of subtle or mild glomerular abnormalities Acute kidney injury is characterized by rapid decline in GFR (within hours to days), w concurrent dysregulation of fluid & electrolyte balance, and retention of metabolic waste products normally excreted by kidney including urea and creatinine In its most severe forms it is manifested by oliguria or anuria (reduced or no urine flow) AKI can result from glomerular, interstitial, vascular or acute tubular injury
- 50. Marc Imhotep Cray, MD Clinical Manifestations (6) 50 Chronic kidney disease (chronic renal failure) is defined as: presence of a diminished GFR that is persistently <60 mL /minute for at least 3 months, from any cause, and/or persistent albuminuria CKD may present w clinically silent decline in renal excretory function in milder forms, and in more severe cases, by prolonged Sx and Sn of uremia It is end result of all chronic renal parenchymal diseases
- 51. Etiologies of CKD Buja LM, Krueger GR. Netter’s Illustrated Human Pathology 2nd Ed. Philadelphia: Saunders-Elsevier, 2014. Main groups of causes are: Chronic vascular disease (e.g. long-standing hypertension Disease of glomeruli, e.g. glomerulonephritis and diabetic glomerular disease Disease of tubules and interstitium infective, toxic and obstructive damage to tubules and renal papillae Some congenital kidney diseases, e.g. autosomal dominant polycystic kidney disease (ADPKD) Note: A kidney in which virtually all nephrons have been destroyed is called an end-stage kidney 46
- 52. Marc Imhotep Cray, MD Clinical Manifestations (8) 52 End-stage renal disease (ESRD) GFR is less than 5% of normal this is terminal stage of uremia NDD-CKD vs. ESRD The term "non-dialysis-dependent chronic kidney disease" (NDD-CKD) is used to encompass status of those persons w established chronic kidney disease (CKD) who do not yet require life-supporting treatments for kidney failure known as renal replacement therapy RRT, including maintenance dialysis or kidney transplantation Condition of individuals w CKD, who require either of two types of RRT (dialysis or transplant) referred to as the end-stage kidney disease (ESKD)
- 53. 53 Staging of chronic kidney disease Stevens A, Lowe J, Scott I. Core Pathology, 3rd Ed. St. Louis: Mosby-Elsevier, 2009. NB: Patients with CKD should be prescribed medication with care, as renal disease impairs excretion of certain drugs. Note: Stages 4 and 5 are considered to equate to irreversible CKD.
- 54. 54 End-stage renal disease, microscopic Klatt EC. Robbins and Cotran Atlas of Pathology, 3rd Ed. Philadelphia: Saunders, 2015.
- 55. Marc Imhotep Cray, MD Clinical Manifestations (11) 55 Renal tubular defects are dominated by nocturia, and electrolyte disorders (e.g., metabolic acidosis) A result of diseases that either directly affect tubular structures (e.g., nephronophthisis-medullary cystic disease complex) or cause defects in specific tubular functions o Latter can be inherited (e.g., familial nephrogenic diabetes, cystinuria, renal tubular acidosis [RTA]) or o acquired (e.g., lead nephropathy)
- 56. Marc Imhotep Cray, MD Clinical Manifestations (12) 56 Urinary tract obstruction and renal tumors have varied clinical manifestations based on specific anatomic location and nature of lesion Urinary tract infection (UTI) is characterized by bacteriuria and pyuria (bacteria and leukocytes in urine) Infection may be symptomatic or asymptomatic, and it may affect kidney (pyelonephritis) or bladder (cystitis) Nephrolithiasis (renal stones) is manifested by spasms of severe pain (renal colic) and hematuria, often w recurrent stone formation
- 57. Marc Imhotep Cray, MD Question 57 A patient with acute renal failure is referred for dialysis. The following are all indications for dialysis EXCEPT? A. Severe metabolic acidosis B. Uncontrollable hyperkalemia C. Pulmonary edema D. Pericarditis E. Anemias
- 58. Marc Imhotep Cray, MD Answer 58 Indications for dialysis include: Uncontrollable hyperkalemia Severe metabolic acidosis Pulmonary edema Overload of fluid not expected to respond to Tx w diuretics Uremic complications (pericarditis, encephalopathy, GI bleeding, platelet dysfunction…) and Intoxication, that is, acute poisoning w a dialyzable substances (eg. SLIME: salicylic acid, lithium, isopropanol, magnesium- containing laxatives, and ethylene glycol) Anemia in a pt. w ARF is not an indication for dialysis.
- 59. Marc Imhotep Cray, MD Nephrotic vs Nephritic Syndrome Capsule 59 Nephrotic Syndrome Proteinuria: b/c of disruption of glomerular charge barrier Hypoalbuminemia: b/c of proteinuria Edema: b/c of decreased plasma oncotic pressure from proteinuria Hyperlipidemia & hypercholesterolemia: b/c of ↑ in lipoprotein synthesis Nephritic Syndrome Oliguria & Azotemia: b/c of renal inflammation Hypertension: results from ↓clearance of sodium & water Hematuria: b/c of leakage of blood into Bowman capsule
- 60. Marc Imhotep Cray, MD Nephrotic syndrome pathophysiology 60 Heavy proteinuria, exceeds 3.5 g/day in nephrotic syndrome Nephrotic syndrome has diverse causes that share a common pathophysiology a derangement in capillary walls of glomeruli results in ↑ permeability to plasma proteins ↑ permeability of GBM may result from structural or physicochemical alterations With long-standing or heavy proteinuria, serum albumin is ↓ giving rise to hypoalbuminemia a drop in plasma colloid osmotic pressure which in turn leads to leakage of fluid from bld into extravascular spaces…cont’d next slide
- 61. Marc Imhotep Cray, MD Nephrotic syndrome cont’d. 61 … resulting ↓in intravascular volume and renal blood flow triggers ↑ release of renin from renal juxtaglomerular cells renin in turn stimulates angiotensin-aldosterone axis which promotes retention of salt and water by kidney This tendency is exacerbated by reductions in cardiac secretion of natriuretic factors ( ANP & BNP) attributed to ↓ intravascular volume In face of continuing proteinuria, salt and water retention further aggravates edema if unchecked may lead to development of generalized edema (termed anasarca) At onset, there is little or no azotemia, hematuria, or hypertension occurs
- 62. Marc Imhotep Cray, MD Nephrotic syndrome cont’d. 62 Genesis of hyperlipidemia is more murky presumably, hypoalbuminemia triggers ↑ synthesis of lipoproteins in liver, or massive proteinuria causes loss of an inhibitor of lipoprotein synthesis There is also is abnormal transport of circulating lipid particles and impairment of peripheral breakdown of lipoproteins associated lipiduria reflects ↑ permeability of GBM to lipoproteins
- 63. Marc Imhotep Cray, MD Nephrotic syndrome cont’d. 63 Hypercoagulability is a clinically significant manifestation of nephrotic syndrome caused by renal losses of proteins C & S and antithrombin III , as well as elevated serum fibrinogen and lipid levels Note: Thyroid-binding globulin is also lost, however, this does not appear to be clinically significant, as pts are euthyroid when free T4 is measured
- 64. Marc Imhotep Cray, MD Pathophysiology of nephrotic syndrome 64 Rubin R, Strayer D (eds.) Rubin’s Pathology. Clinicopathologic Foundations of Medicine, 6th ed. Baltimore: Wolters Kluwer Health, 2012. In nephrotic syndrome, RAS is activated, leading to salt and water retention and edema
- 65. Marc Imhotep Cray, MD Nephrotic syndrome cont’d. 65 Most important of primary glomerular lesions that lead to nephrotic syndrome are focal segmental glomerulosclerosis and minimal-change disease minimal-change disease is more important in children segmental glomerulosclerosis is more important in adults Nephrotic syndrome is also commonly seen in two other primary kidney diseases membranous nephropathy and membranoproliferative glomerulonephritis, and as a complication of systemic disease diabetes mellitus
- 66. Marc Imhotep Cray, MD Nephritic syndrome 66 Nephritic syndrome is a clinical entity caused by glomerular disease and is dominated by acute onset either grossly visible hematuria (RBCs in urine) or microscopic hematuria w dysmorphic red cells and red cell casts on urinalysis diminished GFR mild to moderate proteinuria (< 3.0 g/day), and Mild hypertension It is classic presentation of acute poststreptococcal glomerulonephritis Rapidly progressive glomerulonephritis (RPGN) is characterized as a nephritic syndrome with rapid decline in GFR (within hours to days)
- 67. Marc Imhotep Cray, MD Nephritic syndrome cont’d 67 Nephritic syndrome usually has an acute onset and is caused by inflammatory lesions of glomeruli lesions that cause nephritic syndrome have in common proliferation of cells within glomeruli, often accompanied by an infiltrate of leukocytes inflammatory reaction injures capillary walls permitting blood to pass into urine, and induces hemodynamic changes that lead to a reduction in GFR
- 68. Marc Imhotep Cray, MD Nephritic syndrome cont’d 68 Reduced GFR is manifested clinically by oliguria (or anuria), fluid retention, and azotemia Hypertension is a result of both fluid retention and augmented renin release from ischemic kidneys acute nephritic syndrome may be caused by primary glomerular diseases, such as postinfectious glomerulonephritis GN and various forms of crescentic GN, or as a result of systemic disorders such as systemic lupus erythematosus, amyloidosis, diabetes, hypertension
- 69. 69 Etiologies of most common two renal syndromes Syndrome Common Etiologies Uncommon Etiologies Nephrotic syndrome Minimal change disease, focal segmental glomerulosclerosis, diabetic nephropathy Membranous glomerulonephritis (GN)75% of cases are idiopathic, but 2° causes include SLE, penicillamine, gold, NSAIDs, HBV, HCV, syphilis, and malignancy Renal amyloidosis, SLE WHO Class V (membranous form) Nephritic syndrome Postinfectious GN; IgA nephropathy; RPGN, ANCA-associated, pauci- immune GN; SLE Membranoproliferative GN, HCV, cryoglobulinemia, Goodpasture’s syndrome, vasculitides, TTP, HUS, hereditary nephritis (Alport’s syndrome) Redrawn after: Le T and Bhushan V. First Aid for the Wards, 5th Ed. New York: McGraw-Hill, 2013.
