Tivicay®
•Als PPTX, PDF herunterladen•
3 gefällt mir•2,827 views
Dolutegravir (Tivicay) is an integrase inhibitor approved to treat HIV infection. It is generally dosed as a single 50mg daily tablet for adults and adolescents over 12 years old and weighing at least 40kg. Lower doses are needed when taken with certain other drugs. It is metabolized in the liver and has a half-life of 14 hours. Common side effects include abnormal liver function, fat redistribution, hyperglycemia, insomnia, and headache. Dolutegravir should not be taken with certain antacids, supplements, or seizure and tuberculosis medications due to potential drug interactions.
Melden
Teilen
Melden
Teilen
Empfohlen
Empfohlen
Weitere ähnliche Inhalte
Was ist angesagt?
Was ist angesagt? (20)
Andere mochten auch
Andere mochten auch (20)
¿Hay futuro para el binomio artesanía y joya de autor?

¿Hay futuro para el binomio artesanía y joya de autor?
O IMÓVEL MÓVEL - PROTAN ELMARK - Galpões de Alumínio - Tendas

O IMÓVEL MÓVEL - PROTAN ELMARK - Galpões de Alumínio - Tendas
Ciberseguridad en la identidad digital y la reputación online para tu empresa

Ciberseguridad en la identidad digital y la reputación online para tu empresa
Test del algodón. Saber si tu idea es un proyecto viable.

Test del algodón. Saber si tu idea es un proyecto viable.
Măsuri de protecție împotriva undelor electromagnetice

Măsuri de protecție împotriva undelor electromagnetice
Ähnlich wie Tivicay®
Ähnlich wie Tivicay® (20)
Tigecycline 50 mg powder for solution for infusion smpc taj pharmaceuticals

Tigecycline 50 mg powder for solution for infusion smpc taj pharmaceuticals
Lamivudine 100 mg film coated tablets smpc- taj pharmaceuticals

Lamivudine 100 mg film coated tablets smpc- taj pharmaceuticals
HIV presentation | hiv and various respiratory infections |

HIV presentation | hiv and various respiratory infections |
Diclofenac sodium 50mg gastro resistant tablets smpc- taj pharmaceuticals

Diclofenac sodium 50mg gastro resistant tablets smpc- taj pharmaceuticals
Mehr von velspharmd
Mehr von velspharmd (20)
Anti tuberculosis drug - induced hepatitis – A Case Study

Anti tuberculosis drug - induced hepatitis – A Case Study
Kürzlich hochgeladen
Book Paid Powai Call Girls Mumbai 𖠋 9930245274 𖠋Low Budget Full Independent High Profile Call Girl 24×7
Booking Contact Details
WhatsApp Chat: +91-9930245274
Mumbai Escort Service includes providing maximum physical satisfaction to their clients as well as engaging conversation that keeps your time enjoyable and entertaining. Plus they look fabulously elegant; making an impressionable.
Independent Escorts Mumbai understands the value of confidentiality and discretion - they will go the extra mile to meet your needs. Simply contact them via text messaging or through their online profiles; they'd be more than delighted to accommodate any request or arrange a romantic date or fun-filled night together.
We provide -
Flexibility
Choices and options
Lists of many beauty fantasies
Turn your dream into reality
Perfect companionship
Cheap and convenient
In-call and Out-call services
And many more.
29-04-24 (Smt)Book Paid Powai Call Girls Mumbai 𖠋 9930245274 𖠋Low Budget Full Independent H...

Book Paid Powai Call Girls Mumbai 𖠋 9930245274 𖠋Low Budget Full Independent H...Call Girls in Nagpur High Profile
Kürzlich hochgeladen (20)
VIP Service Call Girls Sindhi Colony 📳 7877925207 For 18+ VIP Call Girl At Th...

VIP Service Call Girls Sindhi Colony 📳 7877925207 For 18+ VIP Call Girl At Th...
Call Girls Haridwar Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Haridwar Just Call 8250077686 Top Class Call Girl Service Available
Book Paid Powai Call Girls Mumbai 𖠋 9930245274 𖠋Low Budget Full Independent H...

