Neoadjuvant, Adjuvant, or Both: How to Solve the Puzzle of Perioperative Immunotherapy, Individualize Treatment Plans, and Improve Cure Rates in Resectable NSCLC
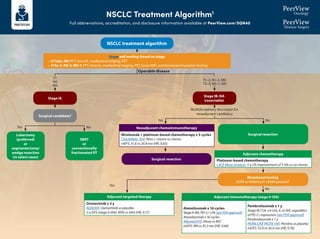
- 1. NSCLC Treatment Algorithm1 Full abbreviations, accreditation, and disclosure information available at PeerView.com/SQN40 Stage and workup based on stage • cT1abc, N0: PFT, bronch, mediastinal staging, PET • cT2a-4, N0-3, M0-1: PFT, bronch, mediastinal staging, PET, brain MRI, and biomarker/mutation testing Surgical candidate? Lobectomy (preferred) or segmentectomy/ wedge resection (in select cases) SBRT or conventionally fractionated RT Surgical resection Mutational testing EGFR ex19del/ex21 L858R present? Surgical resection T1 N0 M0 Operable disease Yes Yes Yes No No No Multidisciplinary discussion for neoadjuvant candidacy T1–2, N1–2, M0 T3–4, N0–1, M0 Neoadjuvant chemoimmunotherapy Nivolumab + platinum-based chemotherapy x 3 cycles CheckMate -816: Nivo + chemo vs chemo mEFS: 31.6 vs 20.8 mo (HR, 0.63) Adjuvant chemotherapy Platinum-based chemotherapy LACE Meta-analysis: 5-y OS improvement of 5.4% vs no chemo Adjuvant immunotherapy (stage II-IIIA) Atezolizumab x 16 cycles StageII-IIIA,PD-L1≥1%(seeFDAapproval) Atezolizumab x 16 cycles IMpower010: Atezo vs BSC mDFS: NR vs 35.3 mo (HR, 0.66) Pembrolizumab x 1 y Stage IB (T2a ≥4 cm), II, or IIIA, regardless of PD-L1 expression (see FDA approval) Pembrolizumab x 1 y PEARLS/KEYNOTE-091: Pembro vs placebo mDFS: 53.6 vs 42.0 mo (HR, 0.76) Adjuvant targeted therapy Osimertinib x 3 y ADAURA: Osimertinib vs placebo 2-y DFS (stage II-IIIA): 90% vs 44% (HR, 0.17) NSCLC treatment algorithm Stage IB-IIIA (resectable) Stage IA
- 2. NSCLC Treatment Algorithm1 Full abbreviations, accreditation, and disclosure information available at PeerView.com/SQN40 Stage IIIA (unresectable) or IIIB/C Definitive chemoradiation → durvalumab Concurrent platinum-based chemotherapy and radiation with consolidation durvalumab PACIFIC: Durvalumab vs placebo mPFS: 16.8 vs 5.6 mo (HR, 0.52) BRAF V600E Dabrafenib + trametiniba BRF113928: Dabrafenib + trametinib single arm ORR: 64% (95% CI, 46-79) 2L: KRAS G12C Sotorasib CodeBreaK100: Sotorasib single arm ORR: 37.1% (95% CI, 29-46); mPFS: 6.8 mo ALK Alectiniba ALEX: Alectinib vs crizotinib 1-y PFS: 68.4% vs 48.7% (HR, 0.47) Brigatiniba ALTA-1L: Brigatinib vs crizotinib mPFS: 24 vs 11.1 mo (HR, 0.48) Lorlatiniba CROWN: Lorlatinib vs crizotinib mPFS: NR vs 9.3 mo, (HR, 0.28); 1-y PFS: 78% vs 39% Ceritinib ASCEND-4: Ceritinib vs chemo mPFS: 16.6 vs 8.1 mo (HR, 0.55) Crizotinib PROFILE 1007: Crizotinib vs chemo mPFS: 7.7 vs 3 mo (HR, 0.49) NTRK Larotrecteniba Entrectiniba ALKA/STARTRK: Entrectinib single arm ORR: 70% (NSCLC) RET Selpercatiniba LIBRETTO-001: Selpercatinib single arm ORR: 64%; mDOR: 17.5 mo Pralsetiniba ARROW: Pralsetinib single arm ORR: 61% (95% CI, 50–71) 2L: EGFR (ex20) Amivantamab CHRYSALIS: Amivantamab single arm CBR: 74% (95%CI, 63-83); mPFS: 8.3 mo Mobocertinib AP32788-15-101: Mobocertinib single arm DCR: 78% (95% CI, 69-85); mPFS: 7.3 mo ROS1 Crizotiniba PROFILE 1001: Crizotinib single arm ORR: 72% (95% CI, 58-84) Entrectiniba ALKA STARTRK: Entrectinib single arm ORR: 67.1%; mPFS: 19 mo Ceritinib YONSEI: Ceritinib single arm ORR: 67% (95% CI, 48-81) EGFR (ex19 del or L858R) Osimertiniba FLAURA: Osimertinib vs erlotinib/gefitinib mPFS: 18.9 vs 10.2 mo (HR, 0.46) Erlotinib EURTAC: Erlotinib vs chemo mPFS: 9.7 vs 5.2 mo (HR, 0.37) Afatinib LUX-Lung 3: Afatinib vs cis/pemetrexed