- 70. Marc Imhotep Cray, MD Lecture 2: Glomerular Diseases 70
- 71. Marc Imhotep Cray, MD Glomerular Diseases 71 Terminology use to describe syndromes is explained in next 4 slides. Many renal disorders are caused by injury to glomerulus Glomeruli may be only major site of disease (1° glomerular disease; e.g., immunoglobulin [Ig]A nephropathy) or Part of a disease affecting several organs (2° glomerular disease; e.g., lupus glomerulonephritis) Signs (Sn) & symptoms (Sx) of fall into one of following categories: Asymptomatic proteinuria Asymptomatic hematuria Nephrotic syndrome Acute nephritic syndrome Rapidly progressive nephritic syndrome Chronic kidney injury ESRD
- 72. Marc Imhotep Cray, MD Terminology describing glomerular syndromes 72 All are histologic determinations after a renal biopsy: Focal vs diffuse: Defines number of glomeruli on biopsy affected If less than half glomeruli are affected it is focal If more than half are affected it is diffuse Segmental vs global: Defines how much of each individual glomerulus is affected, for each affected glomerulus If only part is affected then it is segmental If entire glomerulus is affected it is global Therefore, focal segmental glomerulosclerosis means less than half of glomeruli are affected [focal] and, of those affected, only part of each glomerulus is affected [segmental]
- 73. 73 Patterns of glomerular disease Modified from: Stevens A, Lowe J, Scott I. Core Pathology, 3rd Ed. St. Louis: Mosby-Elsevier, 2009; 367. Global: affecting the whole of the glomerulus uniformly. Segmental: affecting one glomerular segment, leaving other segments unaffected. Diffuse: affecting all glomeruli in both kidneys. Focal: affecting a proportion of glomeruli, others unaffected.
- 74. Marc Imhotep Cray, MD Terminology describing glomerular syndromes 74 Membranous vs proliferative vs membranoproliferative: Membranous, glomerular basement membrane (GBM) becomes thickened in parts thickenings appear as spikes & domes on microscopy b/c of bulging membrane Proliferative indicates cells are proliferating & numerous nuclei seen on microscopy from added cell count Membranoproliferative indicates membranous thickening & proliferation leads to a so-called tram track appearance b/c GBM is rebuilt on top of damaged deposits
- 75. Marc Imhotep Cray, MD Summary of glomerular disorders nomenclature TYPE CHARACTERISTICS EXAMPLE Focal < 50% of glomeruli are involved Focal segmental glomerulosclerosis Diffuse > 50% of glomeruli are involved Diffuse proliferative glomerulonephritis Proliferative Hypercellular glomeruli Membranoproliferative glomerulonephritis Membranous Thickening of glomerular basement membrane(GBM) Membranous nephropathy Primary glomerular disease 1° disease of kidney specifically impacting glomeruli Minimal change disease Secondary glomerular disease Systemic disease or disease of another organ system that also impacts glomeruli SLE, diabetic nephropathy Redrawn after: Le T, Bhushan V, et al. First Aid for the USMLE Step 1 2017. McGraw-Hill Education, 2017. If only part is affected then it is segmental If entire glomerulus is affected it is global
- 76. Marc Imhotep Cray, MD Clinical features of glomerulonephritis relate broadly to histological findings. 76 There are four general rules of thumb which, albeit not absolute, explain vast majority of clinical patterns of disease that relate to each of several types of glomerulonephritis. 1. Structural change in glomerular basement membrane (GBM) (usually thickening) or deposition of excessive mesangial matrix leads to increased loss of protein in urine, which if severe leads to the nephrotic syndrome.
- 77. Marc Imhotep Cray, MD Clinical features of glomerulonephritis relate to histological findings cont’d. 77 2. Glomerular damage associated with proliferation of endothelial or mesangial cells is associated with the development of microscopic hematuria or nephritic syndrome. 3. If there is both damage to GBM and cell proliferation, a mixed nephritic/ nephrotic syndrome is likely. 4. If damage to glomeruli is rapid and widespread, features of acute renal failure develop.
- 78. Marc Imhotep Cray, MD Acute versus Chronic Glomerulonephritis 78 Acute (and Rapidly Progressive) Glomerulonephritis There are several ways to classify acute GN Light microscopy (LM) essential for establishing areas of injury Circulating autoantibodies and measures of complement deposition combined w Immunofluorescence (IF) studies and Electron microscopy (EM) LM, EM & IF allow GN to be categorized into subgroups correlating w other features of disease three patterns emerge: 1. Antiglomerular basement membrane ( anti-GBM ) antibody disease 2. Immune complex glomerulonephritis 3. Anti-neutrophil cytoplasmic antibody (ANCA) disease or pauci-immune GN A brief description of each pattern with disease examples follows…
- 79. Marc Imhotep Cray, MD Acute versus Chronic GN cont’d. 79 1. Anti-GBM antibody disease (eg, Goodpasture syndrome): This disease results from development of circulating antibodies to an antigen intrinsic to GBM Binding of these pathologic anti-GBM antibodies to GBM causes a cascade of inflammation Light microscopy shows crescentic GN, and characteristic linear immunoglobulin deposition in glomerular capillaries is seen on immunofluorescence
- 80. Marc Imhotep Cray, MD Acute versus Chronic GN cont’d. 80 2. Immune complex glomerulonephritis: Immune complex deposition are seen in a variety of diseases On renal biopsy, granular immunoglobulin deposits are suggestive of immune complexes from underlying systemic disease A classic example is postinfectious GN in which there is cross- reactivity betw. an antigen of infecting organism and a host antigen resulting in deposition of immune complexes and complement in glomerular capillaries and mesangium o Resolution of glomerular disease typically occurs weeks after Tx of original infection Other examples include IgA nephropathy, lupus nephritis, & membranoproliferative GN
- 81. Marc Imhotep Cray, MD Acute versus Chronic GN cont’d. 81 3. Anti-neutrophil cytoplasmic antibody (ANCA) disease or pauci-immune GN: Characterized by a necrotizing GN but few or no immune deposits (hence, pauci-immune) seen on immunofluorescence or electron microscopy This pattern is typical of granulomatosis with angiitis, microscopic polyangiitis, or Churg-Strauss syndrome ANCA-negative pauci-immune necrotizing GN occurs less frequently but is also a well-described clinical entity
- 82. Marc Imhotep Cray, MD Acute versus Chronic GN cont’d. 82 Rapidly progressive glomerulonephritis (RPGN) A subset of acute GN in which there is a progressive and dramatic decline (weeks to months) in renal function often leading to complete renal failure and oliguria o It is a nonspecific final pathway in a variety of glomerular diseases Early disease can be subtle, but is marked by proteinuria and hematuria followed by ↓ GFR Often called “crescentic GN,” as characteristic finding on biopsy is cellular crescents in Bowman space Cellular crescents, visible on light microscopy, form in response to severe damage to glomerular capillaries N.B. Recovery without specific treatment is rare
- 83. Marc Imhotep Cray, MD Acute versus Chronic GN cont’d. 83 Chronic Glomerulonephritis Some patients with acute GN develop CKD slowly over a period of 5–20 years pathogenesis includes: Cellular proliferation, in either mesangium or capillary, is a pathologic structural hallmark in some of these cases whereas, others are notable for obliteration of glomeruli = sclerosing chronic GN includes both focal and diffuse subsets and yet, others display irregular subepithelial proteinaceous deposits w uniform involvement of individual glomeruli (membranous GN)
- 84. 84 Chronic glomerulonephritis, gross Klatt EC. Robbins and Cotran Atlas of Pathology, 3rd Ed. Philadelphia: Saunders, 2015.