Book Paid Powai Call Girls Mumbai 𖠋 9930245274 𖠋Low Budget Full Independent H...
Call Girls Gwalior Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Gwalior Just Call 9907093804 Top Class Call Girl Service Available
All Time Service Available Call Girls Marine Drive 📳 9820252231 For 18+ VIP C...

All Time Service Available Call Girls Marine Drive 📳 9820252231 For 18+ VIP C...
The Most Attractive Hyderabad Call Girls Kothapet 𖠋 6297143586 𖠋 Will You Mis...

The Most Attractive Hyderabad Call Girls Kothapet 𖠋 6297143586 𖠋 Will You Mis...
Call Girls Jabalpur Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Jabalpur Just Call 8250077686 Top Class Call Girl Service Available
Top Rated Bangalore Call Girls Mg Road ⟟ 9332606886 ⟟ Call Me For Genuine S...

Top Rated Bangalore Call Girls Mg Road ⟟ 9332606886 ⟟ Call Me For Genuine S...
(Rocky) Jaipur Call Girl - 09521753030 Escorts Service 50% Off with Cash ON D...

(Rocky) Jaipur Call Girl - 09521753030 Escorts Service 50% Off with Cash ON D...
Call Girls Bhubaneswar Just Call 9907093804 Top Class Call Girl Service Avail...

Call Girls Bhubaneswar Just Call 9907093804 Top Class Call Girl Service Avail...
Call Girls Ooty Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Ooty Just Call 8250077686 Top Class Call Girl Service Available
VIP Hyderabad Call Girls Bahadurpally 7877925207 ₹5000 To 25K With AC Room 💚😋

VIP Hyderabad Call Girls Bahadurpally 7877925207 ₹5000 To 25K With AC Room 💚😋
Best Rate (Guwahati ) Call Girls Guwahati ⟟ 8617370543 ⟟ High Class Call Girl...

Best Rate (Guwahati ) Call Girls Guwahati ⟟ 8617370543 ⟟ High Class Call Girl...
Top Rated Bangalore Call Girls Richmond Circle ⟟ 9332606886 ⟟ Call Me For Ge...

Top Rated Bangalore Call Girls Richmond Circle ⟟ 9332606886 ⟟ Call Me For Ge...
(Low Rate RASHMI ) Rate Of Call Girls Jaipur ❣ 8445551418 ❣ Elite Models & Ce...

(Low Rate RASHMI ) Rate Of Call Girls Jaipur ❣ 8445551418 ❣ Elite Models & Ce...
Top Rated Bangalore Call Girls Ramamurthy Nagar ⟟ 9332606886 ⟟ Call Me For G...

Top Rated Bangalore Call Girls Ramamurthy Nagar ⟟ 9332606886 ⟟ Call Me For G...
Night 7k to 12k Navi Mumbai Call Girl Photo 👉 BOOK NOW 9833363713 👈 ♀️ night ...

Night 7k to 12k Navi Mumbai Call Girl Photo 👉 BOOK NOW 9833363713 👈 ♀️ night ...
Premium Bangalore Call Girls Jigani Dail 6378878445 Escort Service For Hot Ma...

Premium Bangalore Call Girls Jigani Dail 6378878445 Escort Service For Hot Ma...
(👑VVIP ISHAAN ) Russian Call Girls Service Navi Mumbai🖕9920874524🖕Independent...

