alkynes (2).ppt
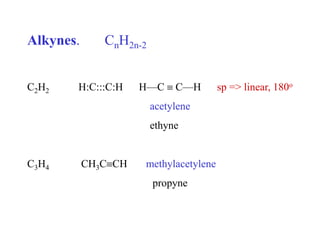
- 1. Alkynes. CnH2n-2 C2H2 H:C:::C:H H—C C—H sp => linear, 180o acetylene ethyne C3H4 CH3CCH methylacetylene propyne
- 2. nomenclature: common names: “alkylacetylene” IUPAC: parent chain = longest continuous carbon chain that contains the triple bond. alkane drop –ane add -yne prefix locant for the triple bond, etc. CH3CH2CCCH3 2-pentyne ethylmethylacetylene
- 3. “terminal” alkynes have the triple bond at the end of the chain: CH3 CH3CH2CCH HCCCHCH2CH3 1-butyne 3-methyl-1-pentyne ethylacetylene sec-butylacetylene
- 4. physical properties: weakly or non-polar, no H-bonding relatively low mp/bp water insoluble
- 5. Synthesis, alkynes: 1. dehydrohalogenation of vicinal dihalides H H H | | | — C — C — + KOH — C = C — + KX + H2O | | | X X X H | — C = C — + NaNH2 — C C — + NaX + NH3 | X
- 6. H H | | — C — C — + 2 KOH — C C — + KX + H2O | | heat X X CH3CH2CHCH2 + KOH; then NaNH2 CH3CH2CCH Br Br “ + 2 KOH, heat
- 7. alkene vicinal dihalide alkyne X2 1. KOH 2. NaNH2 CH3CH=CH2 CH3CHCH2 CH3CCH Br Br Br2 1. KOH 2. NaNH2
- 8. Synthesis of propyne from propane CH3CH2CH3 Br2, heat CH3CH2CH2-Br + CH3CHCH3 Br KOH(alc) CH3CH=CH2 Br2 CH3CHCH2 Br Br KOH CH3CH CH Br NaNH2 CH3C CH
- 9. 2. coupling of metal acetylides with 1o/CH3 alkyl halides R-CC-Na+ + R´X R-CC-R´ + NaX a) SN2 b) R´X must be 1o or CH3X CH3CC-Li+ + CH3CH2-Br CH3CCCH2CH3
- 10. CH3C C Na + CH3CCH3 CH3 Br CH3C CH + CH3C CH2 CH3 3o alkyl halide E2 elimination! note: R-X must be 1o or CH3 to get SN2!
- 12. Reactions, alkynes: 1. addition of H2 (reduction) 2. addition of X2 3. addition of HX 4. addition of H2O, H+ 5. as acids 6. Ag+ 7. oxidation
- 13. 1. Addition of H2 H H | | — C C — + 2 H2, Ni — C — C — | | H H alkane requires catalyst (Ni, Pt or Pd)
- 14. HCCH + 2 H2, Pt CH3CH3 [ HCCH + one mole H2, Pt CH3CH3 + CH2=CH2 + HCCH ] H / Na or Li C = C anti- NH3(liq) / H — C C — / H2, Pd-C C = C syn- Lindlar catalyst / H H
- 15. CH3 H / Na or Li C = C anti- NH3(liq) / H CH3 trans-2-butene CH3CCCH3 H H / H2, Pd-C C = C syn- Lindlar catalyst / CH3 CH3 cis-2-butene
- 16. 2. Addition of X2 X X X | | | — C C— + X2 — C = C — + X2 — C — C — | | | X X X Br Br Br CH3CCH + Br2 CH3C=CH + Br2 CH3-C-CH Br Br Br
- 17. 3. Addition of hydrogen halides: H H X | | | — C C— + HX — C = C — + HX — C — C — | | | X H X a) HX = HI, HBr, HCl b) Markovnikov orientation Cl CH3CCH + HCl CH3C=CH2 + HCl CH3CCH3 Cl Cl
- 18. 4. Addition of water. Hydration. O — C C — + H2O, H+, HgO — CH2 — C— H OH — C = C — “enol” keto-enol tautomerism Markovnikov orientation.
- 19. CH3CH2CCH + H2O, H2SO4, HgO 1-butyne O CH3CH2CCH3 2-butanone
- 20. 5. As acids. terminal alkynes only! a) with active metals CH3CCH + Na CH3CC-Na+ + ½ H2 b) with bases CH3CCH + CH3MgBr CH4 + CH3C CMgBr SA SB WA WB
- 21. acid strength: CH4 < NH3 < HCCH < ROH < H2O < HF HC CH + NaOH NR ( H2O = stronger acid! ) CH3CH2CCH + LiNH2 NH3 + CH3CH2CC-Li+ SA WA
- 22. 6. Ag+ terminal alkynes only! CH3CH2CCH + AgNO3 CH3CH2CC-Ag+ CH3CCCH3 + AgNO3 NR (not terminal) formation of a precipitate is a test for terminal alkynes.
- 23. 7. Oxidation KMnO4 R-CC-R´ hot KMnO4 RCOOH + HOOCR´ carboxylic acids O3; then Zn, H2O
- 24. CH3CH2CCCH3 + KMnO4 CH3CCH + hot KMnO4 CH3CCCH3 + O3; then Zn, H2O CH3CH2COOH + HOOCCH3 CH3COOH + CO2 2 CH3COOH
- 25. Alkynes Nomenclature Syntheses 1. dehydrohalogenation of vicinal dihalide 2. coupling of metal acetylides with 1o/CH3X
- 26. Reactions, alkynes: 1. addition of H2 (reduction) 2. addition of X2 3. addition of HX 4. addition of H2O, H+ 5. as acids 6. Ag+ 7. oxidation