- 85. Marc Imhotep Cray, MD Diagnostic features of glomerular diseases 85 I. Light microscopic (LM) features A. Increased cellularity Infiltration by leukocytes (e.g., neutrophils, monocytes, macrophages) Proliferation of “endocapillary” cells (i.e., endothelial and mesangial cells) Proliferation of “extracapillary” cells (i.e., epithelial cells) (crescent formation) B. Increased extracellular material Localization of immune complexes Thickening or replication of GBM Increases in collagenous matrix (sclerosis) Insudation (collection) of plasma proteins (hyalinosis) Fibrinoid necrosis Deposition of amyloid
- 86. Marc Imhotep Cray, MD Dx features of glomerular diseases (2) 86 II. Immunofluorescence (IF) features A. Linear staining of GBM Anti-GBM antibodies Multiple plasma proteins (e.g., in diabetic glomerulosclerosis) Monoclonal light chains (Amyloid nephropathy ) B. Granular immune complex staining Mesangium (e.g., IgA nephropathy) Capillary wall (e.g., membranous glomerulopathy) Mesangium and capillary wall (e.g., lupus glomerulonephritis) C. Irregular (fluffy) staining Monoclonal light chains (AL amyloidosis) AA protein (AA amyloidosis)
- 87. Marc Imhotep Cray, MD Dx features of glomerular diseases (3) 87 III. Electron microscopic features A. Electron-dense immune complex deposits Mesangial (e.g., IgA nephropathy) Subendothelial (e.g., lupus glomerulonephritis) Subepithelial (e.g., membranous glomerulopathy) B. GBM thickening (e.g., diabetic glomerulosclerosis) C. GBM replication (e.g., membranoproliferative glomerulonephritis) D. Collagenous matrix expansion (e.g., focal segmental glomerulosclerosis) E. Fibrillary deposits (e.g., amyloidosis) To view plates see: LM, IF and EM of Select Glomerular Diseases_pdf.
- 88. Marc Imhotep Cray, MD Mechanisms of Glomerular Injury & Disease 88 Immune mechanisms underlie most primary glomerular diseases & many secondary glomerular diseases Two mechanisms of antibody deposition in glomerulus have been established: 1. deposition of circulating antigen-antibody complexes in glomerular capillary wall or mesangium, and 2. antibodies reacting in situ within glomerulus either with fixed (intrinsic) glomerular antigens or with extrinsic molecules that are planted in glomerulus o Deposition of circulating immune complexes gives a granular immunofluorescence pattern o Anti-GBM antibody GN is characterized by a linear immunofluorescence pattern there is no immune deposit formation in this disease
- 89. Marc Imhotep Cray, MD Mechanisms of Glomerular Injury & Disease cont’d. 89 Once antigen-antibody complexes are deposited or formed in glomeruli they produce injury by activating complement and recruiting leukocytes Binding of immune complexes to Fc receptors on leukocytes also may contribute to activation of cells and injury Morphologically, affected glomeruli exhibit leukocytic infiltrates and proliferation of mesangial and parietal epithelial cells Electron microscopy reveals electron-dense immune deposits in one or more of three locations: 1. Betw. endothelial cells and GBM (subendothelial deposits) 2. Betw. outer surface of GBM and podocytes (subepithelial deposits), 3. In the mesangium
- 90. Marc Imhotep Cray, MD Antibody-mediated glomerular injury. Injury can result either from deposition of circulating immune complexes or from antibody-binding to glomerular components followed by formation of complexes in situ 90 Kumar V, Abbas AK, Aster JC. Robbins Basic Pathology, 10th ed. Philadelphia: Elsevier, 2018.
- 91. Marc Imhotep Cray, MD Two patterns of deposition of immune complexes as seen by immunofluorescence microscopy. (A) Granular, characteristic of circulating and in situ immune complex deposition. (B) Linear, characteristic of classic anti-glomerular basement membrane (anti-GBM) antibody glomerulonephritis. 91 Kumar V, Abbas AK, Aster JC. Robbins Basic Pathology, 10th ed. Philadelphia: Elsevier, 2018
- 92. 92 Localization of immune complexes in glomerulus: Kumar V, Abbas AK, Aster JC. Robbins and Cotran Pathologic Basis of Disease, 9th ed. Philadelphia: Saunders-Elsevier, 2015. 1. Subepithelial humps as in acute glomerulonephritis 2. Epimembranous deposits as in membranous nephropathy 3. Subendothelial deposits as in lupus nephritis & membranoproliferative glomerulonephritis 4. Mesangial deposits as in IgA nephropathy EN, Endothelium; EP, epithelium; GBM, glomerular basement membrane; LD, lamina densa; LRE, lamina rara externa; LRI, lamina rara interna; MC, mesangial cell; MM, mesangial matrix.
- 93. Marc Imhotep Cray, MD Clinical presentations of glomerulopathy / glomerulonephritis(GN) 93 Clinical presentation of all various types of GN can take one of six forms: 1. Nephritic syndrome: characterized by oliguria, hematuria, edema, and hypertension 2. Nephrotic syndrome: characterized by massive proteinuria, edema, hypoalbuminemia, and hyperlipidemia 3. Acute renal failure (now called acute kidney injury [AKI]) 4. Chronic renal failure (now called chronic kidney disease[CKD]) 5. Isolated (asymptomatic) proteinuria 6. Isolated (asymptomatic) hematuria Asymptomatic meaning no functional abnormalities assoc. w reduced GFR, edema, or hypertension.
- 94. Marc Imhotep Cray, MD Suggested approach to reading/studying the data that follows. 94 The discussions of glomerular disorders that follow are presented in a concise and consistent format. Data for each disease commences by presenting a trigger case illustrating the clinical relevance of the pathologic disorder under consideration. Then the etiology and epidemiology, pathology and pathogenesis, clinical manifestations, treatment options and prognosis are presented. Learners are encouraged read the trigger case with close attention and extract all data thought to be relevant to the diagnosis before proceeding to the knowledge-base. This approach should make for good practice in appreciating the intimate relationship of basic science pathology to clinical medicine.
- 95. Marc Imhotep Cray, MD Trigger Case 1 95 A 5-year-old boy presents to the emergency room with a 1-week history of generalized edema and fatigue. Your history reveals that he suffered from a viral URI 1 week before this visit. Serum and urine studies reveal massive proteinuria, hyperlipidemia, and hypoalbuminemia. You suspect that a renal biopsy would show normal-appearing glomeruli on electron microscopy except for fusion of the epithelial foot processes and you begin the child on prednisone. What is the Diagnosis?
- 96. Marc Imhotep Cray, MD Minimal Change Disease (Lipoid Nephrosis) 96 Etiology & Epidemiology Etio. unknown, but usually occurs following a viral URI also assoc. w Hodgkin disease & hypersensitivity reactions Most often seen in young children, but can occur in older children and adults Pathology LM: Normal-appearing glomeruli can see lipid accumulation in renal tubular cells EM: Fusion of epithelial foot processes
- 97. Marc Imhotep Cray, MD MCD cont’d. 97 Clinical Manifestations Nephrotic syndrome Complications include infection by gram-positive organism, thromboembolism, and protein malnutrition Treatment (Tx) & Prognosis (Px) Prednisone; cyclophosphamide or chlorambucil for steroid-resistant cases Response is excellent Note: Minimal change disease is the prototype of nephrotic syndrome
- 98. Marc Imhotep Cray, MD MCD (A) When viewed with a LM, silver methenamine–stained glomerulus appears normal, w a delicate basement membrane. (B) Schematic diagram illustrating diffuse effacement of foot processes of podocytes with no immune deposits. 98 Kumar V, Abbas AK, Aster JC. Robbins and Cotran Pathologic Basis of Disease, 9th ed. Philadelphia: Saunders-Elsevier, 2015.
- 99. 99 MCD, Electron micrograph Klatt EC. Robbins and Cotran Atlas of Pathology, 3rd Ed. Philadelphia: Saunders, 2015.
- 100. Marc Imhotep Cray, MD Trigger Case 2 100 A 40-year-old woman with a history of SLE presents to your office with a chief complaint of increased swelling in her legs. She had been referred by her primary care physician, who suspected a secondary illness to her lupus. Recent laboratory studies show proteinuria, hypoalbuminemia, hyperlipidemia, and hypercholesterolemia. You suspect that a renal biopsy would demonstrate immune complex deposition on electron microscopy as well as a “spike and dome” appearance on silver methenamine stain. What is the Diagnosis?
