Pathology - Cancer
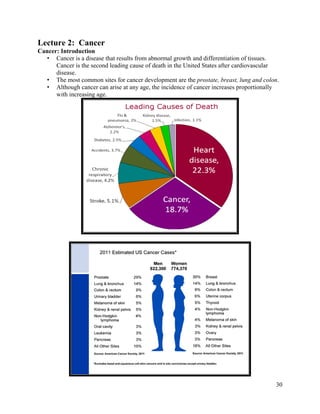
- 1. 30 Lecture 2: Cancer Cancer: Introduction • Cancer is a disease that results from abnormal growth and differentiation of tissues. Cancer is the second leading cause of death in the United States after cardiovascular disease. • The most common sites for cancer development are the prostate, breast, lung and colon. • Although cancer can arise at any age, the incidence of cancer increases proportionally with increasing age.
- 2. 31 PATHOPHYSIOLOGIC CHANGES Normal Cell: Cell Growth • Tissue growth and repair involve cell proliferation and differentiation. 1) Cell Proliferation ()ﺗﻜﺎﺛﺮ • Cell proliferation is the process by which cells divide and reproduce. The process of cell growth and division is called the cell cycle. • Cell division provides the body with the means for replacing cells that have a limited life span such as skin and blood cells, increasing tissue mass during periods of growth, and providing for tissue repair and wound healing. • In normal tissue, cell proliferation is regulated so that the number of cells actively dividing is equivalent to the number dying or being shed. • These controls are absent in cancer cells, and cell production exceeds cell loss. • Consequently, cancer cells enter the cell cycle more frequently and at different rates. Cell Cycle The mitotic phase (M) actually includes both mitosis and cytokinesis. This is when the nucleus and then the cytoplasm divide. The other three phases (G1, S, and G2) are generally grouped together as Interphase 1) Growth Phase 1 (G1): during this phase, the cell grows rapidly, while performing routine metabolic processes. It also makes proteins needed for DNA replication and copies some
- 3. 32 of its organelles in preparation for cell division. A cell typically spends most of its life in this phase. This phase is sometimes referred to as Gap 1. 2) Synthesis Phase (S): during this phase, the cell’s DNA is copied in the process of DNA replication. 3) Growth Phase 2 (G2): during this phase, the cell makes final preparations to divide. For example, it makes additional proteins and organelles. This phase is sometimes referred to as Gap 2. Control of the Cell Cycle Checkpoints in the cell cycle ensure that the cell is ready to proceed before it moves on to the next phase of the cycle. The G1 checkpoint, just before entry into S phase, makes the key decision of whether the cell should divide. The S checkpoint determines if the DNA has been replicated properly. The mitotic spindle checkpoint occurs at the point in metaphase where all the chromosomes should have aligned at the mitotic plate.
- 4. 33 Cell Cycle Summary The cell cycle is a repeating series of events that cells go through. It includes growth, DNA synthesis, and cell division. There are two growth phases, and cell division includes mitosis. The cell cycle is controlled by regulatory proteins at three key checkpoints in the cycle. The proteins signal the cell to either start or delay the next phase of the cycle. Cancer is a disease that occurs when the cell cycle is no longer regulated. Cancer cells grow rapidly and may form a mass of abnormal cells called a tumor. 2) Cell differentiation ()ﺍﻟﺘﻤﻴﻴﺰ Cell differentiation is the process whereby proliferating cells are transformed into different and more specialized cell types. It determines the structure, function, and life span of a cell. For example, a red blood cell is programmed to develop into a concave disk that functions as a vehicle for oxygen transport and lives approximately 120 days. There are three types of cells: o The well differentiated neurons and cells of skeletal and cardiac muscle that are unable to divide and reproduce; o The parent, or progenitor cells, that continue to divide and reproduce, such as blood cells, skin cells, and liver cells. o The undifferentiated stem cells that can be triggered to enter the cell cycle and produce large numbers of progenitor cells when the need arises. As a cell line becomes more differentiated, it becomes more highly specialized in its function and less able to divide.
