Guide to Molecular Cloning - Download the Guide
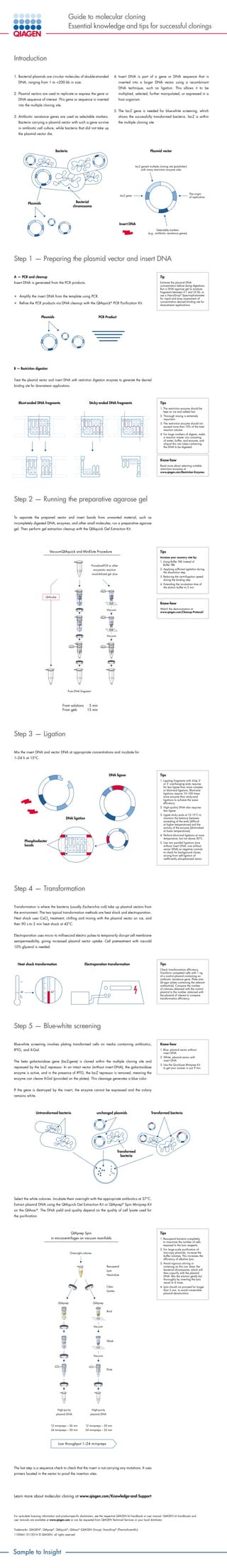
- 1. Guide to molecular cloning Essential knowledge and tips for successful clonings Introduction 1. Bacterial plasmids are circular molecules of double-stranded DNA, ranging from 1 to 200 kb in size. 2. Plasmid vectors are used to replicate or express the gene or DNA sequence of interest. This gene or sequence is inserted into the multiple cloning site. 3. Antibiotic resistance genes are used as selectable markers. Bacteria carrying a plasmid vector with such a gene survive in antibiotic cell culture, while bacteria that did not take up the plasmid vector die. 4. Insert DNA is part of a gene or DNA sequence that is inserted into a larger DNA vector using a recombinant DNA technique, such as ligation. This allows it to be multiplied, selected, further manipulated, or expressed in a host organism. 5. The lacZ gene is needed for blue-white screening, which shows the successfully transformed bacteria. lacZ is within the multiple cloning site Step 1 — Preparing the plasmid vector and insert DNA A — PCR and cleanup Insert DNA is generated from the PCR products. • Amplify the insert DNA from the template using PCR. • Refine the PCR products via DNA cleanup with the QIAquick® PCR Purification Kit. B — Restriction digestion Treat the plasmid vector and insert DNA with restriction digestion enzymes to generate the desired binding site for downstream applications. Step 2 — Running the preparative agarose gel To separate the prepared vector and insert bands from unwanted material, such as incompletely digested DNA, enzymes, and other small molecules, run a preparative agarose gel. Then perform gel extraction cleanup with the QIAquick Gel Extraction Kit. Step 3 — Ligation Mix the insert DNA and vector DNA at appropriate concentrations and incubate for 1–24 h at 15°C. Step 4 — Transformation Transformation is where the bacteria (usually Escherichia coli) take up plasmid vectors from the environment. The two typical transformation methods are heat shock and electroporation. Heat shock uses CaCl2 treatment, chilling and mixing with the plasmid vector on ice, and then 90 s to 2 min heat shock at 42°C. Electroporation uses micro- to millisecond electric pulses to temporarily disrupt cell membrane semipermeability, giving increased plasmid vector uptake. Cell pretreatment with ice-cold 10% glycerol is needed. Step 5 — Blue-white screening Blue-white screening involves plating transformed cells on media containing antibiotics, IPTG, and X-Gal. The beta galactosidase gene (lacZ-gene) is cloned within the multiple cloning site and repressed by the lacZ repressor. In an intact vector (without insert DNA), the galactosidase enzyme is active, and in the presence of IPTG, the lacZ repressor is removed, meaning the enzyme can cleave X-Gal (provided on the plates). This cleavage generates a blue color. If the gene is destroyed by the insert, the enzyme cannot be expressed and the colony remains white. Select the white colonies. Incubate them overnight with the appropriate antibiotics at 37°C. Extract plasmid DNA using the QIAquick Gel Extraction Kit or QIAprep® Spin Miniprep Kit on the QIAvac® . The DNA yield and quality depend on the quality of cell lysate used for the purification. Sample to Insight Tip Estimate the plasmid DNA concentration before doing digestions. Run a DNA agarose gel to analyze fragments between 0.1 and 25 kb, or use a NanoDrop® Spectrophotometer for rapid and easy assessment of concentration.desired binding site for downstream applications. Tips 1. The restriction enzyme should be kept on ice and added last. 2. Thorough mixing is extremely important. 3. The restriction enzyme should not exceed more than 10% of the total reaction volume. 