- 101. 101 Membranous Glomerulonephritis (also called M. Glomerulopathy or M. Nephropathy) Etio. & Epidem. An immune complex disease of unknown etiology Secondary disease seen in 10% of SLE patients (type V Lupus Nephritis) is sometimes assoc. w infections (eg, hepatitis B and C, syphilis, malaria), drugs (eg, gold salts, penicillamine, NSAIDs), or malignancy Incidence is highest in adults Pathology LM: Diffuse capillary wall thickening & basement membrane (BM) thickening IF: Granular pattern of IgG or C3 deposits (lumpy-bumpy) EM: Electron dense immune complex deposition in subepithelial locations within BM of glomerular capillary walls Silver methenamine stain: A spike-and-dome appearance resulting from extension of BM betw. and around immune deposits (spikes = basement membrane, domes = immune complex deposits)
- 102. Marc Imhotep Cray, MD Membranous Glomerulopathy cont’d. 102 Clinical Manifestations Nephrotic syndrome accompanied by azotemia Complications include renal vein thrombosis and higher incidence of occult neoplasms of lung, stomach, and colon Tx Cyclophosphamide or steroids ACE inhibitors (reduce urinary protein loss) renal transplantation for severe cases Note: Membranous glomerulopathy is second most common cause of nephrotic syndrome in adults, w focal segmental glomerulosclerosis (FSGS) recently becoming most common
- 103. 103 Membranous nephropathy A. Silver methenamine stain. Note marked diffuse thickening of capillary walls without an increase in number of cells. There are prominent “spikes” of silver-staining matrix (arrow) projecting from basement membrane lamina densa toward urinary space, which separate and surround deposited immune complexes that lack affinity for silver stain. B. EM showing electron-dense deposits(arrow) along epithelial side of basement membrane (B). Note effacement of foot processes overlying deposits. CL, Capillary lumen; End, endothelium; Ep, epithelium; US, urinary space. C. Characteristic granular immunofluorescent deposits of IgG along glomerular basement membrane. D. Diagrammatic representation of membranous nephropathy. Kumar V, Abbas AK, Aster JC. Robbins and Cotran Pathologic Basis of Disease, 9th ed. Philadelphia: Saunders-Elsevier, 2015.
- 104. Marc Imhotep Cray, MD Trigger Case 3 104 A 40-year-old HIV-positive man is admitted to the hospital complaining of generalized edema and fatigue. A complete history reveals that he is a habitual IV drug user. Laboratory studies show hypoalbuminemia, hyperlipidemia, proteinuria, and microscopic hematuria. You suspect that his current presentation is related to his HIV and you prepare the patient for a renal biopsy to determine the exact diagnosis. What is the Diagnosis?
- 105. Marc Imhotep Cray, MD Focal Segmental Glomerulosclerosis (FSGS) 105 Etio. & Epidem. Often idiopathic; has been assoc. w heroin use, HTN, prior GN and HIV infection (=collapsing glomerulopathy) Most often occurs in older patients Pathology LM: Sclerosis within capillary tufts of deep juxtaglomerular glomeruli w focal and segmental distribution hyalinosis (deposition of hyaline masses) also seen IF: granular mesangial fluorescence for IgM and C3 EM: Fusion of epithelial foot processes
- 106. Marc Imhotep Cray, MD FSGS cont’d. 106 Clinical Manifestations nephrotic syndrome more severe disease in HIV and IV drug users Lab findings: 80% have microscopic hematuria at presentation Tx & Px Prednisone Most patients progress to ESRD in 5–10 years Notes: FSGS most common cause of nephrotic syndrome in adults in U.S. b/c of focal nature of FSGS, early cases can be difficult to distinguish from MCD (How might one differentiate the two clinically?) FSGS, w no cellular proliferation, is different from focal segmental glomerulonephritis (FSGN), which involves cellular proliferation
- 107. Marc Imhotep Cray, MD FSGS, microscopic (PAS stain) 107 A Low-power view showing segmental sclerosis in one of three glomeruli (at 3 o’clock) B High-power view showing hyaline insudation (arrow) and lipid (small vacuoles) in sclerotic area Kumar V, Abbas AK, Aster JC. Robbins and Cotran Pathologic Basis of Disease, 9th ed. Philadelphia: Saunders-Elsevier, 2015.
- 108. Marc Imhotep Cray, MD Collapsing Glomerulopathy 108 Visible are retraction of glomerular tuft (arrows), narrowing of capillary lumens, proliferation and swelling of visceral epithelial cells (double arrows), and prominent accumulation of intracellular protein absorption droplets in the visceral epithelial cells (arrowheads). Kumar V, Abbas AK, Aster JC. Robbins and Cotran Pathologic Basis of Disease, 9th ed. Philadelphia: Saunders-Elsevier, 2015. A morphologic variant of FSGS, characterized by retraction and/or collapse of entire glomerular tuft May be idiopathic, but it also has been assoc. w some drug toxicities (e.g., pamidronate) Most characteristic lesion of HIV-associated nephropathy Typically assoc. w prominent tubular injury It has a very poor prognosis
- 109. Marc Imhotep Cray, MD Trigger Case 4 109 A 60-year-old African Americans man with a 20-year history of type II DM presents for a nephrology consult after his primary care physician found a progressive increase of proteinuria in recent laboratory studies. These laboratory studies also showed hyperlipidemia and hypercholesterolemia. On physical examination, the patient has bilateral diabetic retinopathy and 2+ edema in both legs. You start the patient on an ACE inhibitor and you suspect that a renal biopsy would show Kimmelstiel-Wilson nodules. What is the Diagnosis?
- 110. Marc Imhotep Cray, MD Diabetic Nephropathy 110 Etio. & Epidem. Assoc. w long-standing diabetes T1DM carries 30%–40% chance of diabetic nephropathy after 20 years T2DM carries 15%–20% chance after 20 years however, b/c there are more pts w type II diabetes ESRD is more prevalent among type II diabetics higher risk of developing diabetic nephropathy among men, African Americans and Native Americans w DM Pathology LM: Increase in mesangial matrix, resulting in either diffuse glomerulosclerosis (diffusely distributed ↑ in mesangial matrix) or nodular glomerulosclerosis (Kimmelstiel- Wilson nodules—nodular accumulations of mesangial matrix material) EM: Striking ↑ in GBM thickening
- 111. Marc Imhotep Cray, MD Diabetic Nephropathy cont’d. 111 Clinical Manifestations Nephrotic syndrome diabetic retinopathy is invariably present Lab findings: Microalbuminuria (early sign), proteinuria (late sign) Tx & Px Strict glycemic control Treatment of hypertension and microalbuminuria w ACE inhibitors during early stages to slow progression Often progresses to ESRD and dialysis Note: Diabetic nephropathy is most common cause of ESRD in U.S.
- 112. 112 Nodular glomerulosclerosis, microscopic Klatt EC. Robbins and Cotran Atlas of Pathology, 3rd Ed. Philadelphia: Saunders, 2015.
- 113. 113 Diffuse glomerulosclerosis, microscopic Klatt EC. Robbins and Cotran Atlas of Pathology, 3rd Ed. Philadelphia: Saunders, 2015.
- 114. 114 A 60-year-old man complains of chronic back pain and fatigue, excessive urination, and increased thirst. X-ray examination reveals numerous lytic lesions in the lumbar vertebral bodies. Laboratory studies show hypoalbuminemia, mild anemia, and thrombocytopenia. Urinalysis displays 4+ proteinuria. A monoclonal immunoglobulin light-chain peak is demonstrated on serum electrophoresis. A bone marrow biopsy discloses foci of plasma cells, which account for 20% of all hematopoietic cells. A kidney biopsy is obtained (shown in image). Which of the following is the most likely cause of nephrotic syndrome in this patient? A. Amyloid nephropathy B. Crescentic glomerulonephritis C. IgA nephropathy (Berger disease) D. Membranous glomerulonephritis E. Nodular glomerulosclerosis (Kimmelstiel-Wilson disease) Question
- 115. Marc Imhotep Cray, MD 115 Diagnosis (A) , Amyloid nephropathy, multiple myeloma Neoplastic plasma cells typically secrete a homogeneous immunoglobulin chain, which can be detected in serum or urine by electrophoresis. Amyloid nephropathy is caused by the deposition of secreted light chains in the glomerular basement membranes and mesangial matrix. Amorphous acellular material expands the mesangium and obstructs the glomerular capillaries. Deposits of AL amyloid may also appear in the tubular basement membranes and in the walls of renal vessels. Renal amyloidosis usually presents with nephrotic syndrome. The deposits of amyloid may take on a nodular appearance, reminiscent of Kimmelstiel-Wilson lesion of diabetic glomerulosclerosis (choice E). However, amyloid deposits are not PAS positive and are identifiable by Congo red staining with characteristic apple-green birefringence. IgA nephropathy (choice C) and membranous glomerulonephritis (choice D) are unrelated to light-chain disease.
