Up Close and Personalizing Gastric Cancer Care: Precision Medicine With HER2-Targeted Therapies and Other Novel Agents
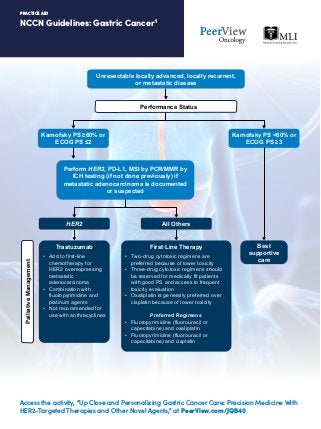
- 1. NCCN Guidelines: Gastric Cancer1 PRACTICE AID Access the activity, “Up Close and Personalizing Gastric Cancer Care: Precision Medicine With HER2-Targeted Therapies and Other Novel Agents,” at PeerView.com/JQB40 Unresectable locally advanced, locally recurrent, or metastatic disease Karnofsky PS ≥60% or ECOG PS ≤2 Karnofsky PS <60% or ECOG PS ≥3 Perform HER2, PD-L1, MSI by PCR/MMR by ICH testing (if not done previously) if metastatic adenocarcinoma is documented or suspected Best supportive care HER2 All Others Trastuzumab • Add to first-line chemotherapy for HER2 overexpressing metastatic adenocarcinoma • Combination with fluoropyrimidine and platinum agents • Not recommended for use with anthracyclines First·Line Therapy • Two-drug cytotoxic regimens are preferred because of lower toxicity • Three-drug cytotoxic regimens should be reserved for medically fit patients with good PS and access to frequent toxicity evaluation • Oxaliplatin is generally preferred over cisplatin because of lower toxicity Preferred Regimens • Fluoropyrimidine (fluorouracil or capecitabine) and oxaliplatin • Fluoropyrimidine (fluorouracil or capecitabine) and cisplatin Performance Status PalliativeManagement
- 2. NCCN Guidelines: Gastric Cancer1 PRACTICE AID CPS: combined positive score; dMMR: deficient mismatch repair; ECOG: Eastern Cooperative Oncology Group; ICH: International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use; MMR: mismatch repair; MSI: microsatellite instability; MSI-H: MSI-high; PCR: polymerase chain reaction; PD-L1: programmed death ligand-1; PS: performance status. 1. National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines. Gastric Cancer. Version 1.2020 https://www.nccn.org/professionals/physician_gls/pdf/gastric_blocks.pdf. Access the activity, “Up Close and Personalizing Gastric Cancer Care: Precision Medicine With HER2-Targeted Therapies and Other Novel Agents,” at PeerView.com/JQB40 Systemic Therapy for Unresectable Locally Advanced, Recurrent, or Metastatic Disease (When Local Therapy Is Not Indicated) Second-Line and Subsequent Therapy Second-Line Therapy Category Preferred regimens Ramucirumab + paclilaxel 1 Docetaxel 1 Paclitaxel 1 lrinotecan 1 Fluorouracil + irinotecan 2A Pembrolizumab (MSI-H or dMMR tumors) 2A Other recommended regimens Ramucirumab 1 lrinotecan + cisplatin 2A Entrectinib or larotrectinib (NTRK gene fusion–positive tumors) 2A Docetaxel + irinotecan 2B Useful in certain circumstances Fluorouracil + irinotecan + ramucirumab 2B Third-Line Therapy Category Preferred regimens Trifluridine + tipiracil 1 Pembrolizumab (MSI-H or dMMR tumors) 2A Pembrolizumab (gastric adenocarcinoma with PD-L1 expression levels by CPS ≥1) 2A
- 3. NCCN’s Principles of Pathologic Review and Biomarker Testing1 PRACTICE AID Access the activity, “Up Close and Personalizing Gastric Cancer Care: Precision Medicine With HER2-Targeted Therapies and Other Novel Agents,” at PeerView.com/JQB40 lmmunohistochemical Criteria for Scoring HER2 Expression in Gastric Cancer Surgical Specimen Expression Pattern, lmmunohistochemistry Biopsy Specimen Expression Pattern, lmmunohistochemistry HER2 Overexpression Assessment 0 No reactivity or membranous reactivity in <10% of cancer cells No reactivity or no membranous reactivity in any cancer cell Negative 1+ Faint or barely perceptible membranous reactivity in ≥10% of cancer cells; cells are reactive only in part of their membrane Cluster of five or more cancer cells with a faint or barely perceptible membranous reactivity irrespective of percentage of cancer cells positive Negative 2+ Weak to moderate complete, basolateral, or lateral membranous reactivity in ≥10% of cancer cells Cluster of five or more cancer cells with a weak to moderate complete, basolateral, or lateral membranous reactivity irrespective of percentage of cancer cells positive Equivocal 3+ Strong complete, basolateral, or lateral membranous reactivity in ≥10% of cancer cells Cluster of five or more cancer cells with a strong complete, basolateral, or lateral membranous reactivity irrespective of percentage of cancer cells positive Positive