mPFS: 13.6 vs 6.9 mo (HR, 0.47) Gefitinib IFUM: Gefitinib single arm mPFS: 9.7 mo Dacomitinib ARCHER 1050: Dacomitinib vs gefitinib mOS: 34.1 vs 27 mo (HR, 0.75) Erlotinib + ramucirumab RELAY: Erlotinib + ramucirumab vs erlotinib mPFS: 19.4 vs 12.4 mo (HR, 0.59) Erlotinib + bevacizumab ARTEMIS-CTONG1509: Erlotinib + bevacizumab vs erlotinib mPFS: 17.9 vs 11.2 mo (HR, 0.55) MET (exon 14) Capmatiniba GEOMETRY mono-1: Capmatinib single arm mPFS: 12.4 mo Tepotiniba VISION: Tepotinib single arm mPFS: 8.5–11 mo 2L: HER2 Trastuzumab deruxtecan DESTINY-Lung01 T-DXd single arm ORR: 55% (95% CI, 44-65); mPFS: 8.2 mo T1-2, N2–3, M0 T3, N1–3, M0 T4, N0–3, M0 Tx Nx M1 Actionable mutation detected • EGFR (ex19, ex20ins) • ALK • ROS1 • BRAF V600E • RET • MET (ex14) • HER2 • NTRK1/2/3 • KRAS G12C Mutation (minimum EGFR; broad NGS if possible) and PD-L1 testing NSCLC treatment algorithm Stage and workup based on stage • cT1abc, N0: PFT, bronch, mediastinal staging, PET • cT2a-4, N0-3, M0-1: PFT, bronch, mediastinal staging, PET, brain MRI, and biomarker/mutation testing Please see the next page for recommendations if no actionable mutation is detected Stage IV Adagrasib KRYSTAL-1: Adagrasib single arm ORR: 43% (95% CI, 34-53); mDOR: 8.5 mo
- 3. NSCLC Treatment Algorithm1 Full abbreviations, accreditation, and disclosure information available at PeerView.com/SQN40 a Denotes NCCN-preferred regimens. 1. Created by Aakash Desai, MBBS, MPH, and Matthew Ho, MD, PhD. Used with permission from the authors. PD-L1 1% IMMUNOTHERAPY + CHEMOTHERAPY SQUAMOUS: • Pembrolizumab + chemotherapya (carboplatin + paclitaxel/nab-paclitaxel) KEYNOTE-407: Pembro + chemo vs chemo mPFS: 6.4 vs 4.8 mo (HR, 0.56); mOS: 15.9 vs 11.3 mo (HR, 0.64) NONSQUAMOUS: • Pembrolizumab + chemotherapy (carboplatin + pemetrexed)a KEYNOTE-189: Pembro + chemo vs chemo mPFS: 8.8 vs 4.9 mo (HR, 0.52); 12-mo OS: 69% vs 49% (HR, 0.49) • Atezolizumab + chemotherapy (carboplatin + paclitaxel + bevacizumab) IMpower150: Atezo + chemo vs chemo mPFS: 8.3 vs 6.8 mo (HR, 0.62) DUAL IMMUNOTHERAPY + CHEMOTHERAPY Nivolumab + ipilimumab + chemo (2 cycles) CheckMate -9LA: Nivo/ipi + chemo vs chemo mOS: 14.1 vs 10.7 mo Durvalumab + tremelimumab + chemo (4 cycles) POSEIDON: Durva/treme + chemo vs chemo mOS: 14 vs 11.7 mo (HR, 0.77) DUAL IMMUNOTHERAPY Nivolumab + ipilimumab CheckMate -227: Nivo/ipi vs chemo mOS: 17.1 vs 14.9 mo DUAL IMMUNOTHERAPY + CHEMOTHERAPY Nivolumab + ipilimumab + chemo (2 cycles) CheckMate -9LA: Nivo/ipi + chemo vs chemo mOS: 14.1 vs 10.7 mo Durvalumab + tremelimumab + chemo (4 cycle) POSEIDON: Durva/treme + chemo vs chemo mOS: 14 vs 11.7 mo (HR, 0.77) IMMUNOTHERAPY MONOTHERAPY Pembrolizumab KEYNOTE-042: Pembro vs plat-based chemo mOS: 16.7 vs 12.1 mo (HR, 0.81) Ramucirumab + docetaxela REVEL: Ram/docetaxel vs docetaxel; mOS: 10.5 vs 9.1 mo (HR, 0.86) Docetaxela TAX320: Docetaxel vs vinorelbine/ifosfamide; 1-y OS: 32% vs 19% Gemcitabine DUAL IMMUNOTHERAPY Nivolumab + ipilimumab CheckMate -227: Nivo/ipi vs chemo mOS: 17.1 vs 14.9 mo DUAL IMMUNOTHERAPY + CHEMOTHERAPY Nivolumab + ipilimumab + chemo (2 cycles) CheckMate -9LA: Nivo/ipi + chemo vs chemo OS: 14.1 vs 10.7 mo Durvalumab + tremelimumab + chemo (4 cycles) POSEIDON: Durva/treme + chemo vs chemo mOS: 14 vs 11.7 mo (HR, 0.77) PD-L1 1%-49% IMMUNOTHERAPY + CHEMOTHERAPY SQUAMOUS: • Pembrolizumab+chemotherapya (carboplatin+paclitaxel/nab-paclitaxel) KEYNOTE-407: Pembro + chemo vs chemo mPFS: 6.4 vs 4.8 mo (HR, 0.56); mOS: 15.9 vs 11.3 mo (HR, 0.64) NONSQUAMOUS: • Pembrolizumab + chemotherapy (carboplatin + pemetrexed)a KEYNOTE-189: Pembro + chemo vs chemo mPFS: 8.8 vs 4.9 mo (HR, 0.52); 12-mo OS: 69% vs 49% (HR, 0.49) • Atezolizumab + chemotherapy (carboplatin + paclitaxel + bevacizumab) IMpower150 : Atezo + chemo vs chemo mPFS: 8.3 vs 6.8 mo (HR, 0.62) • Cemiplimab + chemotherapy (carboplatin + pemetrexed) EMPOWER-Lung 3: Cemi + chemo vs chemo mOS: 21.9 vs 13 mo (HR, 0.7) PD-L1 ≥50% IMMUNOTHERAPY MONOTHERAPY Pembrolizumaba KEYNOTE-024: Pembro vs platinum-based chemo mPFS: 10.3 vs 6 mo (HR, 0.50) Atezolizumaba IMpower110: Atezo vs platinum-based chemo mOS: 20.1 vs 13.1 mo (HR, 0.59) Cemiplimaba EMPOWER-Lung1: Cemi vs platinum-based chemo mPFS: 8.2 vs 5.7 mo; mOS: NR vs 14.2 mo (HR, 0.57) IMMUNOTHERAPY + CHEMOTHERAPY SQUAMOUS: • Pembrolizumab + chemotherapya (carboplatin + paclitaxel/nab- paclitaxel) KEYNOTE-407: Pembro + chemo vs chemo mPFS: 6.4 vs 4.8 mo (HR, 0.56); mOS: 15.9 vs 11.3 mo (HR, 0.64) NONSQUAMOUS: • Pembrolizumab + chemotherapya (carboplatin + pemetrexed) KEYNOTE-189: Pembro + chemo vs chemo mPFS: 8.8 vs 4.9 mo (HR, 0.52); 12-mo OS: 69% vs 49% (HR, 0.49) • Atezolizumab + chemotherapy (carboplatin + paclitaxel + bevacizumab) IMpower150: Atezo + chemo vs chemo mPFS: 8.3 vs 6.8 mo (HR, 0.62) No actionable mutation detected (stratify based on PD-L1 staining %) Second-line therapy
- 4. Immune-Related Adverse Events of Cancer Immunotherapies Become Aware and Stay Vigilant Full abbreviations, accreditation, and disclosure information available at PeerView.com/SQN40 What Are irAEs?1 • Immune checkpoint inhibitors are associated with important clinical benefits, but general immunologic enhancement can also lead to a unique spectrum of immune-related adverse events • Any organ system can be affected, but more commonly occurring are pulmonary (pneumonitis), dermatologic (rash, pruritus, blisters, ulcers, vitiligo), gastrointestinal (diarrhea, enterocolitis, transaminitis, hepatitis, pancreatitis), and endocrine (thyroiditis, hypophysitis, adrenal insufficiency) irAEs Endocrine Hyper- or hypothyroidism Hypophysitis Adrenal insufficiency Diabetes Hepatic Hepatitis Renal Nephritis Dermatologic Rash Pruritus Psoriasis Vitiligo DRESS Stevens-Johnson Hematologic Hemolytic anemia Thrombocytopenia Neutropenia Hemophilia Ocular Uveitis Conjunctivitis Scleritis, episcleritis Blepharitis Retinitis Respiratory Pneumonitis Pleuritis Sarcoid-like granulomatosis Cardiovascular Myocarditis Pericarditis Vasculitis Gastrointestinal Colitis Ileitis Pancreatitis Gastritis Neurologic Neuropathy Guillain Barŕe Myelopathy Encephalitis Myasthenia Musculoskeletal Arthritis Dermatomyositis 01 Prevention 02 Anticipation 03 Detection 04 Treatment 05 Monitoring 01 02 03 04 05
- 5. Immune-Related Adverse Events of Cancer Immunotherapies Become Aware and Stay Vigilant Full abbreviations, accreditation, and disclosure information available at PeerView.com/SQN40 Guidance for Surgeons: Suspect, Detect, and Refer for Treatment2,3 • irAEs frequently occur in the perioperative setting, either before or after surgical intervention • irAEs occurring during neoadjuvant immunotherapy are generally manageable and in most cases should not exclude patients from surgery • The onus is on the surgeon to have a high degree of suspicion for potential toxicities in patients treated with immunotherapy • Vague symptoms should not be dismissed, because nonspecific ailments can be indicative of severe toxicity – Rheumatologic toxicities and endocrinopathies