- 116. 116 Renal Amyloidosis Etiology: Amyloidosis is complication of chronic inflammatory disorders, such as RA, osteomyelitis or CA= multiple myeloma etc. kidneys, liver, spleen, and adrenals most common organs involved accumulation of proteins in form of abnormal, insoluble fibers=amyloid fibrils Pathology: Amyloidosis leads to nephrotic syndrome and renal failure LM-Congo red stain shows apple-green birefringence under polarized light due to amyloid deposition in mesangium Diagnosis: Congo red staining or tissue biopsy gold standard methods of Dx Amyloid nephropathy. In a section stained w Congo red and examined under polarized light, amyloid deposits in glomerulus and adjacent arteriole show a characteristic apple-green birefringence Rubin R, Strayer D (eds.) Rubin’s Pathology. Clinicopathologic Foundations of Medicine, 6th ed. Baltimore: Wolters Kluwer Health, 2012.
- 117. 117 Amyloid nephropathy Disorder is initially assoc. w accumulation of characteristic fibrillar deposits in mesangium. These inert masses, which are fibrillar by EM, extend along inner surface of basement membrane (BM), frequently obstructing capillary lumen. Focal extension of amyloid through BM may elevate epithelial cell, in which case irregular spikes are seen along outer surface of BM Amyloid nephropathy, EM Deposits of fibrils (10 nm diameter) in a glomerulus adjacent to podocyte cytoplasm with effaced foot processes. Rubin R, Strayer D (eds.) Rubin’s Pathology. Clinicopathologic Foundations of Medicine, 6th ed. Baltimore: Wolters Kluwer Health, 2012.
- 118. Marc Imhotep Cray, MD Trigger Case 5 118 A 45-year-old white woman with a 15-year history of SLE presents to the nephrologist after her primary care physician found hematuria and proteinuria on a routine urinalysis. The patient has edema of the ankles on physical examination and she is experiencing a SLE flare-up with an extensive malar rash visible over her face. A renal biopsy is obtained and examination by light microscopy reveals wire-loop abnormalities. You examine the patient’s current immunosuppressive therapy to see what additional therapies should be added. What is the Diagnosis?
- 119. Marc Imhotep Cray, MD Lupus Nephropathy 119 Etiology Renal component of systemic lupus erythematosus Pathology Five distinct renal histologic patterns: (1) Type I: normal (2) Type II (mesangial form): focal & segmental glomerular involvement w ↑ in mesangial matrix (3) Type III (focal proliferative form): involves less than half of glomeruli, causing extensive damage to individual glomeruli (4) Type IV (diffuse proliferative form): most severe form involving all glomeruli w marked inflammation, mesangial proliferation, and scarring o LM: wire-loop abnormality caused immune complex deposition and gross thickening of GBM o EM: endothelial cell proliferation o IF: marked subendothelial immune complex deposition= granular pattern (5) Type V (membranous form): similar to membranous glomerulonephritis
- 120. Marc Imhotep Cray, MD Lupus Nephropathy cont’d. 120 Clinical Manifestations Type I: No clinical findings Types II and III: Mild to moderate proteinuria and hematuria Type IV: Combination of nephrotic and nephritic syndromes Type V: Nephrotic syndrome Tx Types I and II: No treatment required Types III, IV, and V: Immunosuppression (corticosteroids, cyclophosphamide, and/or azathioprine) Note: Renal lesion severity often determines overall prognosis of SLE patients
- 121. 121 A, Focal proliferative glomerulonephritis, with two focal necrotizing lesions at the 11 0'clock and 2 0'clock positions (H&E stain). Extracapillary proliferation is not prominent in this case. B, Diffuse proliferative glomerulonephritis. Note marked increase in cellularity throughout glomerulus (H&E stain). Lupus nephritis Kumar V, Abbas AK, Aster JC. Robbins and Cotran Pathologic Basis of Disease, 9th ed. Philadelphia: Saunders-Elsevier, 2015.
- 122. 122Kumar V, Abbas AK, Aster JC. Robbins and Cotran Pathologic Basis of Disease, 9th ed. Philadelphia: Saunders-Elsevier, 2015. Lupus nephritis C, Lupus nephritis showing a glomerulus w several 'Wire loop" lesions representing extensive subendothelial deposits of immune complexes (PSA stain). D, EM of a renal glomerular capillary loop from a patient w SLE nephritis. Subendothelial dense deposits (arrowheads) correspond to "wire loops" seen by light microscopy. B (with arrow) refers to the basement membrane.
- 123. 123 E, Deposition of lgG antibody in a granular pattern, detected by immunofluorescence. Lupus nephritis Kumar V, Abbas AK, Aster JC. Robbins and Cotran Pathologic Basis of Disease, 9th ed. Philadelphia: Saunders-Elsevier, 2015.
- 124. Marc Imhotep Cray, MD Trigger Case 6 124 A 10-year-old girl presents to the clinic complaining of eye swelling. You note that the child was seen 3 weeks ago in clinic for a chief complaint of sore throat. Upon taking a history and performing a physical, you find that the patient has pronounced periorbital edema, has been urinating very little despite adequate fluid intake, and has a blood pressure of 150/90. Laboratory findings include azotemia, hematuria, red cell casts in the urine, and an elevated ASO antibody titer. You reassure the parents that their child’s condition will likely resolve on its own. What is the Diagnosis?
- 125. Marc Imhotep Cray, MD Poststreptococcal Glomerulonephritis (Acute Proliferative GN/ Acute GN) 125 Etio. & Epidem. Most frequently seen in children (6-10 yrs.) following infection w nephritogenic strains of group A β-hemolytic streptococci decreasing in frequency in U.S but, fairly common disorder worldwide Pathology Gross : Characterized by intense inflammatory reaction involving all glomeruli in both kidneys resulting in punctate hemorrhages on kidney surfaces LM: Enlarged, hypercellular, swollen glomeruli w proliferation of mesangial and endothelial cells; normal GBM thickness EM: Electron-dense humps on the epithelial side of the basement membrane (subepithelial localization) IF: Coarse granular immunofluorescence for IgG or C3 (lumpy-bumpy)
- 126. Marc Imhotep Cray, MD Poststreptococcal GN cont’d. 126 Clinical Manifestations Nephritic syndrome & periorbital edema; pulmonary congestion not uncommon Lab findings: UA RBCs and/or red cell casts, decreased serum C3, elevated ASO antibody titer (evidence of recent streptococcal infection) Tx & Px Resolves spontaneously More than 95% of affected children eventually recover renal function w conservative therapy aimed at maintaining sodium and water balance In adults disease is less benign in up to 40% affected in epidemics fail to resolve quickly Note: Poststreptococcal glomerulonephritis is an immune complex disease w antigen-antibody complex of streptococcal origin and is prototype of nephritic syndrome & prototypical glomerular disease of immune complex etiology
- 127. 127 Nonstreptococcal Acute Glomerulonephritis (Postinfectious Glomerulonephritis) A similar form of glomerulonephritis occurs sporadically in assoc. w other infections, including those of bacterial (e.g., staphylococcal endocarditis, pneumococcal pneumonia, and meningococcemia), viral (e.g., hepatitis B, hepatitis C, mumps, HIV infection, varicella, and infectious mononucleosis), and parasitic (malaria, toxoplasmosis) origin In these settings, granular immunofluorescent deposits and subepithelial humps characteristic of immune complex nephritis are present Postinfectious glomerulonephritis due to staphylococcal infections differs by sometimes producing immune deposits containing IgA rather than IgG
- 128. 128 A Normal glomerulus. B Glomerular hypercellularity is due to intracapillary leukocytes and proliferation of intrinsic glomerular cells. C Typical electron-dense subepithelial “hump” and a neutrophil in lumen. D Immunofluorescent stain demonstrates discrete, coarsely granular deposits of complement protein C3 (stain for IgG was similar), corresponding to “humps” illustrated in part C. Acute proliferative glomerulonephritis Kumar V, Abbas AK, Aster JC. Robbins and Cotran Pathologic Basis of Disease, 9th ed. Philadelphia: Saunders-Elsevier, 2015.
- 129. Marc Imhotep Cray, MD Trigger Case 7 129 A 40-year-old man is admitted to the hospital with complaints of blood in his sputum and urine. A thorough history also reveals fever, malaise, and a 10-pound weight loss over the past month. On physical examination, you find that his blood pressure is 160/95 and that he has several abnormal lung sounds. A urine dipstick demonstrates hematuria. CXR reveals several nodular lesions and blood tests show the presence of C-ANCA and an elevated ESR. You start the patient on a high dose of corticosteroids and you suspect that a renal biopsy would demonstrate crescent moon shapes between the Bowman capsule and the glomerular tuft. What is the Diagnosis?
