Emergency Medicine and Immuno-Oncology Intersect: Recognizing and Managing Cancer Immunotherapy–Related Adverse Effects in the Emergency Department
•
2 gefällt mir•160 views
Daniel Pallin, MD, MPH, and Douglas B. Johnson, MD, MSCI, prepared useful practice aids pertaining to immune-related adverse events for this CME/MOC/CE activity titled "Emergency Medicine and Immuno-Oncology Intersect: Recognizing and Managing Cancer Immunotherapy–Related Adverse Effects in the Emergency Department." For the full presentation, monograph, complete CME/MOC/CE information, and to apply for credit, please visit us at http://bit.ly/2PRv8Ul. CME/MOC/CE credit will be available until November 20, 2019.
Melden
Teilen
Melden
Teilen
Downloaden Sie, um offline zu lesen
Empfohlen
James T. Kenney, RPh, MBA, and Michael B. Atkins, MD, prepared useful Practice Aids pertaining to cancer immunotherapies for this CME/MOC/CE/CPE activity titled "Incorporating Cancer Immunotherapies Into the Oncology Treatment Arsenal in Managed Care Settings: Navigating the Complexities of Value Assessment & Cost Optimization in the Era of Immuno-Oncology." For the full presentation, monograph, complete CME/MOC/CE/CPE information, and to apply for credit, please visit us at http://bit.ly/2Er15gR. CME/MOC/CE/CPE credit will be available until December 23, 2019.Incorporating Cancer Immunotherapies Into the Oncology Treatment Arsenal in M...

Incorporating Cancer Immunotherapies Into the Oncology Treatment Arsenal in M...PVI, PeerView Institute for Medical Education
Evan J. Lipson, MD, Andrew Stolbach, MD, MPH, and Trish Brothers, BSN, RN, OCN®, prepared useful practice aids pertaining to oncologic emergencies for this CME/MOC/CNE/CPE activity titled "Urgent Care of Patients Receiving Cancer Immunotherapy: Recognition and Management of Immune-Mediated Adverse Reactions in the ED." For the full presentation, monograph, complete CME/MOC/CNE/CPE information, and to apply for credit, please visit us at http://bit.ly/2TGpnYl. CME/MOC/CNE/CPE credit will be available until February 21, 2020.Urgent Care of Patients Receiving Cancer Immunotherapy: Recognition and Manag...

Urgent Care of Patients Receiving Cancer Immunotherapy: Recognition and Manag...PVI, PeerView Institute for Medical Education
Empfohlen
James T. Kenney, RPh, MBA, and Michael B. Atkins, MD, prepared useful Practice Aids pertaining to cancer immunotherapies for this CME/MOC/CE/CPE activity titled "Incorporating Cancer Immunotherapies Into the Oncology Treatment Arsenal in Managed Care Settings: Navigating the Complexities of Value Assessment & Cost Optimization in the Era of Immuno-Oncology." For the full presentation, monograph, complete CME/MOC/CE/CPE information, and to apply for credit, please visit us at http://bit.ly/2Er15gR. CME/MOC/CE/CPE credit will be available until December 23, 2019.Incorporating Cancer Immunotherapies Into the Oncology Treatment Arsenal in M...

Incorporating Cancer Immunotherapies Into the Oncology Treatment Arsenal in M...PVI, PeerView Institute for Medical Education
Evan J. Lipson, MD, Andrew Stolbach, MD, MPH, and Trish Brothers, BSN, RN, OCN®, prepared useful practice aids pertaining to oncologic emergencies for this CME/MOC/CNE/CPE activity titled "Urgent Care of Patients Receiving Cancer Immunotherapy: Recognition and Management of Immune-Mediated Adverse Reactions in the ED." For the full presentation, monograph, complete CME/MOC/CNE/CPE information, and to apply for credit, please visit us at http://bit.ly/2TGpnYl. CME/MOC/CNE/CPE credit will be available until February 21, 2020.Urgent Care of Patients Receiving Cancer Immunotherapy: Recognition and Manag...

Urgent Care of Patients Receiving Cancer Immunotherapy: Recognition and Manag...PVI, PeerView Institute for Medical Education
A slide series to learn and appreciate the importance and the potential of Personalized/Individualized Genomic Medicine. It briefly goes through the idea of biotechnology and the advancements we have made in biology and technology. A series of applications for genomic medicine is then explored, not failing to mention the challenges we have to overcome as well, for the next medical revolution.
A case for personalized medicine is presented.The case for Genomic Medicine, (Personalized, Individualized Medicine). Medic...

The case for Genomic Medicine, (Personalized, Individualized Medicine). Medic...Mathura Shanmugasundaram PhD
Arjun Balar, MD, and Petros Grivas, MD, PhD, prepared useful practice aids pertaining to bladder cancer management for this CME activity titled "Keeping Pace With Immunotherapy Advances in Bladder Cancer: Tools for Winning the Race and Optimizing Patient Outcomes." For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2GpacAq. CME credit will be available until December 30, 2019.Keeping Pace With Immunotherapy Advances in Bladder Cancer: Tools for Winning...

Keeping Pace With Immunotherapy Advances in Bladder Cancer: Tools for Winning...PVI, PeerView Institute for Medical Education
Roy H. Decker, MD, PhD, and Sarah B. Goldberg, MD, MPH, prepared useful practice aids pertaining to lung cancer for this CME activity titled "The Era of Immunotherapy in Stage III NSCLC: Exploring the Evidence and Practicalities of Integrating Checkpoint Inhibition Into the Multimodal Treatment Arsenal." For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2PU3iaZ. CME credit will be available until December 6, 2019.The Era of Immunotherapy in Stage III NSCLC: Exploring the Evidence and Pract...

The Era of Immunotherapy in Stage III NSCLC: Exploring the Evidence and Pract...PVI, PeerView Institute for Medical Education
Weitere ähnliche Inhalte
Was ist angesagt?
A slide series to learn and appreciate the importance and the potential of Personalized/Individualized Genomic Medicine. It briefly goes through the idea of biotechnology and the advancements we have made in biology and technology. A series of applications for genomic medicine is then explored, not failing to mention the challenges we have to overcome as well, for the next medical revolution.
A case for personalized medicine is presented.The case for Genomic Medicine, (Personalized, Individualized Medicine). Medic...

