Enzymes dr arifullah [compatibility mode]
•
6 gefällt mir•603 views
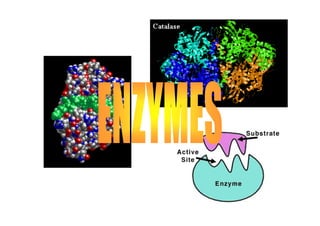
Enzymes are proteins that act as catalysts to speed up biochemical reactions. They have several key characteristics - they are effective in small quantities, efficient, specific, and their activity is affected by their surroundings. There are six main classes of enzymes classified by their catalytic activity. Factors like enzyme concentration, substrate concentration, pH, temperature, and inhibitors can affect an enzyme's activity. Inhibitors bind to enzymes to disrupt their activity, and can be competitive, noncompetitive, or irreversible.
Melden
Teilen
Melden
Teilen
Downloaden Sie, um offline zu lesen
Empfohlen
Empfohlen
Weitere ähnliche Inhalte
Was ist angesagt?
Was ist angesagt? (20)
Andere mochten auch
Andere mochten auch (20)
Molecular Cell Biology Lodish 6th.ppt - Chapter 6 genes, genomics, and chro...

Molecular Cell Biology Lodish 6th.ppt - Chapter 6 genes, genomics, and chro...
Molecular Cell Biology Lodish 6th.ppt - Chapter 12 cellular energetics

Molecular Cell Biology Lodish 6th.ppt - Chapter 12 cellular energetics
Molecular Cell Biology Lodish 6th.ppt - Chapter 5 molecular genetic techniques

Molecular Cell Biology Lodish 6th.ppt - Chapter 5 molecular genetic techniques
Molecular Cell Biology Lodish 6th.ppt - Chapter 1 life begins with cells

Molecular Cell Biology Lodish 6th.ppt - Chapter 1 life begins with cells
Molecular Cell Biology Lodish 6th.ppt - Chapter 3 protein structure and fun...

Molecular Cell Biology Lodish 6th.ppt - Chapter 3 protein structure and fun...
Molecular Cell Biology Lodish 6th.ppt - Chapter 15 cell signaling i signal ...

Molecular Cell Biology Lodish 6th.ppt - Chapter 15 cell signaling i signal ...
Molecular Cell Biology Lodish 6th.ppt - Chapter 4 basic molecular genetic m...

Molecular Cell Biology Lodish 6th.ppt - Chapter 4 basic molecular genetic m...
Ähnlich wie Enzymes dr arifullah [compatibility mode]
Ähnlich wie Enzymes dr arifullah [compatibility mode] (20)
Enzyme. defination ,classification and application

Enzyme. defination ,classification and application
D. Pharm BIOCHEMISTRY AND CLINICAL PATHOLOGY Enzyme

D. Pharm BIOCHEMISTRY AND CLINICAL PATHOLOGY Enzyme
Mehr von Rione Drevale
Mehr von Rione Drevale (20)
Kürzlich hochgeladen
Kürzlich hochgeladen (20)
Polkadot JAM Slides - Token2049 - By Dr. Gavin Wood

Polkadot JAM Slides - Token2049 - By Dr. Gavin Wood
Boost Fertility New Invention Ups Success Rates.pdf

Boost Fertility New Invention Ups Success Rates.pdf
Axa Assurance Maroc - Insurer Innovation Award 2024

Axa Assurance Maroc - Insurer Innovation Award 2024
Navi Mumbai Call Girls 🥰 8617370543 Service Offer VIP Hot Model

Navi Mumbai Call Girls 🥰 8617370543 Service Offer VIP Hot Model
Why Teams call analytics are critical to your entire business

Why Teams call analytics are critical to your entire business
"I see eyes in my soup": How Delivery Hero implemented the safety system for ...

"I see eyes in my soup": How Delivery Hero implemented the safety system for ...
Apidays New York 2024 - The value of a flexible API Management solution for O...

Apidays New York 2024 - The value of a flexible API Management solution for O...
Strategize a Smooth Tenant-to-tenant Migration and Copilot Takeoff

Strategize a Smooth Tenant-to-tenant Migration and Copilot Takeoff
EMPOWERMENT TECHNOLOGY GRADE 11 QUARTER 2 REVIEWER

EMPOWERMENT TECHNOLOGY GRADE 11 QUARTER 2 REVIEWER
TrustArc Webinar - Unlock the Power of AI-Driven Data Discovery

TrustArc Webinar - Unlock the Power of AI-Driven Data Discovery
ICT role in 21st century education and its challenges

ICT role in 21st century education and its challenges
Apidays Singapore 2024 - Building Digital Trust in a Digital Economy by Veron...