- 130. 130 Rapidly Progressive (Crescentic) Glomerulonephritis RPGN is a clinical syndrome assoc. w severe glomerular injury, but does not denote a specific etiologic form of GN Type 1=20% (Anti-GBM antibody-mediated disease); Idiopathic or Goodpasture syndrome (=Ab against BM lung alveoli hemoptysis) Type 2=25% (immune complexes): Idiopathic; postinfectious GN; SLE; IgA nephropathy; Henoch- Schönlein purpura Type 3=55% (pauci-immune type): Idiopathic; ANCA-assoc. vasculitides= Wegener granulomatosis; microscopic polyangiitis etc. Pathology LM: Formation of crescent-moon shape betw. Bowman capsule and glomerular tuft resulting from deposition of fibrin in Bowman space and proliferation of parietal epithelial cells of Bowman capsule IF: Type 1, linear pattern deposits of IgG & C3; Type 2=granular deposits IgG &C3
- 131. Marc Imhotep Cray, MD RPGN cont’d. 131 Clinical Manifestations Nephritic syndrome progressing rapidly to renal failure within months Sn & Sx specific to each etiology (eg, hemoptysis and anti- GBM antibodies in Goodpasture syndrome) Tx & Px Treat w diuretics and often eventual dialysis immunosuppression if appropriate for underlying cause may require transplantation Rapid course to renal failure Note: RPGN refers to a syndrome assoc. w severe and progressive glomerular injury It encompasses many different etiologies
- 132. Marc Imhotep Cray, MD RPGN Schematic 132 Rapidly proliferating epithelial crescent crushes glomerular tuft, which may show a range of changes including o focal proliferative GN, o segmental tuft necrosis, o mesangiocapillary GN, or o acute proliferative GN Note: An epithelial crescent is result of breaks in GBM permitting leak of fibrin, blood proteins and WBCs (particularly monocyte/ macrophages) into urinary space Stevens A, Lowe J, Scott I. Core Pathology, 3rd Ed. St. Louis: Mosby- Elsevier, 2009.
- 133. 133 Rapidly progressive glomerulonephritis, microscopic Klatt EC. Robbins and Cotran Atlas of Pathology, 3rd Ed. Philadelphia: Saunders, 2015.
- 134. Marc Imhotep Cray, MD RPGN, Electron micrograph 134 EM showing characteristic wrinkling of GBM w focal disruptions (arrows). Kumar V, Abbas AK, Aster JC. Robbins and Cotran Pathologic Basis of Disease, 9th ed. Philadelphia: Saunders-Elsevier, 2015.
- 135. 135 RPGN, immunofluorescence Klatt EC. Robbins and Cotran Atlas of Pathology, 3rd Ed. Philadelphia: Saunders, 2015.
- 136. Marc Imhotep Cray, MD Trigger Case 8 136 A 10-year-old boy brought to the clinic by his mother complaining of a red tinge to his urine. A more detailed history reveals that he was diagnosed with mild nerve deafness 2 years earlier and also developed posterior cataracts 1 year ago. Laboratory studies confirm hematuria as well as the presence of erythrocyte casts. You begin to wonder if his conditions may be related to a genetic disorder. What is the Diagnosis?
- 137. Marc Imhotep Cray, MD Alport Syndrome 137 Etiology: Genetic disorder w heterogenous inheritance (usually X-linked dominant) results in mutation of α-5 chain of type IV collagen caused by mutations in COL4A3, COL4A4, and COL4A5, three of six genes involved in basement membrane (type IV) collagen biosynthesis Pathology EM: Irregular foci of thickening or attenuation in GBM with longitudinal splitting of lamina densa Clinical Manifestations: Triad of nephritis, nerve deafness, and various eye disorders (cataracts, lens dislocation, corneal dystrophy) often initially presents w hematuria and RBC casts Treatment ACE inhibitors; renal transplantation
- 138. 138 Alport syndrome, microscopic Klatt EC. Robbins and Cotran Atlas of Pathology, 3rd Ed. Philadelphia: Saunders, 2015.
- 139. Marc Imhotep Cray, MD Trigger Case 9 139 A 25-year-old woman with a history of SLE is admitted to the hospital with generalized edema, malaise, and fatigue. You take a thorough history, which reveals that she had cold symptoms 2 weeks earlier. Laboratory studies show hypoalbuminemia, hypercholesterolemia, proteinuria, and low complement levels. When a renal biopsy shows reduplication of the basement membrane on electron microscopy, you adjust the patient’s current corticosteroid dose and decide to add an antiplatelet drug to her regimen. What is the Diagnosis?
- 140. Marc Imhotep Cray, MD Diffuse Membranoproliferative GN (Mesangiocapillary GN) 140 Etio. and Epidem. Assoc. w inherited complement component deficiency Type I MPGN (90%) is seen in SLE, hepatitis B and C, and involves classic & alternative pathway activation Type II MPGN (10%) (dense deposit disease) dysregulation of alternative complement pathway Most patients are under the age of 30 Pathology Types I and II: LM: reduplication of basement membrane (splitting) and lobular proliferation of mesangial matrix into capillary loops (tram track appearance) Type I: EM: subendothelial electron-dense deposits Type II: EM: dense deposit of homogeneous material within GBM
- 141. Marc Imhotep Cray, MD Membranoproliferative GN cont’d. 141 Clinical Manifestations Type I: Commonly presents w nephrotic syndrome Type II: Commonly presents w hematuria and chronic renal failure Lab Findings: Low complement levels (↓ C3) , ↑BUN and Cr, RBCs and/or RBC casts in urine Tx & Px: Corticosteroids (poor response) and immunosuppression if appropriate for underlying cause Type II, particularly, poor prognosis slowly progresses to CKD 50% develop CKD within 10 year Both types have high incidence of recurrence of disease in transplanted kidneys
- 142. Marc Imhotep Cray, MD Schematic representation of MPGN 142 Patterns in two types of MPGN In type I there are subendothelial deposits Type II is characterized by intramembranous dense deposits (=dense-deposit disease) In both, BM appear split when viewed in light microscope Kumar V, Abbas AK, Aster JC. Robbins and Cotran Pathologic Basis of Disease, 9th ed. Philadelphia: Saunders-Elsevier, 2015.
- 143. 143 MPGN, microscopic Klatt EC. Robbins and Cotran Atlas of Pathology, 3rd Ed. Philadelphia: Saunders, 2015.
- 144. Marc Imhotep Cray, MD A MPGN, type I. Note discrete electron-dense deposits (arrows) incorporated into glomerular capillary wall betw. duplicated (split) basemen membranes (double arrows), and in mesangial regions (M); CL, Capillary lumen. B Dense-deposit disease (type II MPGN). There are dense homogeneous deposits within basement membrane. In both, mesangial interposition gives appearance of split BM when viewed in LM 144 Kumar V, Abbas AK, Aster JC. Robbins and Cotran Pathologic Basis of Disease, 9th ed. Philadelphia: Saunders-Elsevier, 2015.
- 145. Marc Imhotep Cray, MD Question 145 A 26-year-old woman with a history of mitral valve prolapse comes in with 1 week of fever that started 3 days after a dental procedure. Her urine contains red cells and her rheumatoid factor is elevated. Which of the following serologic abnormalities is expected to be present? A. Anti-GBM antibody B. Low serum complement levels C. Antineutrophil cytoplasmic antibody D. Elevated IgA levels
- 146. Marc Imhotep Cray, MD Answer 146 The answer is B. (Robbins, 9th/e, p 926.) This clinical description is classic for membranoproliferative glomerulonephritis, which is associated with bacterial endocarditis. Low complement levels are typically found in membranoproliferative glomerulonephritis, but not in the other disorders listed. Choice A. describes anti-GBM antibody disease or Goodpasture’s syndrome when the lung is involved. Choice C. describes pauci-immune glomerulonephritis, such as Wegener’s. Choice D. describes IgA nephropathy.
- 147. Marc Imhotep Cray, MD Trigger Case 10 147 A 15-year-old Asian boy presents to the emergency room complaining of blood in his urine. Upon taking a complete history, you learn that he has also been suffering from fevers, myalgias, and arthralgias for the last 2 days. Serum studies reveal increased serum IgA levels and normal serum complement levels. You begin him on prednisone and you suspect that he is afflicted with the most common form of acute glomerulonephritis in the United States. What is the Diagnosis?