- 4. NCCN’s Principles of Pathologic Review and Biomarker Testing1 PRACTICE AID Access the activity, “Up Close and Personalizing Gastric Cancer Care: Precision Medicine With HER2-Targeted Therapies and Other Novel Agents,” at PeerView.com/JQB40 • Used for locally advanced, recurrent, or metastatic gastric cancer in patients who are candidates for treatment with PD-1 inhibitors. • Performed on formalin-fixed, paraffinembedded (FFPE) tissue • Results interpreted as MSl-high (MSI-H) or mismatch repair-deficient (dMMR) in accordance with CAP DNA Mismatch Repair Biomarker Reporting Guidelines • Refer patients with MSI-H or dMMR tumors to a genetics counselor for further assessment • No loss of nuclear expression of MMR proteins: no evidence of deficient mismatch repair (low probability of MSI-H) • Loss of nuclear expression of one or more MMR proteins: deficient mismatch repair • Microsatellite stable (MSS) • MSI-Low (MSI-L) - 1%-29% of markers exhibit instability - 1 of the 5 National Cancer Institute (NCI) or mononucleotide markers exhibits instability • MSI-High (MSI-H) - ≥30% of the markers exhibit instability - 2 or more of the 5 NCI or mono-nucleotide markers exhibit instability Microsatellite Instability (MSI) or Mismatch Repair (MMR) Testing MMR Interpretation MSI Interpretation
- 5. NCCN’s Principles of Pathologic Review and Biomarker Testing1 PRACTICE AID CAP: College of American Pathologists; NCCN: National Comprehensive Cancer Network; PD-1: programmed cell death protein 1; PD-L1: programmed death-ligand 1. 1. National Comprehensive Cancer Network (NCCN) Clinical Practice Guidelines. Gastric Cancer. Version 1.2020 https://www.nccn.org/professionals/physician_gls/pdf/gastric_blocks.pdf. Access the activity, “Up Close and Personalizing Gastric Cancer Care: Precision Medicine With HER2-Targeted Therapies and Other Novel Agents,” at PeerView.com/JQB40 • Used for locally advanced, recurrent, or metastatic gastric carcinomas in patients who are candidates for treatment with PD-1 inhibitors • An FDA-approved companion diagnostic test for use on FFPE tissue is available as an aid in identifying patients for treatment with PD-1 inhibitors • Should be performed only in CLIA-approved laboratories • This is a qualitative immuno-histochemical assay using anti–PD-L1 antibodies for the detection of PD-L1 protein in FFPE tissues from gastric adenocarcinoma • A minimum of 100 tumor cells must be present in the PD-L1–stained slide for the specimen to be considered adequate for PD-L1 evaluation • A specimen is considered to have PD-L1 expression if the Combined Positive Score (CPS) ≥1 - CPS is the number of PD-L1 staining cells (ie, tumor cells, lymphocytes, macrophages) divided by the total number of viable tumor cells, multiplied by 100 PD-L1 Testing Assessment of PD-L1 Protein Expression in Gastric Cancers
- 6. Selected Clinical Trials in Gastric Cancer1 PRACTICE AID Access the activity, “Up Close and Personalizing Gastric Cancer Care: Precision Medicine With HER2-Targeted Therapies and Other Novel Agents,” at PeerView.com/JQB40 HER2+ Gastric Cancer PD-1 Inhibitor + Monoclonal Antibody + CTX NCT03615326: KEYNOTE-811 Recruiting 732 participants Pembrolizumab + trastuzumab + CTX + placebo ADC NCT03329690: DESTINY-Gastric01 Active (220), not recruiting Trastuzumab deruxtecan vs physician's choice (irinotecan or paclitaxel) ADC + PD-1 Inhibitor NCT04280341 Not yet recruiting (50) RC48-ADC + JS001 Bispecific Antibody + CTX NCT03929666 Recruiting 115 participants ZW25 vs physician's choice ADC NCT03556345 Active (127), not recruiting RC48-ADC ADC NCT04014075: DESTINY-Gastric02 Recruiting 72 participants Trastuzumab deruxtecan CTXorCTLA-4Inhibitor+PD-1Inhibitor+MonoclonalAntibody NCT03409848: INTEGA Recruiting 97 participants Ipilimumab or FOLFOX + nivolumab + trastuzumab