The case for Genomic Medicine, (Personalized, Individualized Medicine). Medic...Mathura Shanmugasundaram PhD
Arjun Balar, MD, and Petros Grivas, MD, PhD, prepared useful practice aids pertaining to bladder cancer management for this CME activity titled "Keeping Pace With Immunotherapy Advances in Bladder Cancer: Tools for Winning the Race and Optimizing Patient Outcomes." For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2GpacAq. CME credit will be available until December 30, 2019.Keeping Pace With Immunotherapy Advances in Bladder Cancer: Tools for Winning...

Keeping Pace With Immunotherapy Advances in Bladder Cancer: Tools for Winning...PVI, PeerView Institute for Medical Education
Was ist angesagt? (19)
The case for Genomic Medicine, (Personalized, Individualized Medicine). Medic...

The case for Genomic Medicine, (Personalized, Individualized Medicine). Medic...
Keeping Pace With Immunotherapy Advances in Bladder Cancer: Tools for Winning...

Keeping Pace With Immunotherapy Advances in Bladder Cancer: Tools for Winning...
Dr. Theoklis Zaoutis - Antimicrobial Use and Stewardship in the Pediatric Out...

Dr. Theoklis Zaoutis - Antimicrobial Use and Stewardship in the Pediatric Out...
1. Rational use of antiobiotics and antibiotics resistance

1. Rational use of antiobiotics and antibiotics resistance
Immunological Checkpoints and Cancer Immunotherapy

Immunological Checkpoints and Cancer Immunotherapy
Personalized medicine - Mathura Shanmugasundaram PhD

Personalized medicine - Mathura Shanmugasundaram PhD
Pharmacology for Physiotherapy Book By Padmaja Udaykumar Second Edition.

Pharmacology for Physiotherapy Book By Padmaja Udaykumar Second Edition.
The immunotherapy of cancer: past, present & the next frontier

The immunotherapy of cancer: past, present & the next frontier
Ähnlich wie Emergency Medicine and Immuno-Oncology Intersect: Recognizing and Managing Cancer Immunotherapy–Related Adverse Effects in the Emergency Department
Roy H. Decker, MD, PhD, and Sarah B. Goldberg, MD, MPH, prepared useful practice aids pertaining to lung cancer for this CME activity titled "The Era of Immunotherapy in Stage III NSCLC: Exploring the Evidence and Practicalities of Integrating Checkpoint Inhibition Into the Multimodal Treatment Arsenal." For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2PU3iaZ. CME credit will be available until December 6, 2019.The Era of Immunotherapy in Stage III NSCLC: Exploring the Evidence and Pract...

The Era of Immunotherapy in Stage III NSCLC: Exploring the Evidence and Pract...PVI, PeerView Institute for Medical Education
Edward B. Garon, MD, MS, Jamie E. Chaft, MD, and Matthew D. Hellmann, MD, prepared useful Practice Aids pertaining to lung cancer management for this CME/MOC/CE activity titled "Improving Patient Outcomes With Cancer Immunotherapies Throughout the Lung Cancer Continuum: State of the Science and Implications for Practice." For the full presentation, monograph, complete CME/MOC/CE information, and to apply for credit, please visit us at http://bit.ly/2ATq0qp. CME/MOC/CE credit will be available until November 21, 2019.Improving Patient Outcomes With Cancer Immunotherapies Throughout the Lung Ca...

Improving Patient Outcomes With Cancer Immunotherapies Throughout the Lung Ca...PVI, PeerView Institute for Medical Education
Chair, David M. O'Malley, MD, Ana Oaknin, MD, PhD, and Matthew A. Powell, MD, prepared useful Practice Aids pertaining to endometrial cancer for this CME/MOC/AAPA activity titled “Endometrial Cancer Care in the Age of Immunotherapy: Translating Clinical Evidence Into Meaningful Improvements in Patient Outcomes Across the Disease Continuum.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/AAPA information, and to apply for credit, please visit us at https://bit.ly/40bmalK. CME/MOC/AAPA credit will be available until July 3, 2024.Endometrial Cancer Care in the Age of Immunotherapy: Translating Clinical Evi...

Endometrial Cancer Care in the Age of Immunotherapy: Translating Clinical Evi...PVI, PeerView Institute for Medical Education
Co-Chairs, Nasser Altorki, MD, and Jonathan D. Spicer, MD, PhD, FRCSC, prepared useful Practice Aids pertaining to NSCLC for this CME/MOC activity titled “Can the Addition of Immunotherapy to Multimodal Management of Stage I-III NSCLC Help Break the Stalled Cycle of Poor Outcomes?” For the full presentation, downloadable Practice Aids, and complete CME/MOC information, and to apply for credit, please visit us at https://bit.ly/3m1OV2m. CME/MOC credit will be available until February 27, 2023.Can the Addition of Immunotherapy to Multimodal Management of Stage I-III NSC...

Can the Addition of Immunotherapy to Multimodal Management of Stage I-III NSC...PVI, PeerView Institute for Medical Education
Chair and Presenters Sumanta Kumar Pal, MD, FASCO, Prof. Laurence Albiges, MD, PhD, and David F. McDermott, MD, prepared useful Practice Aids pertaining to renal cell carcinoma for this CME/MOC/AAPA activity titled “Leveling Up Our RCC Care Strategy: Real-World Translation of Key Evidence Across Treatment Settings.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/AAPA information, and to apply for credit, please visit us at https://bit.ly/3LtPuyF. CME/MOC/AAPA credit will be available until December 10, 2024.Leveling Up Our RCC Care Strategy: Real-World Translation of Key Evidence Acr...

Leveling Up Our RCC Care Strategy: Real-World Translation of Key Evidence Acr...PVI, PeerView Institute for Medical Education
William F. C. Rigby, MD, and Gregg J. Silverman, MD, prepared useful Practice Aids pertaining to rheumatoid arthritis for this CME activity titled "Improving Outcomes for Patients With Refractory Rheumatoid Arthritis: The Role of JAK Inhibitors." For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2MLFVtI. CME credit will be available until October 8, 2019.Improving Outcomes for Patients With Refractory Rheumatoid Arthritis: The Rol...

