Melden
Teilen
Downloaden Sie, um offline zu lesen
Empfohlen
Empfohlen
MS medium supplemented with different
concentrations of auxins 2,4-dichloro-phenoxyacetic acid (2,4-D),
α-naphthalene acetic acid (NAA) and combination with kinetin
were studied to obtain a suitable protocol of callus initiation of
Trigonella foenum graecum. Callus was induced from hypocotyls
and cotyledons explants which were collected from the seedlings
of the mentioned plant. The explants were cultured in MS
medium supplemented with two auxins 2,4-D and NAA
separately with different concentrations (0.0 as control, 0.1, 0.5,
1.0, 2.0, 3.0, 4.0 and 5.0 mg/l). Concentration of kinetin (0.5 mg/l)
was used in combination with all concentrations of 2, 4-D and
NAA hormones. The callus was successfully induced in all
different concentrations of two mentioned auxins and
combinations of different concentrations of two auxins separately
with (0.5 mg/l) of kinetin. No callus formation was observed in
the absence of plant growth regulators. Hypocotyl explants of
T.foenum- graecum were much better in inducing callus than
cotyledons explants. Combinations of NAA+Kin and 2,4-D+Kin
were found to be more effective for inducing callus from
hypocotyls explants compared to 2,4-D and NAA alone, at the
same time callus induced by 2,4-D using cotyledons explants
gave the best results compared to the other hormone. Among the
different concentrations of auxin and combinations with kinetin
,the highest mean of callusing index from hypocotyls segments
was (3.50±0.15) with 100% 0f callusing in sixth week by 4.0 mg/l
NAA+ 0.5mg/l Kin. In the case of callus induction from
cotyledons segments the highest mean of callusing index
(2.41±0.18) with 100% of callusing in sixth week was observed by
1.0 mg/l 2, 4-DEFFECT OF DIFFERENT CONCENTRATIONS OF AUXINS AND COMBINATION WITH KINETIN ON ...

EFFECT OF DIFFERENT CONCENTRATIONS OF AUXINS AND COMBINATION WITH KINETIN ON ...International Journal of Technical Research & Application
Nanobiosensors exploiting specific interactions between an enzyme and herbici...

Nanobiosensors exploiting specific interactions between an enzyme and herbici...Grupo de Pesquisa em Nanoneurobiofisica
Weitere ähnliche Inhalte
Was ist angesagt?
MS medium supplemented with different
concentrations of auxins 2,4-dichloro-phenoxyacetic acid (2,4-D),
α-naphthalene acetic acid (NAA) and combination with kinetin
were studied to obtain a suitable protocol of callus initiation of
Trigonella foenum graecum. Callus was induced from hypocotyls
and cotyledons explants which were collected from the seedlings
of the mentioned plant. The explants were cultured in MS
medium supplemented with two auxins 2,4-D and NAA
separately with different concentrations (0.0 as control, 0.1, 0.5,
1.0, 2.0, 3.0, 4.0 and 5.0 mg/l). Concentration of kinetin (0.5 mg/l)
was used in combination with all concentrations of 2, 4-D and
NAA hormones. The callus was successfully induced in all
different concentrations of two mentioned auxins and
combinations of different concentrations of two auxins separately
with (0.5 mg/l) of kinetin. No callus formation was observed in
the absence of plant growth regulators. Hypocotyl explants of
T.foenum- graecum were much better in inducing callus than
cotyledons explants. Combinations of NAA+Kin and 2,4-D+Kin
were found to be more effective for inducing callus from
hypocotyls explants compared to 2,4-D and NAA alone, at the
same time callus induced by 2,4-D using cotyledons explants
gave the best results compared to the other hormone. Among the
different concentrations of auxin and combinations with kinetin
,the highest mean of callusing index from hypocotyls segments
was (3.50±0.15) with 100% 0f callusing in sixth week by 4.0 mg/l
NAA+ 0.5mg/l Kin. In the case of callus induction from
cotyledons segments the highest mean of callusing index
(2.41±0.18) with 100% of callusing in sixth week was observed by
1.0 mg/l 2, 4-DEFFECT OF DIFFERENT CONCENTRATIONS OF AUXINS AND COMBINATION WITH KINETIN ON ...

EFFECT OF DIFFERENT CONCENTRATIONS OF AUXINS AND COMBINATION WITH KINETIN ON ...International Journal of Technical Research & Application
Nanobiosensors exploiting specific interactions between an enzyme and herbici...

Nanobiosensors exploiting specific interactions between an enzyme and herbici...Grupo de Pesquisa em Nanoneurobiofisica
Was ist angesagt? (20)
EFFECT OF DIFFERENT CONCENTRATIONS OF AUXINS AND COMBINATION WITH KINETIN ON ...