Monoclonal Antibody ± PD-1 Inhibitor ± CTX ± Dual CPI NCT04082364: MAHOGANY Recruiting 850 participants Margetuximab + INCMGA00012 + CTX + MGD013 + trastuzumab Bispecific Antibody + PD-1 Inhibitor + CTX NCT04276493 Recruiting 50 participants ZW25 + tislelizumab + CTX Antiangiogenesis + CTX NCT03081143: RAMIRIS Recruiting 429 participants Ramucirumab + FOLFIRI Monoclonal Antibody + PD-1 Inhibitor NCT02689284 Active (95), not recruiting Margetuximab + pembrolizumab ADC + PD-L1 Inhibitor + CTX NCT04379596: DESTINY-Gastric03 Not yet recruiting (220) Trastuzumab deruxtecan vs trastuzumab deruxtecan ± CTX ± durvaumab Phase 3 Phase 2 Phase 1b/2 Phase 2/3 Phase 1
- 7. Selected Clinical Trials in Gastric Cancer1 PRACTICE AID Access the activity, “Up Close and Personalizing Gastric Cancer Care: Precision Medicine With HER2-Targeted Therapies and Other Novel Agents,” at PeerView.com/JQB40 Antiangiogenesis + PARP Inhibitor + Immunotherapy (RiME) NCT03995017 Recruiting 61 participants Ramucirumab + rucaparib ± nivolumab PARP Inhibitor + Antiangiogenesis NCT03008278 Recruiting 49 participants Olaparib + ramucirumab Immunotherapy + Antiangiogenesis + CTX NCT04069273: SEQUEL Not yet recruiting (58) Pembrolizumab + ramucirumab + paclitaxel Immunotherapy + Antiangiogenesis NCT02572687 Active, not recruiting (114) Ramucirumab + durvalumab Immunotherapy + Antiangiogenesis + CTX NCT03966118 Recruiting 59 participants Avelumab + paclitaxel + ramucirumab CTX + Antiangiogenesis NCT03686488 Recruiting 25 participants trifluridine/tipiracil + ramucirumab Phase 2 Phase 1 Phase 1/2 Antiangiogenesis Combinations
- 8. Selected Clinical Trials in Gastric Cancer1 PRACTICE AID Access the activity, “Up Close and Personalizing Gastric Cancer Care: Precision Medicine With HER2-Targeted Therapies and Other Novel Agents,” at PeerView.com/JQB40 Immunotherapy + CTX NCT03745170: ORIENT-16 Recruiting 650 participants Sintilimab + CTX Immunotherapy + CTX NCT03675737: KEYNOTE-859 Recruiting 1,542 participants Pembrolizumab + CTX Immunotherapy + CTX NCT03221426: KEYNOTE-585 Recruiting 1,000 participants Pembrolizumab + CTX Phase 3 Immunotherapy + PARP Inhibitor NCT02734004: MEDIOLA Active, not recruiting (427) Durvalumab + olaparib + bevacizumab Immunotherapy Combinations NCT03281369 Recruiting 410 participants Atezolizumab + various combinations PD-1 + CTLA-4 Inhibition NCT04202601 Recruiting 80 participants Sintilimab + IBI310 Phase 1/2 Immunotherapy and Combinations Immunotherapy + PARP Inhibitor NCT03579784 Recruiting 40 participants Paclitaxel + olaparib then durvalumab + olaparib + paclitaxel Immunotherapy + Monoclonal Antibody NCT04099641 Recruiting 80 participants Bavituximab + pembrolizumab Immunotherapy + DKK1 Neutralizing Antibody + CTX NCT04363801: DisTinGuish Not yet recruiting (72) DKN-01 + tislelizumab ± CTX Immunotherapy + PARP Inhibitor NCT04209686 Not yet recruiting (36) Paclitaxel + olaparib + pembrolizumab Immunotherapy + TKI NCT04164979 Recruiting 20 participants Cabozantinib + pembrolizumab Phase 2 Phase 1 TKI + PD-L1 Inhibitor NCT03539822: CAMILLA Recruiting 30 participants Cabozantinib + durvalumab
- 9. Selected Clinical Trials in Gastric Cancer1 PRACTICE AID ADC: antibody–drug conjugates; CPI: checkpoint inhibitor; CTLA-4: cytotoxic T-lymphocyte–associated protein 4; CTX: chemotherapy; FGFR: fibroblast growth factor receptor; PARP: poly ADP-ribose polymerase; PD-1: programmed cell death protein 1; PD-L1: programmed death-ligand 1. 1. https://clinicaltrials.gov. Access the activity, “Up Close and Personalizing Gastric Cancer Care: Precision Medicine With HER2-Targeted Therapies and Other Novel Agents,” at PeerView.com/JQB40 PARP Inhibitor NCT03427814: PARALLEL 303 Active, not recruiting (540) BGB-290 vs placebo Claudin 18.2-Positive NCT03653507: GLOW Recruiting 500 participants Zolbetuximab + CTX + placebo FGFR Inhibitor NCT03694522: FIGHT Recruiting 548 participants Bemarituzumab + CTX + placebo Claudin 18.2-Positive NCT03504397: SPOTLIGHT Recruiting 550 participants Zolbetuximab + CTX + placebo CTX Platform Combo NCT03368963 Recruiting 64 participants Trifluridine/tipiracil + nanoliposomal irinotecan Phase 3 Phase 1/2 Other Therapies and Strategies AKT Small Molecule Inhibitor NCT01896531 Active (154), not recruiting Ipatasertib + CTX Claudin 18.2-positive NCT03505320: ILUSTRO Recruiting 112 participants Zolbetuximab + pembrolizumab + CTXFGFR Inhibitor NCT04189445 Not yet recruiting (115) Futibatinib Phase 2