Improving Outcomes for Patients With Refractory Rheumatoid Arthritis: The Rol...PVI, PeerView Institute for Medical Education
Brendon Stiles, MD, Jamie E. Chaft, MD, and David H. Harpole Jr., MD, prepared useful Practice Aids pertaining to immunotherapy in earlier stages of lung cancer for this CME/MOC activity titled, "Immunotherapy as a Component of Multimodal Therapy in Locally Advanced and Earlier Stages of Lung Cancer: Rationale, Evidence, and Implications for the Multidisciplinary Team." For the full presentation, complete CME/MOC information, and to apply for credit, please visit us at https://bit.ly/2UnPFkF. CME/MOC credit will be available until June 24, 2021.Immunotherapy as a Component of Multimodal Therapy in Locally Advanced and Ea...

Immunotherapy as a Component of Multimodal Therapy in Locally Advanced and Ea...PVI, PeerView Institute for Medical Education
Chair, Jamie E. Chaft, MD, and Jonathan D. Spicer, MD, PhD, FRCSC, prepared useful Practice Aids pertaining to NSCLC for this CME/MOC/CC/NCPD activity titled “Marking New Milestones With Immunotherapy in Locally Advanced and Early Lung Cancer: Latest Data Informing Best Practices for Multimodal Management of Stage I-III NSCLC.” For the full presentation, downloadable Practice Aids and complete CME/MOC/CC/NCPD information, and to apply for credit, please visit us at https://bit.ly/3fcc3qs. CME/MOC/CC/NCPD credit will be available until July 11, 2022.
Marking New Milestones With Immunotherapy in Locally Advanced and Early Lung ...

Marking New Milestones With Immunotherapy in Locally Advanced and Early Lung ...PVI, PeerView Institute for Medical Education
Brendon Stiles, MD, prepared useful practice aids pertaining to immunotherapy in earlier stages of lung cancer for this CME/MOC activity titled, "Chair's Take on Immunotherapy as a Component of Multimodal Therapy in Locally Advanced and Earlier Stages of Lung Cancer: Rationale, Evidence, and Implications for the Multidisciplinary Team." For the full presentation, monograph, complete CME/MOC information, and to apply for credit, please visit us at https://bit.ly/3dtEC0y. CME/MOC credit will be available until June 9, 2021.Chair's Take on Immunotherapy as a Component of Multimodal Therapy in Locally...

Chair's Take on Immunotherapy as a Component of Multimodal Therapy in Locally...PVI, PeerView Institute for Medical Education
Ähnlich wie Emergency Medicine and Immuno-Oncology Intersect: Recognizing and Managing Cancer Immunotherapy–Related Adverse Effects in the Emergency Department (20)
PD1PDL1 Pathway and its inhibitors for slideshare.pptx

PD1PDL1 Pathway and its inhibitors for slideshare.pptx
The Era of Immunotherapy in Stage III NSCLC: Exploring the Evidence and Pract...

The Era of Immunotherapy in Stage III NSCLC: Exploring the Evidence and Pract...
ASCO Guideline managmente of immune related advers events

ASCO Guideline managmente of immune related advers events
Improving Patient Outcomes With Cancer Immunotherapies Throughout the Lung Ca...

Improving Patient Outcomes With Cancer Immunotherapies Throughout the Lung Ca...
Brayer j.-l.-et-al.-2015-acute-myeloid-leukemia-and-myelodysplastic-syndrome

Brayer j.-l.-et-al.-2015-acute-myeloid-leukemia-and-myelodysplastic-syndrome
Webinar: Special Considerations for Managing Immuno-Oncology Studies: A New P...

Webinar: Special Considerations for Managing Immuno-Oncology Studies: A New P...
Endometrial Cancer Care in the Age of Immunotherapy: Translating Clinical Evi...

Endometrial Cancer Care in the Age of Immunotherapy: Translating Clinical Evi...
Can the Addition of Immunotherapy to Multimodal Management of Stage I-III NSC...

Can the Addition of Immunotherapy to Multimodal Management of Stage I-III NSC...
Leveling Up Our RCC Care Strategy: Real-World Translation of Key Evidence Acr...

Leveling Up Our RCC Care Strategy: Real-World Translation of Key Evidence Acr...
Improving Outcomes for Patients With Refractory Rheumatoid Arthritis: The Rol...

Improving Outcomes for Patients With Refractory Rheumatoid Arthritis: The Rol...
NON-INVASIVE BIOMARKERS FOR MONITORING THE IMMUNOTHERAPEUTIC RESPONSE TO CANCER 

NON-INVASIVE BIOMARKERS FOR MONITORING THE IMMUNOTHERAPEUTIC RESPONSE TO CANCER
Immunotherapy as a Component of Multimodal Therapy in Locally Advanced and Ea...

Immunotherapy as a Component of Multimodal Therapy in Locally Advanced and Ea...
Marking New Milestones With Immunotherapy in Locally Advanced and Early Lung ...

Marking New Milestones With Immunotherapy in Locally Advanced and Early Lung ...
Chair's Take on Immunotherapy as a Component of Multimodal Therapy in Locally...

Chair's Take on Immunotherapy as a Component of Multimodal Therapy in Locally...
Mehr von PVI, PeerView Institute for Medical Education
Chair Jamie Carroll, APRN, CNP, MSN, discusses breast cancer in this NCPD/ILNA/AAPA activity titled “Nurses at the Forefront of Maximizing the Potential of TROP2-Targeted Therapy in TNBC and HR+, HER2- Breast Cancer: Best Practices for Adverse Event Management and Patient Education.” For the full presentation, downloadable Practice Aids, and complete NCPD/ILNA/AAPA information, and to apply for credit, please visit us at https://bit.ly/3SdnvWt. NCPD/ILNA/AAPA credit will be available until May 8, 2025.Nurses at the Forefront of Maximizing the Potential of TROP2-Targeted Therapy...

Nurses at the Forefront of Maximizing the Potential of TROP2-Targeted Therapy...PVI, PeerView Institute for Medical Education
Chair Jonathan A. Bernstein, MD, discusses chronic spontaneous urticaria in this CME activity titled “BTK Inhibition Transforming the Landscape of Chronic Spontaneous Urticaria Treatment.” For the full presentation, downloadable Practice Aids, and complete CME information, and to apply for credit, please visit us at https://bit.ly/3P0cnvi. CME credit will be available until May 6, 2025.BTK Inhibition Transforming the Landscape of Chronic Spontaneous Urticaria Tr...