- 5. 34 Cancer Cell Differentiation Cancer cells lose the ability to differentiate; that is, they enter a state, called anaplasia, in which they no longer appear or function like the original cell. Anaplasia occurs in varying degrees. The less the cells resemble the cell of origin, the more anaplastic they are said to be. As the anaplastic cells continue to reproduce, they lose the typical characteristics of the original cell. Some anaplastic cells begin functioning as another type of cell, possibly becoming a site for hormone production. For example, oat-cell lung cancer cells often produce antidiuretic hormone (ADH), which is produced by the hypothalamus but stored in and secreted by the posterior pituitary gland. When anaplasia occurs, cells of the same type in the same site exhibit many different shapes and sizes. Mitosis is abnormal and chromosome defects are common.
- 6. 35 Cancer terminology • Tumor or neoplasm Cancers result from a process of altered cell differentiation and growth. The resulting tissue is called neoplasia. The term neoplasm comes from a Greek word meaning new formation. • Benign neoplasm Tumor cells that tend to be clustered in a single mass and are not malignant. Benign tumors usually will not cause death unless they interfere with vital function. • Malignant neoplasm Tumors that have the ability to metastasize or break loose and spread to other areas of the body. If untreated, such tumors can cause great suffering and death.
- 7. 36 Metastasis Metastatic tumors are those in which the cancer cells have traveled from the original or primary site to a second or more distant site. Most commonly, metastasis occurs through the blood vessels and lymphatic system. Tumor cells also can be transported from one body location to another by external means, such as carriage on instruments or gloves during surgery
- 8. 37 Tumor cells enhance their potential for metastatic spread by releasing protease enzymes that digest the extracellular matrix surrounding adjacent cells. Malignant tumor cells may also produce growth factors that stimulate the formation of new blood vessels (angiogenesis), which in turn support the rapid growth of tumor cells. Metastatic sites Typically, the first capillary bed, whether lymphatic or vascular, encountered by the circulating tumor mass determines the location of the metastasis. For example, because the lungs receive all of the systemic venous return, they are a frequent site for metastasis. The liver is also a common site of metastasis for tumors originating in the gastrointestinal tract because blood draining the intestines must first pass through the liver via the hepatic portal system. In breast cancer, the axillary lymph nodes, which are in close proximity to the breast, are a common site of metastasis. Other types of cancer seem most likely to spread to specific organs. This organ tropism may be a result of growth factor or hormones secreted by the target organ or chemotactic factors that attract the tumor. Organ tropism is a term used to describe the affinity of a primary tumor to a specific distant site. For example : o More than 80% of Prostate Cancer patients develop bone metastases, predominantly in the spine, and are generally associated with a poor prognosis . o Another example, Colon Cancers are prone to metastasizing in the liver. As the colon tumor cells travel through the veins of portal circulation which leads to the liver.
- 9. 38 Classification of Neoplasia by Tissue Origin to Tissue Types • Neoplasms are composed of two types of tissue: • Parenchymal tissue and the Stroma or supporting tissue. The parenchymal cells represent the functional components of an organ (Nephron in the Kidney ) –see illustration- The supporting tissue consists of the connective tissue, blood vessels, and lymph structure. The parenchymal cells of a tumor determine its behavior and are the component for which a tumor is named. The supporting tissue carries the blood vessels and provides support for tumor survival and growth. • Tumors usually are named by adding the suffix -oma to the parenchymal tissue type from which the growth originated. • Thus, a benign tumor of glandular epithelial tissue is called an adenoma, • and a benign tumor of bone tissue is called an osteoma • The term carcinoma is used to designate a malignant tumor of epithelial tissue origin. In the case of a malignant adenoma, the term adenocarcinoma is used. • Malignant tumors of mesenchymal origin are called sarcomas (e.g., osteosarcoma). • Papillomas are benign microscopic or macroscopic fingerlike projections that grow on any surface.
- 10. 39 • A polyp is a growth that projects from a mucosal surface, such as the intestine. Although the term usually implies a benign neoplasm, some malignant tumors also appear as polyps. • Oncology is the study of tumors and their treatment.