- 148. Marc Imhotep Cray, MD IgA Nephropathy (Berger Disease) 148 Etio. & Epidem. Primary renal disease of IgA deposition in glomerular mesangium can manifest after infection (viral URI, GI infection, flu-like syndrome) or can be a component of Henoch-Schönlein purpura Most commonly seen in children and young adults w men affected more often than women Pathology LM: mesangial widening and focal and segmental inflammation EM: Mesangial deposits of IgA
- 149. Marc Imhotep Cray, MD IgA Nephropathy (Berger Disease) cont’d. 149 Clinical Manifestations Presents w recurrent hematuria (red or cola-colored urine) 1–2 days after an infection Lab findings: ↑ serum IgA level (50% of cases), nml serum complement levels Tx & Px Prednisone Can progress to chronic renal failure 25-50% go on to develop CKD within 20 yrs. Note: Berger disease is most common form of acute GN in U.S. and worldwide
- 150. 150 IgA nephropathy (Berger disease), microscopic Klatt EC. Robbins and Cotran Atlas of Pathology, 3rd Ed. Philadelphia: Saunders, 2015.
- 151. 151 IgA nephropathy, immunofluorescence Klatt EC. Robbins and Cotran Atlas of Pathology, 3rd Ed. Philadelphia: Saunders, 2015.
- 152. 152Rubin R, Strayer D (eds.) Rubin’s Pathology. Clinicopathologic Foundations of Medicine, 6th ed. Baltimore: Wolters Kluwer Health, 2012. Algorithm demonstrating integration of pathologic findings with clinical data to make a diagnosis of a specific form of primary or secondary glomerulonephritis.
- 153. Nephritic syndrome—due to GBM disruption. Hypertension, ↑ BUN and creatinine, oliguria, hematuria, RBC casts in urine. Proteinuria often in the subnephrotic range (< 3.5 g/day) but in severe cases may be in nephrotic range. • Acute poststreptococcal glomerulonephritis • Rapidly progressive glomerulonephritis • IgA nephropathy (Berger disease) • Alport syndrome • Membranoproliferative glomerulonephritis Nephritic-nephrotic syndrome—severe nephritic syndrome with profound GBM damage that damages the glomerular filtration charge barrier → nephrotic-range proteinuria (> 3.5 g/day) and concomitant features of nephrotic syndrome. Can occur with any form of nephritic syndrome, but is most commonly seen with: • Diffuse proliferative glomerulonephritis • Membranoproliferative glomerulonephritis • Lupus Nephropathy Type IV Nephrotic syndrome—podocyte disruption → charge barrier impaired. Massive proteinuria (> 3.5 g/day) with hypoalbuminemia, hyperlipidemia, edema. May be 1° (eg, direct podocyte damage) or 2° (podocyte damage from systemic process [eg, diabetes]). • Focal segmental glomerulosclerosis (1° or 2°) • Minimal change disease (1° or 2°) • Membranous nephropathy (1° or 2°) • Amyloidosis (2°) • Diabetic glomerulonephropathy (2°) Glomerular diseases capsule 0.25 3.5 > 3.5 GRAMS OF PROTEIN EXCRETED PER DAY (g/day)
- 154. Marc Imhotep Cray, MD Lecture 3: Urinary Tract Infections Tubulointerstitial diseases Obstructive uropathy, hydronephrosis & urolithiasis 154
- 155. Marc Imhotep Cray, MD Urinary Tract Infections 155 A urinary tract infection (UTI) is an infection that affects part of urinary tract When it affects lower urinary tract it is known as a bladder infection (cystitis) When it affects upper urinary tract it is known as kidney infection (pyelonephritis [discussed under tubulointerstitial diseases] ) Cystitis Clinical features: dysuria in absence of vaginal discharge and significant pain, frequency, urgency, suprapubic pain
- 156. Marc Imhotep Cray, MD UTI, Cystitis cont’d. 156 Etio-pathogenesis: Bacteria gain access to urinary tract via urethra Cystitis most frequently involves normal colonic flora o Escherichia coli is most common cause (approx. 80%) o Proteus, Klebsiella, and Enterobacter are also implicated o Staphylococcus saprophyticus causes 10% to 15% of infections in young women o Nosocomial cystitis is frequently caused by Pseudomonas or Staphylococcus aureus
- 157. Marc Imhotep Cray, MD UTI, Cystitis cont’d. 157 Epidemiology Women have a higher incidence of infection b/c they have shorter urethras Other risk factors include sexual activity, pregnancy, urinary obstruction, neurogenic bladder, and vesicoureteral reflux-8 Diagnostic findings Characteristic clinical features are present Pyuria (more than 8 leukocytes/high-power field) Bacterial culture yields >105 organisms/mL Treatment antibiotics Recurrent cystitis may require prophylactic antibiotics
- 158. 158 Acute cystitis Patient died 2 days after surgery, and cystitis was caused by an indwelling catheter A. Several foci of hemorrhage are seen on hyperemic bladder mucosa B. Foci of mucosal hemorrhage C. Acute cystitis. Polymorphonuclear leukocytes infiltrate mucosa Rubin R , Strayer DS Eds. Rubin’s Pathology: Clinicopathologic Foundations of Medicine, 6th Ed. Baltimore: Lippincott Williams & Wilkins, 2012.
- 159. Marc Imhotep Cray, MD Tubulointerstitial Diseases 159 Most forms of tubular injury also involve interstitium so they are discussed together= tubulointerstitial diseases Diseases involving tubules and interstitium may have clinical manifestations of nephritic syndrome, or of specific defects in tubular function, or of acute or chronic kidney disease without more specific defining features In this section we will discuss diseases characterized by: 1. Inflammatory involvement of tubules and interstitium (tubulointerstitial nephritis) Acute and chronic pyelonephritis (Upper UTI) 2. Ischemic and toxic tubular injury leading to acute tubular injury and clinical syndrome of acute kidney injury (ARF)
- 160. Marc Imhotep Cray, MD Tubulointerstitial Diseases cont’d. 160 Acute tubular necrosis (ATN)-now called acute tubular injury (ATI)- is usually due to profound hypotension causing ischemic damage to tubular epithelial cells Infections include pyelonephritis, renal abscesses & TB Drug toxicity usually causes tubulointerstitial nephritis due to a hypersensitivity reaction, but other patterns may occur, including direct toxicity to tubular epithelial cells giving an appearance similar to ATN Mechanical obstruction of ureters or bladder may lead to hydronephrosis and recurrent infection
- 161. Marc Imhotep Cray, MD Tubulointerstitial Nephritis 161 Tubulointerstitial nephritis (TIN) refers to a group of inflammatory kidney diseases that primarily involve interstitium & tubules glomeruli may be spared altogether or affected only late in course In cases of TIN caused by bacterial infection renal pelvis is prominently involved hence a more descriptive term is pyelonephritis (from pyelo, “pelvis”)
- 162. Marc Imhotep Cray, MD Tubulointerstitial Nephritis cont’d. 162 Term interstitial nephritis is reserved for cases of TIN that are nonbacterial in origin includes tubular injury resulting from drugs metabolic disorders such as hypokalemia irradiation viral infections, and immune reactions o For example, acute drug-induced interstitial nephritis caused by penicillin derivatives (eg, methicillin), NSAIDs, and diuretics On basis of clinical manifestations & character of inflammatory exudate TIN can be divided into acute and chronic categories
- 163. Marc Imhotep Cray, MD Trigger Case 11 163 A 25-year-old woman presents to the emergency room with fever, severe flank pain, and costovertebral angle tenderness. After taking a complete history, you find that she is sexually active and has had a 2-week history of burning pain while urinating and increased urinary frequency. Urinalysis reveals white cell casts in the urine and a urine sample is sent for culture. While you await the culture results, you start her on broad spectrum antibiotics. What is the Diagnosis?
- 164. Marc Imhotep Cray, MD Pyelonephritis (Acute and Chronic) 164 Etio. & Epidem. Acute: Caused by infection of renal parenchyma more frequent among women Chronic: Results from chronic urinary tract obstruction & recurrent UTIs Pathology Acute: Affects renal cortex w sparing of glomeruli neutrophilic infiltration and abscess formation within renal interstitium o abscesses may rupture introducing WBCs into tubular lumen Chronic: Asymmetric corticomedullary scarring tubules contain eosinophilic, proteinaceous casts resulting in gross appearance reminiscent of thyroid follicles (thyroidization of kidneys) in later stages results in tubular atrophy and interstitial fibrosis
- 165. Marc Imhotep Cray, MD Acute pyelonephritis, gross 165 Note in this plate that cortical surface is studded w focal pale abscesses, more numerous in upper pole and middle region of kidney, lower pole is relatively unaffected Betw. abscesses, there is dark congestion of renal surface Kumar V, Abbas AK, Aster JC. Robbins and Cotran Pathologic Basis of Disease, 9th ed. Philadelphia: Saunders-Elsevier, 2015.
