Electron,nutron,proton
•Als PPTX, PDF herunterladen•
7 gefällt mir•6,260 views
About electron,nutron,proton
Melden
Teilen
Melden
Teilen
Weitere ähnliche Inhalte
Was ist angesagt?
Was ist angesagt? (20)
Grade 8 Chemistry Structure of Matter : Atoms, Molecules and Ions

Grade 8 Chemistry Structure of Matter : Atoms, Molecules and Ions
Andere mochten auch
Andere mochten auch (20)
Ähnlich wie Electron,nutron,proton
Ähnlich wie Electron,nutron,proton (20)
Kürzlich hochgeladen
Mattingly "AI & Prompt Design: The Basics of Prompt Design"

Mattingly "AI & Prompt Design: The Basics of Prompt Design"National Information Standards Organization (NISO)
Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"

Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"National Information Standards Organization (NISO)
APM Welcome, APM North West Network Conference, Synergies Across Sectors

APM Welcome, APM North West Network Conference, Synergies Across SectorsAssociation for Project Management
Kürzlich hochgeladen (20)
Measures of Dispersion and Variability: Range, QD, AD and SD

Measures of Dispersion and Variability: Range, QD, AD and SD
Web & Social Media Analytics Previous Year Question Paper.pdf

Web & Social Media Analytics Previous Year Question Paper.pdf
Kisan Call Centre - To harness potential of ICT in Agriculture by answer farm...

Kisan Call Centre - To harness potential of ICT in Agriculture by answer farm...
Mattingly "AI & Prompt Design: The Basics of Prompt Design"

Mattingly "AI & Prompt Design: The Basics of Prompt Design"
Interactive Powerpoint_How to Master effective communication

Interactive Powerpoint_How to Master effective communication
Disha NEET Physics Guide for classes 11 and 12.pdf

Disha NEET Physics Guide for classes 11 and 12.pdf
Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"

Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"
APM Welcome, APM North West Network Conference, Synergies Across Sectors

APM Welcome, APM North West Network Conference, Synergies Across Sectors
Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...

Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...
Ecosystem Interactions Class Discussion Presentation in Blue Green Lined Styl...

Ecosystem Interactions Class Discussion Presentation in Blue Green Lined Styl...
Electron,nutron,proton
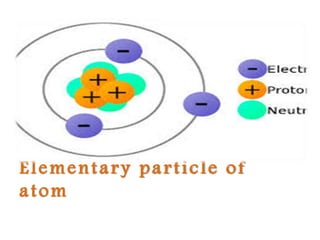
- 3. ELECTRON Electron is a sub atom particle with a negative element. Electric charge. A n electron has no known components or substructure. It is generally though to be elementary particle. An electron has a mass that is approximately 1/1836 that of the proton. Its symbol is e-
- 4. Proton The proton is a sub atomic particle with the symbol P or P+ and a positive electric charge of 1 elementary charge. One or more protons are present in atom . Every proton number is called atomic number. The name of proton was given to the hydrogen nucleus by Earnest Rutherford in1920.
- 5. Neutron The neutron is sub atomic particle .its symbol is n . It has no charge. Its mean ,it is neutral and a mass slightly larger than that of a proton .With exception hydrogen -1 .Nucleic of atoms consist of protons and neutrons . Which are there fore collectively referred to as nucleons.
- 7. Atomic Number In chemistry and physics , the atomic number is the number of protons founds in the nucleus of an atom and therefore identical to the to the charge number of the nucleus .It is conventionally represented by symbol Z . The atomic number uniquely identifies a chemical element .In the atom of neutral charge . The atomic number is equal to the number of electron.
- 8. Mass number The mass number ‘ A’ which is the number of nucleons .The total number of protons and neutrons in the nucleus of atom.
- 9. Atomic mass Relative atomic mass is a dimensional physical quantity. The ration of the average mass of atoms of element to 1/12 the mass of an atom of carbon 12 (known as the unified mass unit)
- 13. Quantum Number: Any set of number that together fully determine the sate of quantum mechanical system by quantifying its individual properties. For example four quantum numbers are used to specify the quantum state of an electron orbiting the nucleus of atom .one characterizes its basic orbital energy level one the shape of its orbit, one the orientation of its orbit of its orbit relative to other orbit and one its spin.