are some of the most difficult to recognize, given their relatively nonspecific presentation » For example, fatigue, poor energy, and low mood could represent hypophysitis or adrenal insufficiency – Other toxicities can be essentially asymptomatic » For example, renal and hepatic toxicity are generally only detected on routine labs – Pneumonitis is another relevant irAE requiring awareness by surgeons, as severe pneumonitis could potentially exclude patients from operative therapy, but significant pneumonitis has been rare in trials to date • A comprehensive workup for irAEs, with a thorough history specifically targeted to potential irAEs, should be conducted • Coordinate and collaborate with oncologists and other multidisciplinary experts to optimally diagnose and manage irAEs in patients who have received/are receiving perioperative immunotherapy • The National Comprehensive Cancer Network (NCCN) and American Society of Clinical Oncology (ASCO) have issued guidelines for recognition and management of immune-related adverse events
- 6. Immune-Related Adverse Events of Cancer Immunotherapies Become Aware and Stay Vigilant Full abbreviations, accreditation, and disclosure information available at PeerView.com/SQN40 General Recommendations for Treating irAEs4-7 Increasing intensity of treatment required Grade 2 Grade 1 Grade 3 Grade 4 Moderate Mild Severe Very severe Symptomatic supportive therapy Stop treatment Oral steroids Intravenous steroids. ------------ • Referral to specialist • Strong immune suppressive treatment Increasing grade of irAE intravenous steroids Steroids (PO/IV): 1-2 mg/kg/d prednisone or equivalent, slowly taper over 4-6 weeks For some AEs, treatment can be restarted after resolution (eg, rash); generally, ICI can be continued with endocrinopathies once managed Managed in outpatient/ community setting Generally requires hospital admission
- 7. Immune-Related Adverse Events of Cancer Immunotherapies Become Aware and Stay Vigilant Full abbreviations, accreditation, and disclosure information available at PeerView.com/SQN40 Hold immunotherapy and reassess in 1-2 weeks Pulse oximetry rest and ambulation Consider chest imaging with CT (with contrast preferred) Repeat in 3-4 weeks Moderate (grade 2): 25%-50% lung involved Severe (grade 3-4) Grade 3: all lobes of lung or 50% of lung parenchyma; limited ADLs, oxygen requirement Grade 4: life threatening Hold immunotherapy Infectious workup (nasal swab, sputum, blood) Consider bronchoscopy and BAL Chest imaging with CT contrast Repeat in 3-4 weeks Consider empiric antibiotics Refractory: methylprednisolone 1-2 mg/m2 /day; if no response in 3-4 days, treat as grade 3 Permanently discontinue immunotherapy and move to inpatient care Infectious workup (nasal swab, sputum, blood) Pulmonary and infectious disease consultation Bronchoscopy with BAL Empiric antibiotics Methylprednisolone 1-2 mg/m2 /day; when grade 1, taper over 6 weeks Refractory: infliximab, mycophenolate, or IVIG How Should Pulmonary irAEs Be Diagnosed and Managed?4,8 Pneumonitis: focal or diffuse inflammation of the lung parenchyma (typically identified on CT imaging) Diagnostic work-up: CXR, CT, pulse oximetry; for grade ≥2, may include infectious work-up Mild (grade 1): 25% lung involved Additional considerations • GI and pneumocystis prophylaxis may be offered to patients on prolonged steroid use (12 weeks) • Consider calcium and vitamin D supplementation with prolonged steroid use • Bronchoscopy and biopsy; if clinical picture is consistent with pneumonitis, no need for biopsy Supportive care: smoking cessation and vaccinations (influenza, pneumococcal)