EFFECT OF DIFFERENT CONCENTRATIONS OF AUXINS AND COMBINATION WITH KINETIN ON ...
International Journal of Pharmaceutical Science Invention (IJPSI)

International Journal of Pharmaceutical Science Invention (IJPSI)
Nanoparticle of plant extract: A Novel approach for cancer therap

Nanoparticle of plant extract: A Novel approach for cancer therap
Evaluation mutagenic potential of pesticides through bioassays with Allium cepa

Evaluation mutagenic potential of pesticides through bioassays with Allium cepa
Ecological assessment of topically applied dsRNA-based products - Jörg Romeis...

Ecological assessment of topically applied dsRNA-based products - Jörg Romeis...
Micromotors to capture and destroy anthrax simulant spores

Micromotors to capture and destroy anthrax simulant spores
Assimilation Of Phosphite By A. Stolonifera L. And Its In Vitro Effect On Mic...

Assimilation Of Phosphite By A. Stolonifera L. And Its In Vitro Effect On Mic...
Nanobiosensors exploiting specific interactions between an enzyme and herbici...

Nanobiosensors exploiting specific interactions between an enzyme and herbici...
Paola Caballero and Anthony Hernandez Scientific Manuscript

Paola Caballero and Anthony Hernandez Scientific Manuscript
Green Synthesis of Silver Nanoparticles exhibits Anti-Metastasis and Anti-Bio...

Green Synthesis of Silver Nanoparticles exhibits Anti-Metastasis and Anti-Bio...
Combined effect of solar drying and gamma radiation on the microbiological qu...

Combined effect of solar drying and gamma radiation on the microbiological qu...
Toxicity Studies on Methanolic Leaf Extract of Rothmannia Longiflora: Biochem...

Toxicity Studies on Methanolic Leaf Extract of Rothmannia Longiflora: Biochem...
Programmatic detection of spatial behaviour in an agent-based model

Programmatic detection of spatial behaviour in an agent-based model
Phylotype Analysis of Ralstonia Solanacearum Causing Bacterial wilt in Eggpla...

Phylotype Analysis of Ralstonia Solanacearum Causing Bacterial wilt in Eggpla...
Andere mochten auch
Andere mochten auch (7)
S2.5 Development and evaluation of a maize variety with resistance against

S2.5 Development and evaluation of a maize variety with resistance against
Integrated vector control approach Dr Kulrajat Bhasin.

Integrated vector control approach Dr Kulrajat Bhasin.
Ähnlich wie Poster_05022016
The Senegalese grasshopper Oedaleus senegalensis (Krauss, 1877) is a serious agricultural pest in Senegal. The use of chemical pesticides on a large scale has raised concerns because of side effects on health and the environment. As an alternative to chemical control, a fungal strain of Metarhizium anisopliae (Metschnikoff, Sorokin) was isolated from the Senegalese grasshopper, and grown in agar culture medium. The effect on O. senegalensis was studied with an oil fungus formulation of 340 × 105 conidia/ml. Spraying took place in the field, and both nymphs and adults were infected with the fungus oil formulation. A total of 1.5 liter oil formulation of fungus was used for 1500 m2. Two methods were used to assess effectiveness: 1) we captured infected insects and fed them fresh grass daily in the laboratory and recorded time to death; 2) we counted insects in the field before and after application. In the field, the number of insects decreased significantly after the fungus treatment. In the laboratory, the lethal time at which 50% of the insects died varied between 8 to 9 days. The effectiveness of M. anisopliae in natural environment decreased with time.Efficacy of Metarhizium Anisoplae (Metschnikoff, Sorokin) on the senegalese g...

Efficacy of Metarhizium Anisoplae (Metschnikoff, Sorokin) on the senegalese g...Journal of Agriculture and Crops
Ähnlich wie Poster_05022016 (20)
The Effect of Dried Leaves Extract of Hyptis suaveolens on Various Stages of ...

The Effect of Dried Leaves Extract of Hyptis suaveolens on Various Stages of ...
Efficacy of Metarhizium Anisoplae (Metschnikoff, Sorokin) on the senegalese g...

Efficacy of Metarhizium Anisoplae (Metschnikoff, Sorokin) on the senegalese g...
Preliminary evaluation of the larvicidal efficacy of coelomic fluid of Eudril...

Preliminary evaluation of the larvicidal efficacy of coelomic fluid of Eudril...
Identification of phytonematodes from vegetable growing areas in malakand div...

Identification of phytonematodes from vegetable growing areas in malakand div...
Atmospheric Exposure to Cr III Powder Causes Genotoxicity in Rattus Norvegicus.