(👑VVIP ISHAAN ) Russian Call Girls Service Navi Mumbai🖕9920874524🖕Independent...
Call Girls Cuttack Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Cuttack Just Call 9907093804 Top Class Call Girl Service Available
Tivicay®
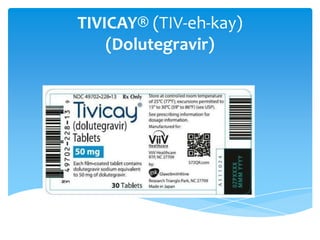
- 2. Date of Approval: August 12, 2013 Approved by :U.S. Food and Drug Administration (FDA) Company: ViiV Healthcare Generic Name : Dolutegravir Brand Name: Tivicay Other Names: DTG Drug Class: Integrase Inhibitors Approved Use: Treatment of HIV Infection Chemical structure: Molecular Weight: 419.3821
- 3. DOSAGE AND ADMINISTRATION: TIVICAY tablets may be taken with or without food. For Adults : 50 mg tablet once a d for people on HIV therapy for the first time . Twice-daily dosing in people who have viral resistance to Isentress and elvitegravir. Expected to have no food restriction Alternative combinations that do not include metabolic inducers should be considered where possible
- 4. Pediatric Patients : Age 12 years and older and weighing at least 40 kg – 50 mg p/o -OD. Efavirenz, fosamprenavir/ritonavir, tipranavir/ritonavi r, or rifampin - 50 mg twice daily. Safety and efficacy of TIVICAY have not been established in pediatric patients younger than 12 years or weighing less than 40 kg, or in pediatric patients who are INSTI-experienced with documented or clinically suspected resistance to other INSTIs (raltegravir, elvitegravir).
- 5. DOSAGE FORMS AND STRENGTHS TIVICAY 50-mg tablets are yellow, round, filmcoated, biconvex tablets . Each tablet contains 50 mg of dolutegravir (as dolutegravir sodium) Pharmacokinetics Absorption Peak plasma time: 2-3 hr Peak plasma concentration: 3.67-4.15 mcg/l Distribution Protein bound: ≥98.9% Vd : 17.4 L; 4-232 ng/mL (CSF)
- 6. Metabolism : Metabolized by UGT1A1 with some contribution from CYP3A Elimination : Half-life: 14 hr Clearance: 1 L/hr Excretion: 53% feces (unchanged); 31% urine (as ether glucuronide, benzylic carbon, or N-dealkylation product); <1% urine (unchanged)
- 7. Mechanism of Action Inhibits catalytic activity of HIV-1 integrase, an HIV encoded enzyme required for viral replication
- 8. Adverse effects Abnormal liver function in participants co-infected with hepatitis B and / or C Fat Redistribution 1-10% •Hyperglycemia (<1-7%) •Insomnia (<1-3%) •Headache (<1-2%) •GI disorders (<2%) •Fatigue (<2%) •Hepatitis (<2%)
- 9. • Myositis (<2%) • Renal impairment (<2%) Pruritus (<2%) Nausea (<1-1%) <1% Abnormal dreams Dizziness Diarrhea Rash Vertigo
- 10. CONTRAINDICATIONS Coadministration of TIVICAY with dofetilide is contraindicated due to the potential for increased dofetilide plasma concentrations and the risk for serious and/or life-threatening events Pregnancy: Pregnancy category B. Breastfeeding: Breastfeeding is NOT recommended
- 11. Drug Interactions Co-administration of TIVICAY with drugs that are strong inducers of UGT1A1 and/or CYP3A4 may result in reduced plasma concentrations of dolutegravir and require dose adjustments of TIVICAY. -TIVICAY should be taken 2 hours before or 6 hours after taking cation-containing antacids or laxatives, sucralfate, oral iron supplements, oral calcium supplements, or buffered medications.
- 12. Anti-seizure medicines: oxcarbazepine • phenytoin • phenobarbital • carbamazepine a medicine that contains metformin rifampin
- 13. Overdose There is no known specific treatment for overdose with TIVICAY. If overdose occurs, the patient should be monitored and standard supportive treatment applied as required. As dolutegravir is highly bound to plasma proteins, it is unlikely that it will be significantly removed by dialysis.
- 14. Patient Information Hypersensitivity Reactions Effects on Serum Liver Biochemistries in Patients With Hepatitis B or C Co-infection Patients should be advised to avoid doing things that can spread HIV-1 infection to others. • Do not share personal items that can have blood or body fluids on them, like toothbrushes and razor blades. • Practice safe sex by using a latex or polyurethane condom to lower the chance of sexual contact with semen, vaginal secretions, or blood.
- 15. • Female patients should be advised not to breastfeed To inform their physician or pharmacist if they develop any unusual symptom, or if any known symptom persists or worsens. If missed a dose - should take it as soon as they remember. If they do not remember until it is within 4 hours of the time for the next dose, they should be instructed to skip the missed dose and go back to the regular schedule.