BTK Inhibition Transforming the Landscape of Chronic Spontaneous Urticaria Tr...PVI, PeerView Institute for Medical Education
Co-Chairs Milind Desai, MD, MBA, FACC, FAHA, FESC, and Andrew Willeford, PharmD, PhD, BCCP, prepared useful Practice Aids pertaining to hypertrophic cardiomyopathy for this CME/MOC/CPE/AAPA/IPCE activity titled “Adapting Managed Care Strategies in the Era of Precision Medicine for Hypertrophic Cardiomyopathy.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/CPE/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/4bb7eKc. CME/MOC/CPE/AAPA/IPCE credit will be available until May 16, 2025.Adapting Managed Care Strategies in the Era of Precision Medicine for Hypertr...

Adapting Managed Care Strategies in the Era of Precision Medicine for Hypertr...PVI, PeerView Institute for Medical Education
Co-Chairs Milind Desai, MD, MBA, FACC, FAHA, FESC, and Andrew Willeford, PharmD, PhD, BCCP, discuss hypertrophic cardiomyopathy in this CME/MOC/CPE/AAPA/IPCE activity titled “Adapting Managed Care Strategies in the Era of Precision Medicine for Hypertrophic Cardiomyopathy.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/CPE/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/4bb7eKc. CME/MOC/CPE/AAPA/IPCE credit will be available until May 16, 2025.Adapting Managed Care Strategies in the Era of Precision Medicine for Hypertr...

Adapting Managed Care Strategies in the Era of Precision Medicine for Hypertr...PVI, PeerView Institute for Medical Education
Chair A. Michael Lincoff, MD, discusses obesity in this CME activity titled “Exploring the Evidence: Improving Cardiovascular Outcomes and the Role of Weight Loss Pharmacotherapy.” For the full presentation, downloadable Practice Aids, and complete CME information, and to apply for credit, please visit us at https://bit.ly/3KAO98K. CME credit will be available until April 25, 2025.Exploring the Evidence: Improving Cardiovascular Outcomes and the Role of Wei...

Exploring the Evidence: Improving Cardiovascular Outcomes and the Role of Wei...PVI, PeerView Institute for Medical Education
Co-Chairs and Planners Saakshi Khattri, MBBS, MD, FAAD, FACR, Marla Dubinsky, MD, Emma Guttman-Yassky, MD, PhD, and Alexis Ogdie, MD, MSCE, discuss immune-mediated inflammatory diseases in this CME/AAPA activity titled “Interdisciplinary Approaches to Management of Immune-Mediated Inflammatory Diseases: Addressing Shared Pathophysiology With JAK Inhibitors.” For the full presentation, downloadable Practice Aids, and complete CME/AAPA information, and to apply for credit, please visit us at https://bit.ly/3JhsIZ7. CME/AAPA credit will be available until April 24, 2025.Interdisciplinary Approaches to Management of Immune-Mediated Inflammatory Di...

Interdisciplinary Approaches to Management of Immune-Mediated Inflammatory Di...PVI, PeerView Institute for Medical Education
Co-Chairs Alicia K. Morgans, MD, MPH, and Neal D. Shore, MD, FACS, discuss prostate cancer in this CME/MOC/NCPD/CPE/AAPA/IPCE activity titled “Treatment Advances and Individualized Therapeutic Strategies in Prostate Cancer: Expert Insights on Key Evidence, Practical Tips for Personalized Therapy, and Clinical Integration Approaches.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/CPE/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3SQrJ6G. CME/MOC/NCPD/CPE/AAPA/IPCE credit will be available until April 24, 2025.Treatment Advances and Individualized Therapeutic Strategies in Prostate Canc...

Treatment Advances and Individualized Therapeutic Strategies in Prostate Canc...PVI, PeerView Institute for Medical Education
Co-Chairs Prof. Nicolas Girard, MD, PhD, and Aaron Lisberg, MD, discuss NSCLC in this CME/MOC/NCPD/AAPA/IPCE activity titled “Charting a New Path to Better Outcomes With TROP2-Targeting ADCs in Lung Cancer: Unveiling Potential, Shaping Tomorrow.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3RmX3dU. CME/MOC/NCPD/AAPA/IPCE credit will be available until April 24, 2025.Charting a New Path to Better Outcomes With TROP2-Targeting ADCs in Lung Canc...

Charting a New Path to Better Outcomes With TROP2-Targeting ADCs in Lung Canc...PVI, PeerView Institute for Medical Education
Co-Chairs, Joseph K. Han, MD, and Seth J. Isaacs, MD, prepared useful Practice Aids pertaining to chronic rhinosinusitis with nasal polyps for this CME/MOC/CC/AAPA/IPCE activity titled “Biologics in CRSwNP: Putting a Paradigm Shift Into Practice.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/CC/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3Tq6n1G. CME/MOC/CC/AAPA/IPCE credit will be available until May 6, 2025.Biologics in CRSwNP: Putting a Paradigm Shift Into Practice

Biologics in CRSwNP: Putting a Paradigm Shift Into PracticePVI, PeerView Institute for Medical Education
Co-Chairs, Joseph K. Han, MD, and Seth J. Isaacs, MD, discuss chronic rhinosinusitis with nasal polyps in this CME/MOC/CC/AAPA/IPCE activity titled “Biologics in CRSwNP: Putting a Paradigm Shift Into Practice.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/CC/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3Tq6n1G. CME/MOC/CC/AAPA/IPCE credit will be available until May 6, 2025.Biologics in CRSwNP: Putting a Paradigm Shift Into Practice

Biologics in CRSwNP: Putting a Paradigm Shift Into PracticePVI, PeerView Institute for Medical Education
Co-Chairs, R. Donald Harvey, PharmD, BCOP, FCCP, FHOPA, FASCO, Zahra Mahmoudjafari, PharmD, MBA, BCOP, FHOPA, and James Davis, PharmD, BCOP, discuss multiple myeloma in this CME/CPE/IPCE activity titled “Prescriptions for Successful Myeloma Care: Pharmacy Strategies for Delivering Effective Therapy With Antibody Platforms.” For the full presentation, downloadable Practice Aids, and complete CME/CPE/IPCE information, and to apply for credit, please visit us at https://bit.ly/4aa0iMX. CME/CPE/IPCE credit will be available until May 2, 2025.Prescriptions for Successful Myeloma Care: Pharmacy Strategies for Delivering...