Atmospheric Exposure to Cr III Powder Causes Genotoxicity in Rattus Norvegicus.
IOSR Journal of Pharmacy (IOSRPHR), www.iosrphr.org, call for paper, research...

IOSR Journal of Pharmacy (IOSRPHR), www.iosrphr.org, call for paper, research...
48.Effect of R394 on Ecdysteroid titre of haemolymph after fourth moult of Si...

48.Effect of R394 on Ecdysteroid titre of haemolymph after fourth moult of Si...
Genotyping and subgenotyping of Trichophyton rubrum isolated from dermatophyt...

Genotyping and subgenotyping of Trichophyton rubrum isolated from dermatophyt...
Secondary Metabolites of the Entomopathogenic Fungus, Cladosporium cladospori...

Secondary Metabolites of the Entomopathogenic Fungus, Cladosporium cladospori...
Assessing the Risk of Resistance in Pseudoperonospora cubensis to the Fungici...

Assessing the Risk of Resistance in Pseudoperonospora cubensis to the Fungici...
Entomopathogenic nematodes against ostrinia furnacalis (asian corn borer)

Entomopathogenic nematodes against ostrinia furnacalis (asian corn borer)
Paulo Guilherme Coconut Constraints Oryctes monocerus

Paulo Guilherme Coconut Constraints Oryctes monocerus
Bioassay of insecticides against three honey bee species in laboratory condit...

Bioassay of insecticides against three honey bee species in laboratory condit...
Microbiological Investigations of Selected Flies of Public Health Importance ...