- 166. Marc Imhotep Cray, MD Acute pyelonephritis, microscopic 166 An extensive infiltrate of neutrophils is present in collecting tubules and interstitial tissue Rubin R , Strayer DS Eds. Rubin’s Pathology: Clinicopathologic Foundations of Medicine, 6th Ed. Baltimore: Lippincott Williams & Wilkins, 2012.
- 167. Marc Imhotep Cray, MD Pyelonephritis cont’d. 167 Pathogenesis Principal causative organisms in acute pyelonephritis are enteric gram-negative bacilli Escherichia coli is most common Other important organisms are Proteus, Klebsiella, Enterobacter, and Pseudomonas these usually are assoc. w recurrent infections, especially in individuals who undergo urinary tract manipulations or have congenital or acquired anomalies of lower urinary tract Bacteria can reach kidneys from lower urinary tract (ascending infection) or through bloodstream (hematogenous infection) N.B. Ascending infection from lower urinary tract is most important & frequent route by which bacteria reach kidney
- 168. Marc Imhotep Cray, MD Pathways of renal infection 168 Hematogenous infection results from bacteremic spread ascending infection results from a combination of o urinary bladder infection, o vesicoureteral reflux, and o intrarenal reflux Kumar V, Abbas AK, Aster JC. Robbins and Cotran Pathologic Basis of Disease, 9th ed. Philadelphia: Saunders-Elsevier, 2015.
- 169. Marc Imhotep Cray, MD 169 Buja LM, Krueger GR. Netter’s Illustrated Human Pathology, 2nd Ed. Philadelphia: Saunders-Elsevier, 2014.
- 170. Marc Imhotep Cray, MD 170 Vesicoureteral reflux demonstrated by a voiding Dye injected into bladder refluxes into both dilated ureters, filling pelvis and calyces In absence of vesicoureteral reflux, infection remains localized in bladder o majority of individuals w repeated or persistent bacterial colonization of urinary tract suffer from cystitis and urethritis (lower UTI) rather than pyelonephritis Kumar V, Abbas AK, Aster JC. Robbins and Cotran Pathologic Basis of Disease, 9th ed. Philadelphia: Saunders-Elsevier, 2015.
- 171. Marc Imhotep Cray, MD Pyelonephritis cont’d. 171 Clinical Manifestations Acute: Fever; flank pain with CVA tenderness; polyuria and dysuria; nausea, vomiting, and diarrhea Chronic: Recurrent episodes of acute pyelonephritis can lead to renal hypertension and ESRD Lab findings: Leukocytosis, WBC and/or WBC casts in urine Treatment Acute: IV antibiotics Chronic: Renal transplantation if progresses to ESRD
- 172. 172 Chronic pyelonephritis, gross Rubin R , Strayer DS Eds. Rubin’s Pathology: Clinicopathologic Foundations of Medicine, 6th Ed. Baltimore: Lippincott Williams & Wilkins, 2012. A Cortical surface contains many irregular, depressed scars (reddish areas) B marked dilation of calyces (caliectasis) caused by inflammatory destruction of papillae, w atrophy and scarring of overlying cortex
- 173. Marc Imhotep Cray, MD Chronic pyelonephritis, microscopic 173 Rubin R , Strayer DS Eds. Rubin’s Pathology: Clinicopathologic Foundations of Medicine, 6th Ed. Baltimore: Lippincott Williams & Wilkins, 2012. A LM shows tubular dilation & atrophy, w many tubules containing eosinophilic hyaline casts resembling colloid of thyroid follicles (so-called thyroidization) interstitium is scarred and contains a chronic inflammatory cell infiltrate
- 174. Marc Imhotep Cray, MD Two major types of chronic pyelonephritis: Left. Vesicoureteral reflux causes infection of peripheral papillae and, therefore, scars in poles of kidney Right. Obstruction of urinary tract leads to high pressure backflow of urine, causes infection of all papillae, diffuse scarring of kidney & thinning of cortex 174 Rubin R , Strayer DS Eds. Rubin’s Pathology: Clinicopathologic Foundations of Medicine, 6th Ed. Baltimore: Lippincott Williams & Wilkins, 2012.
- 175. Marc Imhotep Cray, MD Reflux nephropathy [vesicoureteral reflux] 175 N.B. Most common mechanism in pathogenesis of chronic pyelonephritis is reflux nephropathy Reflux nephropathy is kidney damage (nephropathy) due to urine flowing backward (reflux) from bladder toward kidneys also called vesicoureteral reflux (VUR) Longstanding VUR can result in small and scarred kidneys during first five years of life in affected children End results of reflux nephropathy can include high blood pressure, excessive protein loss in urine, and eventually kidney failure When reflux nephropathy is suspected as a cause of kidney disease, other conditions to consider include chronic pyelonephritis, obstructive uropathy, and analgesic overuse
- 176. Marc Imhotep Cray, MD Renal papillary necrosis & Diffuse cortical necrosis 176 Important notes: Renal papillary necrosis is a complication of acute pyelonephritis in diabetics or analgesics particularly those ingested at least 2 kg in past (=analgesic nephropathy) risk is higher for phenacetin (withdrawn from market in U.S.) and acetaminophen compared to aspirin and other NSAIDs Diffuse cortical necrosis is an acute generalized infarction of renal cortices (medulla is spared)usually b/c of a combination of DIC and end-organ vasospasm in assoc. w obstetric catastrophes or septic shock
- 177. Marc Imhotep Cray, MD Papillary necrosis 177 Bisected kidney shows a dilated renal pelvis and dilated calyces secondary to urinary tract obstruction Papillae are all necrotic and appear as sharply demarcated, ragged, yellowish areas Rubin R , Strayer DS Eds. Rubin’s Pathology: Clinicopathologic Foundations of Medicine, 6th Ed. Baltimore: Lippincott Williams & Wilkins, 2012. Mnemonic for causes of renal papillary necrosis is POSTCARDS: pyelonephritis, obstruction of urogenital tract, sickle cell disease, tuberculosis, cirrhosis, analgesia/alcohol abuse, renal vein thrombosis, diabetes mellitus, and systemic vasculitis
- 178. Marc Imhotep Cray, MD Renal cortical necrosis 178 Cortex of kidney is pale yellow and soft due to diffuse cortical necrosis Rubin R , Strayer DS Eds. Rubin’s Pathology: Clinicopathologic Foundations of Medicine, 6th Ed. Baltimore: Lippincott Williams & Wilkins, 2012. Usually caused by significantly diminished arterial perfusion of kidneys due to spasms of feeding arteries, microvascular injury, or DIC Renal cortical necrosis is the pathological progression of acute tubular necrosis
- 179. Marc Imhotep Cray, MD Case Trigger 12 179 A 68-year-old man presents to the emergency room because he is unable to urinate. After taking a detailed history, you learn that he has had increasing urinary hesitancy and decreased force of his urine stream for several months. He also complains of a sensation of incomplete bladder emptying. On rectal examination, you find a smooth, firm, elastic enlargement of the prostate. Examination of the lower abdomen reveals signs of a distended bladder. You order laboratory studies that show increased urinary sodium excretion and an elevated BUN and creatinine. You determine that the patient requires prompt urethral catheterization to help reverse his renal failure. What is the Diagnosis?
- 180. Marc Imhotep Cray, MD Acute Renal Failure (Prerenal, Intrarenal, and Postrenal Azotemia) 180 Etiology Prerenal: Caused by decreased effective arterial volume (ie, CHF, hypovolemia, systemic vasodilation [sepsis]), or renal vasoconstriction (NSAIDs, ACE inhibitors, RAS) Intrarenal: Caused by acute tubular necrosis, acute interstitial nephritis, glomerulonephritis, and thrombotic microangiopathy Postrenal: Caused by kidney stones, BPH, neurogenic bladder, and neoplasia Pathology and Pathophysiology Prerenal: Renal hypoperfusion leads to decreased GFR resulting in sodium and water retention Intrarenal: Characterized by patchy tubular necrosis leads to tubule obstruction and fluid backflow across necrotic tubule and a resulting decrease in GFR Postrenal: Only develops w bilateral outflow obstruction
- 181. Marc Imhotep Cray, MD Acute Kidney Injury (AKI) [previously ARF] 181 An acute rise in serum Cr has been called ARF It is classified as: prerenal, intrarenal, postrenal azotemia Clinical Manifestations: oliguria; azotemia; hyperkalemia Intrarenal ARF is now called acute kidney injury (AKI) AKI is categorized by portion of kidney that is primarily injured: glomeruli (e.g., acute glomerulonephritis) vessels (e.g., vasculitis), tubules (e.g., ischemic acute tubular injury (previously ATN) or interstitium (acute interstitial nephritis) most common cause intrarenal AKI is ischemic acute tubular injury most common cause of AKI overall is therapeutic drugs