Prescriptions for Successful Myeloma Care: Pharmacy Strategies for Delivering...PVI, PeerView Institute for Medical Education
Co-Chairs, Carlos G. Romo, MD, and Aimee Sato, MD, discuss Neurofibromatosis in this CME/MOC activity titled “Precision & Progress Against NF1: Solutions for Better Outcomes With MEKi & Multimodal Care for NF1 pNF and Other Tumors.” For the full presentation, downloadable Practice Aids, and complete CME/MOC information, and to apply for credit, please visit us at https://bit.ly/3SZRz8p. CME/MOC credit will be available until May 2, 2025.Precision & Progress Against NF1: Solutions for Better Outcomes With MEKi & M...

Precision & Progress Against NF1: Solutions for Better Outcomes With MEKi & M...PVI, PeerView Institute for Medical Education
Chair and Presenters Kathleen N. Moore, MD, MS, Floor J. Backes, MD, and Bhavana Pothuri, MD, MS, prepared useful Practice Aids pertaining to endometrial cancer for this CME/MOC/NCPD/AAPA/IPCE activity titled “Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Potential of Immunotherapy, ADCs, PARP Inhibitors, and Other Emerging Treatment Strategies.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3SjJyuH. CME/MOC/NCPD/AAPA/IPCE credit will be available until April 17, 2025.Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...

Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...PVI, PeerView Institute for Medical Education
Chair and Presenters Kathleen N. Moore, MD, MS, Floor J. Backes, MD, and Bhavana Pothuri, MD, MS, discuss endometrial cancer in this CME/MOC/NCPD/AAPA/IPCE activity titled “Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Potential of Immunotherapy, ADCs, PARP Inhibitors, and Other Emerging Treatment Strategies.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3SjJyuH. CME/MOC/NCPD/AAPA/IPCE credit will be available until April 17, 2025.Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...

Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...PVI, PeerView Institute for Medical Education
Chair and Presenters Bradley J. Monk, MD, FACS, FACOG, Kathleen N. Moore, MD, MS, and Ana Oaknin, MD, PhD, discuss gynecologic cancers in this CME/MOC/NCPD/AAPA/IPCE activity titled “Advancing ADCs in Gynecologic Cancers: Expert Insights on Recent Evidence, Implementation Strategies, and Patient Care.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/4a56tly. CME/MOC/NCPD/AAPA/IPCE credit will be available until April 16, 2025.Advancing ADCs in Gynecologic Cancers: Expert Insights on Recent Evidence, Im...

Advancing ADCs in Gynecologic Cancers: Expert Insights on Recent Evidence, Im...PVI, PeerView Institute for Medical Education
Chair Lecia V. Sequist, MD, MPH, and Patrick Nana-Sinkam, MD, FCCP, prepared useful Practice Aids pertaining to lung cancer for this CME/MOC/AAPA/IPCE activity titled “Screening and Early Intervention as the Keys to Success in Lung Cancer: A Practical Approach to Implementing Lung Cancer Screening for High-Risk Individuals.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/46VvwoP. CME/MOC/AAPA/IPCE credit will be available until April 16, 2025.Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...

Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...PVI, PeerView Institute for Medical Education
Chair Lecia V. Sequist, MD, MPH, and Patrick Nana-Sinkam, MD, FCCP, discuss lung cancer screening in this CME/MOC/AAPA/IPCE activity titled “Screening and Early Intervention as the Keys to Success in Lung Cancer: A Practical Approach to Implementing Lung Cancer Screening for High-Risk Individuals.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/46VvwoP. CME/MOC/AAPA/IPCE credit will be available until April 16, 2025.Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...

Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...PVI, PeerView Institute for Medical Education
Chair and Presenter, Olalekan Oluwole, MBBS, MPH, Veronika Bachanova, MD, PhD, and David L. Porter, MD, prepared useful Practice Aids pertaining to CAR-T therapy for this CME/NCPD activity titled “Democratizing the CAR-T Experience: The Principles and Practice of Outpatient Cellular Therapy.” For the full presentation, downloadable Practice Aids, and complete CME/NCPD information, and to apply for credit, please visit us at https://bit.ly/3TfIABM. CME/NCPD credit will be available until April 15, 2025.Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...

Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...PVI, PeerView Institute for Medical Education
Chair and Presenter, Olalekan Oluwole, MBBS, MPH, Veronika Bachanova, MD, PhD, and David L. Porter, MD, discuss CAR-T therapy in this CME/NCPD activity titled “Democratizing the CAR-T Experience: The Principles and Practice of Outpatient Cellular Therapy.” For the full presentation, downloadable Practice Aids, and complete CME/NCPD information, and to apply for credit, please visit us at https://bit.ly/3TfIABM. CME/NCPD credit will be available until April 15, 2025.Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...

Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...PVI, PeerView Institute for Medical Education
Co-Chairs Lipika Goyal, MD, MPhil, and Riad Salem, MD, MBA, discuss HCC in this CME activity titled “The Convergence of Interventional Radiologists and Oncologists in HCC: Shared Decision-Making and Care Coordination at the Center of Personalized Care Across the Disease Continuum.” For the full presentation, downloadable Practice Aids, and complete CME information, and to apply for credit, please visit us at https://bit.ly/48BAasz. CME credit will be available until April 26, 2025.The Convergence of Interventional Radiologists and Oncologists in HCC: Shared...

The Convergence of Interventional Radiologists and Oncologists in HCC: Shared...PVI, PeerView Institute for Medical Education
Mehr von PVI, PeerView Institute for Medical Education (20)
Nurses at the Forefront of Maximizing the Potential of TROP2-Targeted Therapy...

Nurses at the Forefront of Maximizing the Potential of TROP2-Targeted Therapy...
BTK Inhibition Transforming the Landscape of Chronic Spontaneous Urticaria Tr...