- 10. Access the activity, “Up Close and Personalizing Gastric Cancer Care: Precision Medicine With HER2-Targeted Therapies and Other Novel Agents,” at PeerView.com/JQB40 ASCO Guidance on Allocation of Scarce Resources During COVID-19 Pandemic1,2 PRACTICE AID What special training is necessary to prepare my staff for a local outbreak of COVID-19? Train staff on: • Symptom recognition • Screening procedures • Use of Standard Precautions and personal protective equipment (PPE) • Obtaining SARS-COV2 testing for patients according to current testing guidelines • Protocols for triaging and assessing patients quickly • Identifying and referring patients, families, and coworkers to telephone-based mental health services What patient scheduling changes should be made while pandemic restrictions exist? • Because of reduced waiting areas, the number of appointments may have to be decreased or the time between appointments may have to be increased • Postpone routine follow-up visits of patients not on active cancer treatment, including 6-month and 12-month survivorship visits • Schedule brief, remote check-ins with patients on maintenance therapies to ensure that they have sufficient drug supplies; provide instructions on when they should call their provider • Institute direct telecommunication for survivorship check-ins; create a timeline over the next months to schedule calls • Communicate COVID-19 information and the rationale for changes in appointments via direct telecommunication, websites, and patient portals • Consider home collection of routine lab samples instead of patient visits into the clinic; results can be communicated via telephone • For areas not yet affected by widespread, local transmission, postpone nonurgent visits so urgent visits can be scheduled more immediately • Use telemedicine for patients not requiring a physical exam, treatment, or in-office diagnostics • Ask patients to use telephone triage, patient portals, online assessment tools, or to call and speak with a staff member • Conduct remote check-ins to monitor high-risk patients’ symptoms What workplace changes should be made to prepare for a local outbreak of COVID-19? • If necessary, obtain additional PPE for staff members who do not usually use it • Limit facility access to one point of entry, if possible; vendors, minimal ancillary services, most or all visitors, and people younger than age 18 years should be denied access • Consider remote or virtual support services • Establish outside triage stations with social distancing of 6 feet apart to screen patients and visitors for COVID-19 symptoms and fever before appointments • Install barriers or social distancing mechanisms at front desks if screening is not conducted outside of the facility • Convert waiting areas to allow for distancing of at least 6 feet (eg, move chairs, cordon off every other chair) • Convert open infusion suites to semi-private spaces with at least 6 feet distance between patients and/or use available curtains as a barrier between patients • Suspend (or move to a virtual platform) all onsite group and patient activities (eg, yoga, education seminars, support groups) How should clinical trial investigators respond to the COVID-19 pandemic? • ASCO acknowledges that conducting clinical trials will be particularly challenging during this time • The FDA has issued guidance on management of clinical trial patients during the coronavirus pandemic – Go to: https://www.fda.gov/regulatory-information/ search-fda-guidance-documents/fda-guidance- conduct-clinical-trials-medical-products-during- covid-19-public-health-emergency • The National Cancer Institute has issued guidance on the NCI Central Institutional Review Board – https://www.cancer.gov/research/key-initiatives/ covid-19 • For more guidance and information on COVID-19 and HCC management, visit – https://www.asco.org/asco-coronavirus-information – https://ilca-online.org/management-of-hcc-during- covid-19-ilca-guidance/
