XRD spectroscopy training sheet
•
2 gefällt mir•999 views
Some problems around the application of XRD powder
Melden
Teilen
Melden
Teilen
Downloaden Sie, um offline zu lesen
Empfohlen
Xrd presentation [autosaved] [autosaved] copy (2)![Xrd presentation [autosaved] [autosaved] copy (2)](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Xrd presentation [autosaved] [autosaved] copy (2)](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Xrd presentation [autosaved] [autosaved] copy (2)Sarhad University Science and Information Technology Hayatabad Peshawar Pakistan
Weitere ähnliche Inhalte
Was ist angesagt?
Xrd presentation [autosaved] [autosaved] copy (2)![Xrd presentation [autosaved] [autosaved] copy (2)](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Xrd presentation [autosaved] [autosaved] copy (2)](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Xrd presentation [autosaved] [autosaved] copy (2)Sarhad University Science and Information Technology Hayatabad Peshawar Pakistan
Was ist angesagt? (20)
Crystal Structures and X-Ray Diffraction - Sultan LeMarc

Crystal Structures and X-Ray Diffraction - Sultan LeMarc
Selected topics in x ray diffrcation ahmed abdelmohsen fame master

Selected topics in x ray diffrcation ahmed abdelmohsen fame master
Ähnlich wie XRD spectroscopy training sheet
Ähnlich wie XRD spectroscopy training sheet (20)
From the workshop "High-Resolution Submillimeter Spectroscopy of the Interste...

From the workshop "High-Resolution Submillimeter Spectroscopy of the Interste...
Microstructural and Dielectric Characterization of Sr doped Ba(Fe0.5Ta0.5)O3 ...

Microstructural and Dielectric Characterization of Sr doped Ba(Fe0.5Ta0.5)O3 ...
Mehr von Chris Sonntag
Mehr von Chris Sonntag (20)
Spectroscopic methods in inorganic chemistry 2019 IR

Spectroscopic methods in inorganic chemistry 2019 IR
Chemical Terminology: Lesson 1 elements and reactions

Chemical Terminology: Lesson 1 elements and reactions
Spectroscopic methods 2018 Part 1 uv vis TM complexes

Spectroscopic methods 2018 Part 1 uv vis TM complexes
Kürzlich hochgeladen
Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"

Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"National Information Standards Organization (NISO)
Kürzlich hochgeladen (20)
Disha NEET Physics Guide for classes 11 and 12.pdf

Disha NEET Physics Guide for classes 11 and 12.pdf
Web & Social Media Analytics Previous Year Question Paper.pdf

Web & Social Media Analytics Previous Year Question Paper.pdf
Call Girls in Dwarka Mor Delhi Contact Us 9654467111

Call Girls in Dwarka Mor Delhi Contact Us 9654467111
Measures of Dispersion and Variability: Range, QD, AD and SD

Measures of Dispersion and Variability: Range, QD, AD and SD
Ecosystem Interactions Class Discussion Presentation in Blue Green Lined Styl...

Ecosystem Interactions Class Discussion Presentation in Blue Green Lined Styl...
A Critique of the Proposed National Education Policy Reform

A Critique of the Proposed National Education Policy Reform
Interactive Powerpoint_How to Master effective communication

Interactive Powerpoint_How to Master effective communication
Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"

Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"
XRD spectroscopy training sheet
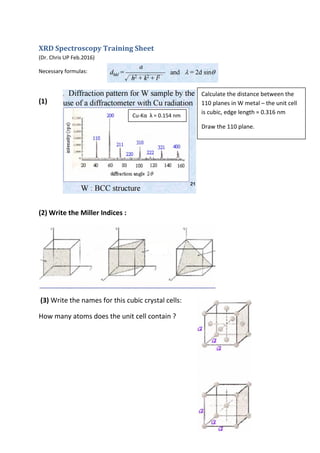
- 1. XRD Spectroscopy Training Sheet (Dr. Chris UP Feb.2016) Necessary formulas: (1) (2) Write the Miller Indices : (3) Write the names for this cubic crystal cells: How many atoms does the unit cell contain ? Calculate the distance between the 110 planes in W metal – the unit cell is cubic, edge length = 0.316 nm Draw the 110 plane. Cu-Kα λ = 0.154 nm
- 2. (4) BCC Fe placed in an x-ray diffractometer using x-ray with λ = 0.1541 nm. Diffraction from {110} planes was obtained at 2θ = 44.704 deg. Calculate lattice constant a. (5) An element that has either BCC or FCC structure shows diffraction peaks at following 2θ angles: 40, 58, 73, 86.8, 100.4 and 114.7. Wavelength of x-ray λ = 0.154 a. BCC or FCC? b. determine the lattice constant a. (6) A reaction between Zn(OAc)2 and NaOH gives a product with IR and XRD Compared to XRD of ZnO:
- 3. Infos IR spectrum of sodium hydroxide
- 4. Answers
- 5. 5 bcc or fcc lattice? For bcc, the first peak should come from 110, for fcc from 111. The second peak comes in both cases from 200. Which is consistent with the 2 formulas ? d(40) = 0.154/sin 20 = 0.45 nm Lattice constant a = 0.45 v = 0.636 nm for (110) or a= = 0.45 √(3) = 0.779 nm for (111) Next peak at 58: d(200) = 0.154/sin 29 = 0.318 nm lattice constant a = 0.318 √(4) = 0.635 nm Therefore the data fit with BCC structure !