BTK Inhibition Transforming the Landscape of Chronic Spontaneous Urticaria Tr...
Adapting Managed Care Strategies in the Era of Precision Medicine for Hypertr...

Adapting Managed Care Strategies in the Era of Precision Medicine for Hypertr...
Adapting Managed Care Strategies in the Era of Precision Medicine for Hypertr...

Adapting Managed Care Strategies in the Era of Precision Medicine for Hypertr...
Exploring the Evidence: Improving Cardiovascular Outcomes and the Role of Wei...

Exploring the Evidence: Improving Cardiovascular Outcomes and the Role of Wei...
Interdisciplinary Approaches to Management of Immune-Mediated Inflammatory Di...

Interdisciplinary Approaches to Management of Immune-Mediated Inflammatory Di...
Treatment Advances and Individualized Therapeutic Strategies in Prostate Canc...

Treatment Advances and Individualized Therapeutic Strategies in Prostate Canc...
Charting a New Path to Better Outcomes With TROP2-Targeting ADCs in Lung Canc...

Charting a New Path to Better Outcomes With TROP2-Targeting ADCs in Lung Canc...
Biologics in CRSwNP: Putting a Paradigm Shift Into Practice

Biologics in CRSwNP: Putting a Paradigm Shift Into Practice
Biologics in CRSwNP: Putting a Paradigm Shift Into Practice

Biologics in CRSwNP: Putting a Paradigm Shift Into Practice
Prescriptions for Successful Myeloma Care: Pharmacy Strategies for Delivering...

Prescriptions for Successful Myeloma Care: Pharmacy Strategies for Delivering...
Precision & Progress Against NF1: Solutions for Better Outcomes With MEKi & M...

Precision & Progress Against NF1: Solutions for Better Outcomes With MEKi & M...
Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...

Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...
Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...

Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...
Advancing ADCs in Gynecologic Cancers: Expert Insights on Recent Evidence, Im...

Advancing ADCs in Gynecologic Cancers: Expert Insights on Recent Evidence, Im...
Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...

Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...
Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...

Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...
Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...

Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...
Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...

Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...
The Convergence of Interventional Radiologists and Oncologists in HCC: Shared...

The Convergence of Interventional Radiologists and Oncologists in HCC: Shared...
Kürzlich hochgeladen
Models Call Girls In Hyderabad 9630942363 Hyderabad Call Girl & Hyderabad Escort ServiceModels Call Girls In Hyderabad 9630942363 Hyderabad Call Girl & Hyderabad Esc...

Models Call Girls In Hyderabad 9630942363 Hyderabad Call Girl & Hyderabad Esc...GENUINE ESCORT AGENCY
Kürzlich hochgeladen (20)
Coimbatore Call Girls in Coimbatore 7427069034 genuine Escort Service Girl 10...

Coimbatore Call Girls in Coimbatore 7427069034 genuine Escort Service Girl 10...
Jogeshwari ! Call Girls Service Mumbai - 450+ Call Girl Cash Payment 90042684...

Jogeshwari ! Call Girls Service Mumbai - 450+ Call Girl Cash Payment 90042684...
Call Girls Jaipur Just Call 9521753030 Top Class Call Girl Service Available

Call Girls Jaipur Just Call 9521753030 Top Class Call Girl Service Available
Dehradun Call Girls Service {8854095900} ❤️VVIP ROCKY Call Girl in Dehradun U...

Dehradun Call Girls Service {8854095900} ❤️VVIP ROCKY Call Girl in Dehradun U...
Top Rated Hyderabad Call Girls Erragadda ⟟ 9332606886 ⟟ Call Me For Genuine ...

Top Rated Hyderabad Call Girls Erragadda ⟟ 9332606886 ⟟ Call Me For Genuine ...
Call Girl In Pune 👉 Just CALL ME: 9352988975 💋 Call Out Call Both With High p...

Call Girl In Pune 👉 Just CALL ME: 9352988975 💋 Call Out Call Both With High p...
Call Girls Mysore Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Mysore Just Call 8250077686 Top Class Call Girl Service Available
💕SONAM KUMAR💕Premium Call Girls Jaipur ↘️9257276172 ↙️One Night Stand With Lo...

💕SONAM KUMAR💕Premium Call Girls Jaipur ↘️9257276172 ↙️One Night Stand With Lo...
Premium Call Girls In Jaipur {8445551418} ❤️VVIP SEEMA Call Girl in Jaipur Ra...

Premium Call Girls In Jaipur {8445551418} ❤️VVIP SEEMA Call Girl in Jaipur Ra...
Models Call Girls In Hyderabad 9630942363 Hyderabad Call Girl & Hyderabad Esc...

Models Call Girls In Hyderabad 9630942363 Hyderabad Call Girl & Hyderabad Esc...
Top Rated Pune Call Girls (DIPAL) ⟟ 8250077686 ⟟ Call Me For Genuine Sex Serv...

Top Rated Pune Call Girls (DIPAL) ⟟ 8250077686 ⟟ Call Me For Genuine Sex Serv...
Call Girls Service Jaipur {8445551418} ❤️VVIP BHAWNA Call Girl in Jaipur Raja...

Call Girls Service Jaipur {8445551418} ❤️VVIP BHAWNA Call Girl in Jaipur Raja...
Russian Call Girls Service Jaipur {8445551418} ❤️PALLAVI VIP Jaipur Call Gir...

Russian Call Girls Service Jaipur {8445551418} ❤️PALLAVI VIP Jaipur Call Gir...
Call Girls Amritsar Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Amritsar Just Call 8250077686 Top Class Call Girl Service Available
Top Rated Call Girls Kerala ☎ 8250092165👄 Delivery in 20 Mins Near Me

Top Rated Call Girls Kerala ☎ 8250092165👄 Delivery in 20 Mins Near Me
Andheri East ) Call Girls in Mumbai Phone No 9004268417 Elite Escort Service ...

Andheri East ) Call Girls in Mumbai Phone No 9004268417 Elite Escort Service ...
Call Girls Service Jaipur {9521753030 } ❤️VVIP BHAWNA Call Girl in Jaipur Raj...

Call Girls Service Jaipur {9521753030 } ❤️VVIP BHAWNA Call Girl in Jaipur Raj...
Premium Bangalore Call Girls Jigani Dail 6378878445 Escort Service For Hot Ma...