- 11. Access the activity, “Up Close and Personalizing Gastric Cancer Care: Precision Medicine With HER2-Targeted Therapies and Other Novel Agents,” at PeerView.com/JQB40 ASCO: American Society of Clinical Oncology; HCC: hepatocellular carcinoma. 1. https://www.sirweb.org/practice-resources/toolkits/covid-19-toolkit/covid-19-planning. 2. https://www.asco.org/asco-coronavirus-information/provider-practice-preparedness-covid-19. ASCO Guidance on Allocation of Scarce Resources During COVID-19 Pandemic1,2 PRACTICE AID What screening and infection prevention and control practices should my workplace undertake? • To address asymptomatic or presymptomatic transmission, everyone should put on a face mask or other face covering—regardless of symptoms—before entering the facility • When scheduling appointments, ask patients to call ahead and discuss the need to reschedule their appointments if they develop symptoms of a respiratory infection (eg, cough, sore throat, fever) on the day they are scheduled to be seen • Contact the patient the day before the appointment and screen for symptoms of cough, sore throat, fever, or other flu-like symptoms – If symptoms are present, use triage protocols to determine if an appointment is necessary or if the patient can be managed from home – If the patient can be managed from home, the patient should be rescheduled until such time when he or she is determined to be no longer infectious What screening and infection prevention and control practices should my workplace undertake upon patient arrival or during the visit? • Allow only essential visitors who are not displaying symptoms of a respiratory or other infection; if essential, limit the number to one visitor per patient for all provider visits • Visitors should not enter communal treatment areas—ask them to wait in vehicles or return after treatment • Encourage use of alternative mechanisms for patient and visitor interactions, such as video-calls on cell phones or tablets • Healthcare personnel should wear facemasks at all times while they are in the healthcare facility; when available, facemasks are preferred over cloth face coverings, because they protect the wearer from exposure to infectious material from others • Healthcare personnel screening patients and visitors at arrival should wear PPE (including masks) and have access to waste bins and cleaning/disinfecting agents • Ask patients and visitors if they have symptoms of cough, sore throat, or fever; if they’ve been out of the country in the past 14 days; or if they’ve been exposed to anyone with respiratory symptoms or known COVID-19; when available, use an infrared thermometer to take temperatures during screening • Provide signage with COVID-19 screening questions and visualization of symptoms for all patient/visitors, as well as patient education materials and illustrations of proper hygiene for infection prevention and symptoms to report • Patients with suspected infection should receive a facemask and be rapidly isolated until more thorough screening or testing can be conducted. Isolation should take place in an exam room or other private area with the door closed • All staff entering the room of a patient with known or suspected COVID-19 should adhere to Standard Precautions and use a N95 respirator or facemask, gown, gloves, and eye protection; cloth face coverings are not proven effective PPE and should not be worn for the care of patients with known or suspected COVID-19 or other situations where a respirator or facemask is warranted • Establish a plan of action for patients who present with respiratory symptoms (eg, resource for testing, schedule patient with primary care or local/health department)