- 11. 40 Theories of oncogenesis Oncogenesis is the process by which normal cells are transformed into cancer cells due to: (1) Abnormalities of proto-oncogenes, tumor suppressor genes and DNA repair genes. (2) Mutation of DNA (3) Hereditary (1) Abnormalities of proto-oncogenes, tumor suppressor genes and DNA repair genes. Cancer is caused by the accumulation of genetic and epigenetic mutations in genes that normally play a role in the regulation of cell proliferation, thus leading to uncontrolled cell growth. These genes can be grouped into 3 general categories: proto-oncogenes (growth promoting), tumor suppressor genes (growth inhibitory) and DNA repair genes, which help prevent mutations that lead to cancer. Chemicals (e.g., from smoking), radiation, viruses, and heredity all contribute to the development of cancer by triggering changes in a cell's genes. Chemicals and radiation act by damaging genes, viruses introduce their own genes into cells, and heredity passes on alterations in genes that make a person more susceptible to cancer. Genes are inherited instructions that reside within a person's chromosomes. Each gene instructs a cell how to build a particular kind of protein. Genes are altered, or "mutated," in various ways as part of the mechanism by which cancer arises.
- 12. 41 1) Oncogenes arise from the mutation of proto-oncogenes. Oncogenes are genes whose PRESENCE in certain forms and/or over activity can stimulate the development of cancer. When oncogenes arise in normal cells, they can contribute to the development of cancer by instructing cells to make abnormal versions or quantities of cellular growth-control proteins that stimulate excessive cell growth and division.
- 13. 42 2) A tumor suppressor gene, Or anti-oncogene, is a gene that protects a cell from one step on the path to cancer. When this gene is mutated to cause a loss or reduction in its function, the cell can progress to cancer, usually in combination with other genetic changes. One particular tumor suppressor gene codes for a protein called "p53" that can trigger cell suicide (apoptosis). In cells that have undergone DNA damage, the p53 protein acts like a brake pedal to halt cell growth and division. If the damage cannot be repaired, the p53 protein eventually initiates cell suicide, thereby preventing the genetically damaged cell from growing out of control.
- 14. 43 3) “DNA repair genes“ code for proteins whose normal function is to correct errors that arise when cells duplicate their DNA prior to cell division. Mutations in DNA repair genes can lead to a failure in repair, which in turn allows subsequent mutations to accumulate. (2) Mutation of DNA Numerous chemical, physical and biologic agents have been shown to be carcinogenic, meaning they can induce the formation of cancers. Many of these agents can damage cellular DNA, either directly or through the production of toxic intermediates such as free radicals. Certain viruses are also oncogenic in that they may induce mutations in host cell DNA or alter rates of cellular transcription. Mutations of cellular DNA can lead to the formation of cells with abnormal growth and differentiation patterns. (3) Hereditary A genetic predisposition has been observed for a number of cancers including colon cancer, breast cancer, retinoblastoma and certain forms of leukemia and lymphoma. A great deal of recent research has focused on identifying certain genetic markers in individuals that might pinpoint them as at risk for the development of certain types of cancer.
- 15. 44
- 16. 45 Carcinogenesis Steps Most of the numerous theories about carcinogenesis suggest that it involves four steps: initiation, promotion, progression and metastasis. 1) Initiation: Initiation refers to the damage to or mutation of DNA that occurs when the cell is exposed to an initiating substance or event (such as chemicals, virus, or radiation) during DNA replication (transcription). Normally, enzymes detect errors in transcription and remove or repair them. But sometimes an error is missed. If regulatory proteins recognize the error and block further division, then the error may be repaired or the cell may self-destruct. If these proteins miss the error again, it becomes a permanent mutation that is passed on to future generations of cells. 2) Promotion Promotion involves the exposure of the mutated cell to factors (promoters) that enhance its growth. This exposure may occur either shortly after initiation or years later. Promoters may be hormones, such as estrogen; food additives, such as nitrates; or drugs, such as nicotine. Promoters can affect the mutated cell by altering: function of genes that control cell growth and duplication , cell response to growth stimulators or inhibitors intercellular communication. 3) Progression refers to further growth and spreading throughout the tissue of origin. This step is irreversible. 4) Metastasis indicates spreading of the cancer to other organs