Premium Bangalore Call Girls Jigani Dail 6378878445 Escort Service For Hot Ma...
Independent Call Girls Service Mohali Sector 116 | 6367187148 | Call Girl Ser...

Independent Call Girls Service Mohali Sector 116 | 6367187148 | Call Girl Ser...
Emergency Medicine and Immuno-Oncology Intersect: Recognizing and Managing Cancer Immunotherapy–Related Adverse Effects in the Emergency Department
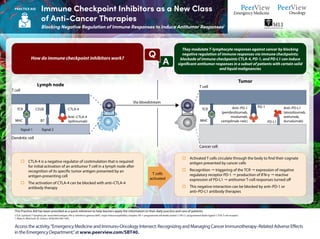
- 1. CTLA: cytotoxic T-lymphocyte–associated antigen; IFN-γ: interferon gamma; MHC: major histocompatibility complex; PD-1: programmed cell death protein 1; PD-L1: programmed death-ligand 1; TCR: T-cell receptor. 1. Ribas A, Wolchock JD. Science. 2018;359:1350-1355. This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. They modulate T-lymphocyte responses against cancer by blocking negative regulation of immune responses via immune checkpoints; blockade of immune checkpoints CTLA-4, PD-1, and PD-L1 can induce significant antitumor responses in a subset of patients with certain solid and liquid malignancies How do immune checkpoint inhibitors work? o Activated T cells circulate through the body to find their cognate antigen presented by cancer cells o Recognition " triggering of the TCR " expression of negative regulatory receptor PD-1 " production of IFN-γ " reactive expression of PD-L1 " antitumor T-cell responses turned off o This negative interaction can be blocked by anti–PD-1 or anti–PD-L1 antibody therapies o CTLA-4 is a negative regulator of costimulation that is required for initial activation of an antitumor T cell in a lymph node after recognition of its specific tumor antigen presented by an antigen-presenting cell o The activation of CTLA-4 can be blocked with anti–CTLA-4 antibody therapy T cells activated A Q Lymph node T cell Dendritic cell TCR MHC Signal 1 Signal 2 CD28 B7 CTLA-4 Anti–CTLA-4 (ipilimumab) Via bloodstream Tumor T cell Cancer cell TCR MHC Anti–PD-L1 (atezolizumab, avelumab, durvalumab) Anti–PD-1 (pembrolizumab, nivolumab, cemiplimab-rwlc) PD-L1 PD-1 Access the activity,“Emergency Medicine and Immuno-Oncology Intersect: Recognizing and Managing Cancer Immunotherapy–Related Adverse Effects in the Emergency Department,”at www.peerview.com/SBT40. Immune Checkpoint Inhibitors as a New Class of Anti-Cancer Therapies Blocking Negative Regulation of Immune Responses to Induce Antitumor Responses1 PRACTICE AID
- 2. CRC: colorectal cancer; CSCC: cutaneous squamous-cell carcinoma; CTLA: cytotoxic T-lymphocyte–associated antigen; dMMR: mismatch repair deficient; HCC: hepatocellular carcinoma; HNSCC: head and neck squamous cell carcinoma; MSI-H: microsatellite instability high; PD-1: programmed death 1; PD-L1: programmed death-ligand 1; RCC: renal cell carcinoma. 1. https://www.fda.gov/drugs/informationondrugs/approveddrugs/ucm279174.htm. Accessed August 21, 2018. This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. Access the activity,“Emergency Medicine and Immuno-Oncology Intersect: Recognizing and Managing Cancer Immunotherapy–Related Adverse Effects in the Emergency Department,”at www.peerview.com/SBT40. Immune Checkpoint Inhibitors as a New Class of Anti-Cancer Therapies Timeline of FDA Approvals and Current Indications1 PRACTICE AID September Pembrolizumab (melanoma) December Nivolumab (melanoma) March Ipilimumab (melanoma) 2011 2014 2015 2016 2017 2018 May Atezolizumab (bladder) August Pembrolizumab (HNSCC) October Pembrolizumab (PD-L1+ NSCLC, first line) Atezolizumab (NSCLC, second line) November Nivolumab (HNSCC) March Nivolumab (squamous NSCLC, second line) October Nivolumab (nonsquamous NSCLC, second line) Pembrolizumab (PD-L1+ NSCLC, second line) Nivolumab + ipilimumab (melanoma, first line) Ipilimumab (melanoma, adjuvant) November Nivolumab (RCC) December Pembrolizumab (melanoma, first line) January Nivolumab (bladder) March Avelumab (Merkel cell carcinoma) May Durvalumab, avelumab, pembrolizumab (bladder) Pembrolizumab + chemotherapy (nonsquamous NSCLC, first line) Pembrolizumab (MSI-H cancers) August Nivolumab (dMMR/MSI-H CRC) September Pembrolizumab (gastric) Nivolumab (HCC) December Nivolumab (melanoma, adjuvant) February Durvalumab (stage III NSCLC) April Nivolumab + ipilimumab (RCC, first line) June Pembrolizumab (PD-L1+ cervical) July Nivolumab + ipilimumab (dMMR/MSI-H CRC) August Nivolumab (SCLC, third line) Pembrolizumab + chemotherapy (nonsquamous NSCLC, first line—full approval) September Cemiplimab (CSCC) Drug Ipilimumab Nivolumab Pembrolizumab Atezolizumab Avelumab Durvalumab Mechanism Anti–CTLA-4 Anti–PD-1 Anti–PD-L1
- 3. aTTP: acquired thrombotic thrombocytopenic purpura; irAE: immune-related adverse effect; ITP: Immune thrombocytopenia. 1. https://www.asco.org/sites/new-www.asco.org/files/content-files/practice-and-guidelines/2018-management-of-irAEs-summary.pdf. Accessed September 14, 2018. This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. Access the activity,“Emergency Medicine and Immuno-Oncology Intersect: Recognizing and Managing Cancer Immunotherapy–Related Adverse Effects in the Emergency Department,”at www.peerview.com/SBT40. Spectrum of Immune-Related Adverse Effects Associated With Immune Checkpoint Inhibitors Used to Treat Patients With Cancer1 Immunotherapies used in oncology are associated with important clinical benefits, but general immunologic enhancement induced by these agents can also lead to a unique spectrum of irAEs Pulmonary Pneumonitis Gastrointestinal Colitis Hepatitis Cardiac Myocarditis Pericarditis Arrhythmias Impaired ventricular function with heart failure and vasculitis Venous thromboembolism Musculoskeletal Inflammatory arthritis Myositis Polymyalgia-like syndrome Dermatologic Rash/Inflammatory dermatitis Bullous dermatoses Severe cutaneous adverse reactions (SCAR) Renal Nephritis Symptomatic nephritis Hematologic Autoimmune hemolytic anemia aTTP Hemolytic uremic syndrome Aplastic anemia Lymphopenia ITP Acquired hemophilia Endocrine Primary hypothyroidism Hyperthyroidism Hypophysitis Primary adrenal insufficiency Diabetes Hepatic Transaminases Hepatitis Ocular Uveitis/iritis Episcleritis Blepharitis Neurologic Neuropathy Meningitis Guillain-Barré syndrome Myasthenia gravis Encephalitis Transverse myelitis irAEs Differ from toxicities of chemotherapies and other cancer therapies Can affect any organ system, but most commonly involve the GI tract, endocrine glands, skin, and liver Can have unpredictable onset (including early or late) Can be difficult to differentiate from other causes (diagnosed by exclusion) PRACTICE AID