Microbiological Investigations of Selected Flies of Public Health Importance ...
Poster_05022016
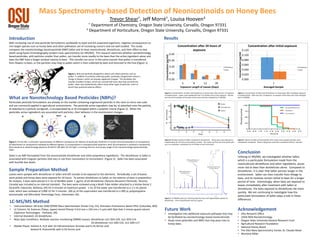
- 1. Sample Preparation Method Leaves were sprayed with dinotefuran or Safari and left outside to be exposed to the elements. Periodically, a set of leaves were picked and honey bees were exposed for 24 hours. To extract dinotefuran or Safari on the exterior of bees in preparation for analysis, 5 bees were placed in 5 mL of distilled water. 1 µg/mL of d3-dinotefuran (Toronto Research Chemicals, Toronto, Canada) was included as an internal standard. The bees were vortexed using a Multi-Tube Holder attached to a Vortex Genie 2 (Scientific Industries, Bohema, NY) for 4 minutes at maximum power. 1 mL of the water was transferred to a 1.5 mL plastic tube, which was vortexed at 5,000 rcf for 5 minutes. 100 µL of the supernatant was transferred to a 300 µL polypropylene autosampler vial (MicroSolv Technology Corp., Eatontown, NJ). LC-MS/MS Method • Instrumentation: AB Sciex 3200 QTRAP Mass Spectrometer (Foster City, CA), Shimadzu Prominence Series HPLC (Columbia, MD) • LC Column: GL Sciences (Tokyo, Japan) Inertsil Phenyl 3 (4.6 mm x 150 mm x 5 µm) with Opti-Solv 2 micron guard column (Optimize Technologies - Portland, OR) • Internal Standard: d3-dinotefuran • Mass Spec Conditions: Multiple reaction monitoring (MRM) masses: dinotefuran m/z 203>129, m/z 203>114 d3-dinotefuran m/z 206>132, m/z 206>117 • Mobile Phase: Solvent A: H2O with 10 mM ammonium formate and 0.1% formic acid Solvent B: Acetonitrile with 0.1% formic acid Results Future Work • Investigation into additional exposure pathways that may be facilitated by nanotechnology based neonicotinoids. • Study more pesticides and NBPs that may pose risks to honey bees. Mass Spectrometry-based Detection of Neonicotinoids on Honey Bees Trevor Shear†, Jeff Morré†, Louisa Hooven‡ † Department of Chemistry, Oregon State University, Corvallis, Oregon 97331 ‡ Department of Horticulture, Oregon State University, Corvallis, Oregon 97331 D ays %Mortality 5 1 0 1 5 2 1 2 6 3 1 3 6 0 2 5 5 0 7 5 1 0 0 W a te r D in o te fu ra n S a fa ri Figure 3. Concentrations of Safari and dinotefuran on honey bees after 24 hours of exposure to treated leaves. Leaves were weathered from 5 to 39 days prior to bee exposure. Results are from three samples of five bees at four time intervals, analyzed by LC-MS/MS. Figure 4. Residual toxicity of Safari compared to dinotefuran. Honey bees were exposed to treated leaves for 24 hours and mortality counted. The results are from ten time points with up to six replicates, consisting of up to 30 bees at each time point. Figure 5. Concentration of Safari and dinotefuran on honey bees after immediate exposure to treated leaves. After one hour of exposure, six samples of five bees each were analyzed with LC-MS/MS. 0 .0 0 .5 1 .0 1 .5 2 .0 2 .5 3 .0 3 .5 4 .0 4 .5 0 2 5 5 0 7 5 1 0 0 H o u r s Percentsurvival D in o te fu ra n S a fa r i C o n tro l Figure 6. Toxicity of Safari and dinotefuran after leaves were sprayed and bees were immediately introduced. Dead or dying bees were then counted at half hour intervals. Introduction With increasing use of new pesticide formulations worldwide to repel and kill unwanted organisms, negative consequences on non-target species such as honey bees and other pollinators are of increasing concern and not well studied. This study compares the nanotechnology based pesticide (NBP) Safari and its base neonicotinoid, dinotefuran, and their effect on bee death using liquid chromatography tandem mass spectrometry (LC-MS/MS). This research examines whether nanotechnology based pesticides, with particles smaller than pollen, can transfer more readily to the bees than the active ingredient alone and does the NBP have a longer residual toxicity to bees. This transfer can occur in the same manner that pollen is transferred from flowers to bees, or the particles may cling to pollen which is then collected by bees and returned to the hive (Figure 1). What are Nanotechnology Based Pesticides (NBPs)? Particulate pesticide formulations are already on the market containing engineered particles in the nano to micro size scale and are commonly applied in agricultural environments. The pesticide active ingredient may be; a) adsorbed onto the particle, b) attached to a particle via ligands, c) encapsulated by or d) entrapped within a polymer matrix (Figure 2). When the pesticides active ingredients are associated with particles, their behavior in the environment changes. Figure 2. On the left, a schematic representation of different nanodevices for delivery of pesticides, fertilizers or nucleic acids (a) adsorption on nanoparticle; (b) attachment on nanoparticle mediated by different ligands; (c) encapsulation in nanoparticulate polymeric shell; (d) entrapment in polymeric nanoparticle. Ghormande et al, Biotechnology Advances 29 (2011) 792–803. On the right, a scanning electron microscopy image of the nanotechnology based pesticide, Safari. Safari is an NBP formulated from the neonicotinoid dinotefuran and other proprietary ingredients. The dinotefuran in Safari is associated with irregular particles that vary in size from nanometers to micrometers (Figure 2). Safari has been associated with bumble bee death. Figure 1. Bees are perfectly designed to attract and collect particles such as pollen. In addition to actively collecting pollen, positively charged bees induce a charge in flowers, which are already negatively charged. This facilitates the transfer of pollen to bees, which are covered with hairs that trap and hold the pollen. Bees may inadvertently collect many other types of particles, some of which have potential adverse effects. 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 Time,min 0.0e0 100% Intensity 11.31 60 70 80 90 100 110 120 130 140 150 160 170 180 190 200 Mass/Charge,Da 129.2 203.2 114.2 87.1 157.2 100.173.1 Figure 7. LC-MS/MS spectrum showing retention time and fragmentation pattern of dinotefuran. Limit of quantitation was 0.01 µg/mL. MRM quantification ion m/z 203.2>129.2 MRM qualification ion m/z 203.2>114.2 dinotefuran parent mass m/z 203.2 (M+H) Acknowledgement • OSU Research Office • USDA NIFA Nanotechnology • Oregon State University General Research Fund • Agricultural Research Foundation • National Honey Board • The OSU Mass Spectrometry Center, Dr. Claudia Maier • Montana Miller Conclusion Utilizing LC-MS/MS, we investigated whether Safari, which is a particulate formulation made from the neonicotinoid dinotefuran and other ingredients, poses more risk to bees than dinotefuran alone. Compared to dinotefuran, it is clear that Safari persists longer in the environment. Safari can then transfer from foliage to bees, and its residues remain lethal to bees for a longer period of time. Interestingly, when bees are exposed to leaves immediately after treatment with Safari or dinotefuran, the bees exposed to dinotefuran die more quickly. We are continuing to investigate how the particulate formulation of Safari plays a role in these differences. 0.00 0.10 0.20 0.30 0.40 5 8 12 39 Concentration(µg/ml) Exposure Length of Leaves (Days) Concentration after 24 hours of exposure Dinotefuran Safari 0.000 0.020 0.040 0.060 0.080 0.100 0.120 1 Concentration(µg/ml) Averaged Sample Concentration after initial exposure Dinotefuran Safari