- 8. Immune-Related Adverse Events of Cancer Immunotherapies Become Aware and Stay Vigilant Full abbreviations, accreditation, and disclosure information available at PeerView.com/SQN40 1. Champiat S et al. Ann Oncol. 2016;27:559-574. 2. Helmink BA et al. Ann Surg Oncol. 2020;27:1533-1545. 3. Stiles BM et al. J Thorac Cardiovasc Surg. 2020;160:1376-1382. 4. Brahmer JR et al. J Clin Oncol. 2018;36:1714-1786. 5. https://www.esmo.org/content/ download/124130/2352601/1/ESMO-Patient-Guide-on-Immunotherapy-Side-Effects.pdf. 6. https://www.nccn.org/professionals/physician_gls/pdf/immunotherapy.pdf. 7. Puzanov I et al. J Immunother Cancer. 2017;5:95. 8. Provided courtesy of Marianne Davies, DNP, ACNP, AOCNP, FAAN, 2021; adapted from AIM with Immunotherapy, NCCN, and CTCAE. 9. https://ascopubs.org/doi/full/10.1200/JCO.21.01440. 10. https://www.esmo.org/content/download/124130/2352601/1/ESMO-Patient-Guide-on-Immunotherapy-Side-Effects.pdf. 11. https://www.nccn.org/professionals/physician_gls/pdf/immunotherapy.pdf. 12. https://www.sitcancer.org/research/cancer-immunotherapy-guidelines/irae/immune-checkpoint-inhibitor-related-adverse-events. Additional Guideline Recommendations for Treating irAEs9-12
- 9. Simple Summaries of Significant Studies CheckMate -816 Study of Nivolumab Plus Chemotherapy Before Surgery for NSCLC1,2 Full abbreviations, accreditation, and disclosure information available at PeerView.com/SQN40 Plain language summaries (also called layperson summaries, lay language summaries, simple summaries, and trial results summaries) have been introduced to communicate research and clinical trial results in an understandable way to a broader audience, and they are intended to make the clinical results of these studies understandable and accessible to healthcare providers, researchers, patients, caregivers, and the general public What Are Plain Language Summaries of Research and Clinical Trial Results? The following pages include a simple infographic summary of the CheckMate -816 trial for use as a printable resource The following additional resources explaining the CheckMate -816 trial are also available • Brief video summary: www.nejm.org/do/10.1056/NEJMdo006524/full • Plain-language summary: https://www.futuremedicine.com/doi/epdf/10.2217/fon-2023-0007
- 10. 1. Provided courtesy of Patrick M. Forde, MD. 2. Forde PM et al. N Engl J Med. 2022;386:1973-1985. 358 people from 14 different countries The researchers created 2 groups Who took part in this study? What treatments were used? Why is this study important? Some people with non–small cell lung cancer (NSCLC) have tumors that can be removed surgically. However, the cancer often comes back or spreads to other parts of the body, which may subsequently lead to death Taking chemotherapy (chemo) before or after surgery can reduce the risk of cancer coming back and may help people live longer. However, this only works for some people Nivolumab (nivo) is an immunotherapy; it works by activating a person’s immune system to fight back against cancer cells The goal of the CheckMate -816 study was to find out if nivo plus chemo works better than chemo alone when given before surgery for NSCLC 179 people in the nivo plus chemo group 179 people in the chemo alone group Average age 64 years All had tumors (4 centimeters or larger) in the lungs (and in some cases, the nearby lymph