- 4. Access the activity,“Emergency Medicine and Immuno-Oncology Intersect: Recognizing and Managing Cancer Immunotherapy–Related Adverse Effects in the Emergency Department,”at www.peerview.com/SBT40. Answers to Common Questions Related to irAEs That Can Occur in Patients With Cancer Treated With Immune Checkpoint Inhibitors1 • Pathophysiology unknown • T-cell, antibody, and cytokine responses may be involved Why Do irAEs Occur? • No prospective trials have defined the best treatments; recommendations based on consensus opinion • Immunosuppression used to $ excess temporary inflammation • Glucocorticoids are usually the first-line immunosuppressive agent • Additional immunosuppressants can be used if glucocorticoids are not initially effective • Side effects of glucocorticoid treatment can occur, and immunosuppression # risk for opportunistic infections How Are irAEs Treated? • Can present at any time, but typically start within the first few weeks to months after treatment initiation and can occur after treatment discontinuation • Dermatologic AEs typically occur first When Do irAEs Occur? PRACTICE AID
- 5. CTLA: cytotoxic T-lymphocyte–associated antigen; irAE: immune-related adverse effect; PD-1: programmed cell death protein 1; PD-L1: programmed death-ligand 1. 1. Postow MA et al. N Engl J Med. 2018;378:158-168. This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. Access the activity,“Emergency Medicine and Immuno-Oncology Intersect: Recognizing and Managing Cancer Immunotherapy–Related Adverse Effects in the Emergency Department,”at www.peerview.com/SBT40. Answers to Common Questions Related to irAEs That Can Occur in Patients With Cancer Treated With Immune Checkpoint Inhibitors1 PRACTICE AID • Clinical outcomes are similar between patients who do and do not require immunosuppression to treat irAEs • Beneficial responses can persist despite the use of immunosuppressive agents Does Immunosuppression to Treat irAEs Affect Response to Immune Checkpoint Blockade? • irAEs associated with one type of agent (eg, anti–CTLA-4 antibodies) may not necessarily recur during subsequent treatment with another agent (eg, anti–PD-1/–PD-L1 antibodies) • The safety of retreatment likely depends on the severity of the initial irAE • Patients who have had a favorable response to immune checkpoint blockade and then discontinue treatment because of irAEs generally maintain responses; prospective data are needed to address the necessity of restarting immunotherapy Can Immunotherapy Be Safely Restarted After a Major irAE? • Patients with # risk for irAEs (eg, those with pre-existing autoimmune conditions) may still benefit from treatment • Age alone should not be used to exclude patients from treatment, since benefit appears to be similar regardless of age Can Patients at an Increased Risk of irAEs Be Safely Treated With Immune Checkpoint Blockade? • Conflicting data exist regarding whether the occurrence of irAEs is associated with # treatment efficacy • irAEs not required for treatment benefit, although certain AEs (eg, vitiligo) are linked to # treatment response Are There Associations Between irAEs and Treatment Efficacy?
- 6. CAR: chimeric antigen receptor; DDx: differential diagnosis; irAE: immune-related adverse effect; PMHx: past medical history. This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. Access the activity,“Emergency Medicine and Immuno-Oncology Intersect: Recognizing and Managing Cancer Immunotherapy–Related Adverse Effects in the Emergency Department,”at www.peerview.com/SBT40. Managing Immune-Related Adverse Effects in the Emergency Department PAST MEDICAL HISTORY o Ask about history of cancer o If positive, ask about cancer therapies received within past year – Does treatment history include cancer immunotherapy (eg, immune checkpoint inhibitors, CAR-T cell therapy)? IMMUNOTHERAPY IN PMHx " MODIFY DDx o Almost any inflammatory conditions, endocrinopathies from checkpoint inhibitors o Cytokine-release syndrome, neurotoxicity from CAR-T cell therapy irAEs in the ED Change your practice! ALWAYS CALL THE ONCOLOGIST! AVOID PREMATURE CLOSURE o Don't settle too early on any oncologic or nononcologic diagnoses o Examples Premature Closure on a Nononcology Diagnosis Your Diagnosis Sepsis Depression Benign headache Cellulitis Viral syndrome Pneumonia Real Diagnosis Cytokine-release syndrome Adrenal insufficiency Hypophysitis Immune-mediated dermatitis or radiation recall Immune-mediated hepatitis Immune-mediated pneumonitis Premature Closure on an Oncology Diagnosis Your Diagnosis Immune-mediated colitis Cytokine-release syndrome Real Diagnosis C. difficile colitis Sepsis SIMPLIFY TREATMENT DECISIONS o Immunosuppression used to reduce excessive state of temporary inflammation o Glucocorticoids should typically be used as first-line immunosuppressive therapy o Additional immunosuppressive agents can be used if glucocorticoids are not initially effective Always coordinate with oncology! PRACTICE AID