4. For large numbers of digests, make a reaction master mix consisting of water, buffer, and enzyme, and aliquot this into tubes containing the DNA to be digested. Tips Increase your recovery rate by: 1. Using Buffer TAE instead of Buffer TBE 2. Applying sufficient agitation during the dissolution step 3. Reducing the centrifugation speed during the binding step 4. Extending the incubation time of the elution buffer to 5 min Know-how Watch the demonstration at www.qiagen.com/Cleanup-Protocol/ Tips 1. Ligating fragments with 4-bp 3’ or 5’ overhanging ends requires far less ligase than more complex or blunt-end ligations. Blunt-end ligations require 10–100 times more enzyme than sticky-end ligations to achieve the same efficiency. 2.High-quality DNA also requires less ligase. 3. Ligate sticky ends at 12–15°C to maintain the balance between annealing of the ends (difficult at higher temperatures) and the activity of the enzyme (diminished at lower temperatures). 4. Perform blunt-end ligations at room temperature, but not above 30°C. 5. Use two parallel ligations (one without insert DNA, one without vector DNA) as negative controls to check for background clones arising from self-ligation of inefficiently phosphatized vector. Tips Check transformation efficiency. Transform competent cells with 1 ng of a control plasmid containing an antibiotic resistance gene. Plate onto LB-agar plates containing the relevant antibiotic(s). Compare the number of colonies obtained with the control plasmid to the number obtained with the plasmid of interest to compare transformation efficiency. Know-how 1. Blue: plasmid vector without insert DNA 2. White: plasmid vector with insert DNA 3. Use the QuickLyse Miniprep Kit to get your answer in just 9 min. Tips 1. Resuspend bacteria completely to maximize the number of cells exposed to the lysis reagents. 2. For large-scale purification of low-copy plasmids, increase the buffer volumes. This increases the efficiency of alkaline lysis. 3. Avoid vigorous stirring or vortexing as this can shear the bacterial chromosome, which will then copurify with the plasmid DNA. Mix the solution gently but thoroughly by inverting the lysis vessel 4–6 times. 4. Lysis should not proceed for longer than 5 min, to avoid irreversible plasmid denaturation. Know-how Read more about selecting suitable restriction enzymes at www.qiagen.com/Restriction-Enzymes. Learn more about molecular cloning at www.qiagen.com/Knowledge-and-Support For up-to-date licensing information and product-specific disclaimers, see the respective QIAGEN kit handbook or user manual. QIAGEN kit handbooks and user manuals are available at www.qiagen.com or can be requested from QIAGEN Technical Services or your local distributor. Trademarks: QIAGEN® , QIAprep® , QIAquick® , QIAvac® (QIAGEN Group); NanoDrop® (ThermoScientific) 1100661 01/2016 © QIAGEN, all rights reserved The last step is a sequence check to check that the insert is not carrying any mutations. It uses primers located in the vector to proof the insertion sites. Bacteria Plasmids Bacterial chromosome Plasmid vector Insert DNA The origin of replication Selectable markers (e.g., antibiotic resistance genes) VacuumQIAquick and MinElute Procedure QIAquick and MinElute Procedure PCR or other enzymatic reaction or solubilized gel slice Pure DNA fragment From solutions 5 min From gels 15 min ProcedurePCR or other enzymatic reaction orsolubilized gel slice QIAcube Pure DNA fragment From solutions 5 min From gels 15 min Vacuum Vacuum lacZ gene lacZ geneA multiple cloning site (polylinker) with many restriction enzyme sites Blunt-ended DNA fragments Sticky-ended DNA fragments Plasmids PCR Product A T T T T T T GT A A A A A A CG G GC C C C T A AA ACA T TT TGC C C G AAT TC T CG A GC G G G G AG C TC G Heat shock transformation Electroporation transformation 42° 0 40 Untransformed bacteria unchanged plasmids Transformed bacteria Transformed bacteria Low throughput 1–24 minipreps Overnight cultures Resuspend Lyse Neutralize Clear lysates QIAprep QIAprep Vacuum Vacuum Bind Wash Elute High-purity plasmid DNA 12 minipreps – 26 min 24 minipreps – 30 min 12 minipreps – 20 min 24 minipreps – 25 min High-purity plasmid DNA QIAprep Spin in microcentrifuges on vacuum manifolds Low throughput 1–24 minipreps AGT CG C TCA GC G Phosphodiester bonds DNA ligation DNA ligase