- 17. 46 Possible Cancer-Causing Agents Chemicals — Many such as benzene, vinyl chloride, cigarette smoke, aromatic hydrocarbons Radiation, radon gas, radioactive materials, ultraviolet radiation Occupational exposure — Asbestos, coal dust, uranium, solvents Oncogenic viruses A number of DNA and RNA viruses have been shown to be “oncogenic,” meaning they can cause cancers in the hosts they infect. • Human Papillomavirus — Cervical carcinoma • Hepatitis B Virus — Liver cancer • Epstein–Barr Virus — Burkitt’s lymphoma, nasopharyngeal cancer • HIV Virus — Kaposi’s sarcoma Dietary factors — High-fat diet, excessive alcohol intake, nitrosamine preservatives, grilled or charred foods Hormones — Estrogens, progesterone
- 18. 47 Manifestations of cancer Many cancers may be asymptomatic in the early stages. As the tumors continue to grow, they affect local tissues as well as the overall body. 1. Local effects of cancer Compression of blood vessels Ischemia Pain Bleeding Infection Altered tissue function 2. Systemic effects of cancer Fatigue Cachexia Bleeding and hemorrhage Anemia due to chronic bleeding or bone marrow destruction; this anemia may be exacerbated by chemotherapy Altered organ function Abnormal hormone production from an affected gland or directly from certain types of hormone-producing tumors. Cachexia A complex syndrome characterized by anorexia, weight loss and lean body (muscle) wasting seen in a significant percent of patients with cancer and AIDS. A number of metabolic abnormalities have been demonstrated in patients with cachexia that lead to poor utilization of nutrients and overall malnutrition. A key factor in cachexia appears to be the production of cytokines such as tumor necrosis factor and interleukins in response to the presence of cancer. These substances are produced by many cells within the body and appear to be protective against bacterial and viral infections as well as malignant cells. Unfortunately, these substances also appear to be responsible for many of the effects of cachexia including anorexia and lean body wasting.
- 19. 48 Tumor cell markers Some cancer cells release substances that normally aren't present in the body or are present only in small quantities. These substances, called tumor markers or biologic markers, are produced either by the cancer cell's genetic material during growth and development or by other cells in response to the presence of cancer. Markers may be found on the cell membrane of the tumor or in the blood, cerebrospinal fluid, or urine. Tumor cell markers include hormones, enzymes, genes, antigens, and antibodies. Tumor cell markers have many clinical uses, for example: screening people who are at high risk of cancer diagnosing a specific type of cancer in conjunction with clinical manifestations monitoring the effectiveness of therapy detecting recurrence. Tumor cell markers provide a method for detecting and monitoring the progression of certain types of cancer. Unfortunately, several disadvantages of tumor markers may preclude their use alone. For example, By the time the tumor cell marker level is elevated, the disease may be too far advanced to treat. Most tumor cell markers are not specific enough to identify one certain type of cancer. Some nonmalignant diseases, such as pancreatitis or ulcerative colitis, also are associated with tumor cell markers. Perhaps the worst drawback is that the absence of a tumor cell marker does not mean that a person is free of cancer. For example, mucinous ovarian cancer tumors typically do not express the ovarian cancer marker CA-125, so that a negative test doesn't eliminate the possibility of ovarian malignancy.
- 20. 49 Examples of Tumor Cell Markers Alpha-fetoprotein for liver and yolk sac (ovarian and testicular) cancers Carcinoembryonic antigen for colorectal cancer Human chorionic gonadotropin (hCG) for many tumors, including choriocarcinoma (usually cancer of the uterus) Acid phosphatase and prostate-specific antigen (PSA) for prostate cancer Monoclonal immunoglobulin (one subtype of antibody) for multiple melanoma CA-125, a protein released from female reproductive organs as well as from the lining of the chest and peritoneal cavities. It increases with inflamed or injured tissue and is a marker for ovarian cancer.