nodes) that could be removed with surgery IIIA IIB IIA IB As the number and letter goes up, this represents a bigger tumor and/or more spread Staging is part of the lung cancer diagnosis. This study included people with stages IB, IIA, IIB, and IIIA NSCLC 7 in 10 were men 6 in 10 had stage IIIA NSCLC Each treatment was taken once every 3 weeks for a total of 3 times Surgery planned to happen within 6 weeks of the last dose CheckMate -816 Study: Nivolumab Plus Chemotherapy Given Before Surgery for Non–Small Cell Lung Cancer1,2
- 11. 1. Provided courtesy of Patrick M. Forde, MD. 2. Forde PM et al. N Engl J Med. 2022;386:1973-1985. What did the researchers look at? What were the main results? Nivo plus chemo Nivo plus chemo Nivo plus chemo Median EFS (number of months half of the people lived without the cancer getting worse or spreading) People who took nivo plus chemo and had a pCR after surgery lived longer without the cancer getting worse or spreading than those who did not have a pCR People alive at 2 years without the cancer getting worse or spreading Chemo Chemo Chemo 8 in 10 7-8 in 10 0-1 in 10 2-3 in 10 4-5 in 10 Event-free survival (EFS) How long did each person live without the cancer getting worse or spreading? PRIMARY ASSESSMENTS ADDITIONAL ASSESSMENTS Pathological complete response (pCR) Were there any cancer cells remaining in the tissue samples obtained from the lungs and lymph nodes after surgery? Overall survival How long did each person live after starting treatment? What adverse events did people have? Most people went on to have surgery in both the nivo plus chemo and chemo groups People who took nivo plus chemo lived longer without the cancer getting worse or spreading (EFS) There was a trend for people who took nivo plus chemo to live longer overall than those who took chemo alone. This remains to be confirmed over time in the study More people who took nivo plus chemo than who took chemo alone had no remaining cancer cells in tissue samples obtained from the lungs and lymph nodes after surgery (pCR) 32 months 21 months CheckMate -816 Study: Nivolumab Plus Chemotherapy Given Before Surgery for Non–Small Cell Lung Cancer1,2
- 12. 1. Provided courtesy of Patrick M. Forde, MD. 2. Forde PM et al. N Engl J Med. 2022;386:1973-1985. What were the adverse events? What do these findings mean? A serious adverse event is one that is life-threatening, requires going to the hospital, or results in death In CheckMate -816, people who took nivo plus chemo instead of chemo alone before their surgery: Lived longer without the cancer getting worse or spreading Were more likely to have lungs and lymph nodes clear of cancer cells after surgery Had a trend to live longer in general, which needs more time to be confirmed Did not have more adverse events About 1 in 10 people in each group had a serious adverse events from treatment Most adverse events from surgery were mild or moderate Most adverse events from treatment were mild or moderate No people in the nivo plus chemo group died because of serious adverse events or serious surgery-related adverse events due to the treatment Chemo Nivo plus chemo 1–2 in 10 1 in 10 Few people had severe or life threatening adverse events from surgery Nivo plus chemo is now an approved treatment in the United States for adults with NSCLC whose tumors are 4 centimeters or larger or have spread to nearby lymph nodes CheckMate -816 Study: Nivolumab Plus Chemotherapy Given Before Surgery for Non–Small Cell Lung Cancer1,2