Apidays Singapore 2024 - Building Digital Trust in a Digital Economy by Veron...
Cloud Frontiers: A Deep Dive into Serverless Spatial Data and FME

Cloud Frontiers: A Deep Dive into Serverless Spatial Data and FME
Enzymes dr arifullah [compatibility mode]
- 2. ENZYMES Outline Definition Characteristics of enzymes Types of enzymes Factors effecting enzyme activity
- 3. The definition & Characteristics of enzymes • A protein that acts as a catalyst, speeding the rate at which a biochemical reaction proceeds. a type of protein Characters: • a catalyst • Effective in smaller quantities • efficient and specific • reaction can be reversed • activities affected by surroundings • need helpers – cofactors/prostethic grps • involve in multiple steps of biochemical pathways
- 4. Classification of enzymes 6 main classes according to International Union of Biochemistry and Molecular Biology (IUBMB): 1. oxidoreductase 2. transferase 3. hydrolase 4. lyase 5. isomerase 6. ligase
- 6. • Function: catalyzes oxidation-reduction reactions • e.g. alcohol dehydrogenase • Other e.g. Biliverdin reductase; Glucose oxidase
- 7. • Function: catalyzes reactions involving transfer of functional groups • e.g. Hexokinase • Other e.g. Glycoaldehyde transferase; DNA nucleotidylexotransferase
- 8. • Function: catalyzes hydrolytic reactions involving use of water mol. • e.g. Triacylglycerol lipase • Other e.g. -amino acid esterase; Oxaloacetase H2O
- 9. • Function: catalyzes cleavage of C-C, C-O, C- N and other bonds by other means than by hydrolysis or oxidation • e.g. Lysine decarboxylase • other e.g.: threonine aldolase [EC 4.1.2.5]; cystine lyase
- 10. • Function: catalyzes intramolecular arrangement • e.g. Maleate isomerase • Other e.g. Inositol-3-phosphate synthase; Maltose epimerase]
- 11. • Function: catalyzes the joining of two molecules with concomitant hydrolysis of the diphosphate bond in ATP or a similar triphosphate • e.g. Pyruvate carboxylase • Other e.g. GMP synthase; DNA ligase
- 12. Enzyme as protein • exhibits characteristics like other proteins • primary structure amino acid sequence e.g.: human pancreatic lipase (467 amino acids) N-Met1-…-Ser171-...-Asp194-...-His281-…-Cys467-C human trypsin (247 amino acids) N-Met1-…-His63-…-Asp107-…-Ser200-…-Ser247-C
- 14. Aspartate carbamoyltransferase’s quartenary structure 2 catalytic trimers 3 regulatory dimers
- 16. How enzyme reacts E + S ES E + P
- 17. Enzyme reaction…hypothesis • lock and key hypothesis: proposed by Emil Fischer (1894) perfect match like specific key & lock
- 20. • induced-fit hypothesis: proposed by Daniel Koshland (1958) shows elasticity/flexibility at active site well-accepted
- 23. Factors affecting enzyme activity • enzyme concentration • substrate concentration • pH • temperature • inhibitors
- 25. A B
- 26. Change in pH effects the pull/push force of polar/nonpolar intramolecules which change the enzyme shape as well as active site – in acidic conditions, basic grps are protonized while in basic conditions, acidic grps are deprotonized.
- 30. A C B
- 31. • substances which bind to enzyme & disrupt the enzyme activity by blocking the production of ES-complex or E + P • reversible & irreversible
- 32. • involves the noncovalent links between inhibitor and enzyme • 2 types: competitive inhibitor noncompetitive inhibitor uncompetitive inhibitor Reversible inhibitors
- 33. competitive inhibitor mol. similar to substrate compete with substrate for active site e.g.: succinate dehydrogenase (E); succinate (S); malonate (I)
- 34. noncompetitive inhibitor mol./ion attaches to second site (other than active site) at enzyme surface e.g.: prostaglandin synthase (E); arachidonate (S); aspirin (I)
- 35. uncompetitive inhibitor binds to ES complex, forming an inactive ESI complex e.g..: polymerase (E); nucleic acid (S); nevirapine (I) - HIV
- 36. • covalently bonded – react with functional grp at active site, blocking active site from substrate rendering the enzyme inactive • mostly are toxic substances • e.g.: (see attached list) Irreversible inhibitors