- 21. 50 Grading and Staging of Malignant Neoplasia Classification process that is helpful in determining prognosis and treatment. • Grading - a histologic method used by pathologists when they examine tissue or cell specimens • Looking for differentiation • Severity of malignancy can be assessed by degree of dysplasia or anaplasia present (remember anaplastic cells have de-differentiated or regressed from normal mature form) • Growth rate can be loosely determined by number of mitotic cells present- an increase in numbers means that cells are reproducing faster Grading: Recognized grades of malignancy: Grade I - cells are well differentiated (closely resemble tissue of origin), with little mitoses. Prognosis is good. Grade II - cells are moderately differentiated (some structural similarity to parent tissue), with moderate mitoses. Prognosis is fair. Grade III - Cells are poorly differentiated (little resemblance to their origin), with many mitoses. Prognosis is fair to poor. Grade IV - Cells are de-differentiated (bizarre and primitive with unrecognizable origins), with many mitoses. Prognosis is poor. Staging : A classification based on clinical findings by the physician (often oncologist) • stage relates to degree of spread (whereas grade relates to malignancy) • staging is based on size of primary tumor and amount of metastasis or secondary tumors Rules of Staging Follow TNM protocol: T refers to tumor size (1 to 4) N refers to numbers of lymph nodes affected (local invasion) M means the extent of metastasis Staging usually has a better correlation with prognosis. Grading and staging (Summary) Grade: GX: Grade cannot be assessed (Undetermined grade) G1 Well-differentiated (Low grade) G2 Moderately differentiated (Intermediate grade) G3 Poorly differentiated (High grade) G4 Undifferentiated (High grade) Staging systems (various): carcinoma • Stage 1: confined to organ • Stage 2: locally invasive • Stage 3: lymph node invasion • Stage 4: spread to distant sites
- 23. 52 Diagnosis Visualization Radiography, computer tomography (CT scans), magnetic resonance imaging (MRI) Endoscopy : may also be utilized to visually detect tumors in the bronchi and gastrointestinal tract . Identifies the presence of a tumor or tumors; can also be used to evaluate metastasis Biopsy Removal of a piece of suspect tissue for detailed histologic or histochemical analysis May be accomplished surgically, by a needle biopsy, by scraping cells from a surface (Pap smear) or by endoscopic biopsy Rationale for therapy Cancer treatment can be multifaceted and may include surgical removal of tumors, as well as chemotherapy and/or radiation therapy to kill or arrest rapidly growing tumor cells. A number of immune-based treatments are currently under investigation as alternatives to toxic chemotherapy and radiation therapy. Treatment with specific hormones has also been shown to inhibit the growth of certain types of cancers. Treatment of cancer 1) Surgical removal • If accessible, tumors should be surgically removed. Often accompanied by • chemotherapy or radiation therapy to kill any cancer cells that are not • removed or have metastasized. 2) Chemotherapy • Drugs used for chemotherapy of cancer fall into several categories : Alkylating agents and nitrosureas (examples: cyclophosphamide, carmustine) Cytotoxic to cancer cells due to alkylation of cancer cell DNA Major toxicities include nausea and vomiting, and bone marrow suppression Antimetabolites (examples: methotrexate, fluorouracil) Inhibit synthesis of essential nucleotides and nucleic acids in cancer cells Major toxicities include myelosuppression, nausea, vomiting, oral and gastrointestinal ulceration Plant alkaloids (examples: vinblastine, vincristine) Disrupt mitosis in cancer cells by interfering with formation of the mitotic spindle Numerous toxicities including cardiotoxicity, bone marrow depression, neurologic and muscle effects as well as alopecia
- 24. 53 Antibiotics (examples: doxorubicin, bleomycin) Bind directly to cancer cell DNA to block the formation of new RNA or DNA Major toxicities include bone marrow suppression, alopecia 3) Hormonal therapy Sex hormones are routinely used to inhibit tumor growth in breast, prostate and uterine cancer. The estrogen inhibitor tamoxifen has also been shown to be effective in the treatment of breast cancer and may eventually be used as a prophylactic agent in women who are at a high risk for developing breast cancer. The androgen inhibitor flutamide has also been approved for treatment of prostate cancer. 4) Radiation therapy Radiation therapy utilizes ionizing or particle beam radiation to destroy cancer cells that are highly mitotic and most susceptible to the lethal effects of radiation. Radiation therapy can have a number of localized and systemic side effects including alopecia, diarrhea, tissue irritation and organ inflammation. 5) Immune-based therapies “Biologic response modifiers” such as interferons, immunomodulators, tumor antigens and lymphokines/cytokines are being investigated as means of enhancing the immune system response of individuals with cancer. Monoclonal antibodies have also been studied as a highly specific means of delivering chemotherapeutic drugs directly to and only to cancer cells. --------------------------------
