Breast cancer written report
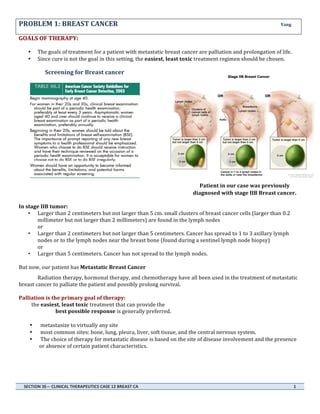
- 1. PROBLEM 1: BREAST CANCER Yang GOALS OF THERAPY: • • The goals of treatment for a patient with metastatic breast cancer are palliation and prolongation of life. Since cure is not the goal in this setting, the easiest, least toxic treatment regimen should be chosen. Screening for Breast cancer Patient in our case was previously diagnosed with stage IIB Breast cancer. In stage IIB tumor: • Larger than 2 centimeters but not larger than 5 cm. small clusters of breast cancer cells (larger than 0.2 millimeter but not larger than 2 millimeters) are found in the lymph nodes or • Larger than 2 centimeters but not larger than 5 centimeters. Cancer has spread to 1 to 3 axillary lymph nodes or to the lymph nodes near the breast bone (found during a sentinel lymph node biopsy) or • Larger than 5 centimeters. Cancer has not spread to the lymph nodes. But now, our patient has Metastatic Breast Cancer Radiation therapy, hormonal therapy, and chemotherapy have all been used in the treatment of metastatic breast cancer to palliate the patient and possibly prolong survival. Palliation is the primary goal of therapy: the easiest, least toxic treatment that can provide the best possible response is generally preferred. • metastasize to virtually any site • most common sites: bone, lung, pleura, liver, soft tissue, and the central nervous system. • The choice of therapy for metastatic disease is based on the site of disease involvement and the presence or absence of certain patient characteristics. SECTION 3E-‐– CLINICAL THERAPEUTICS CASE 12 BREAST CA 1
- 2. Treatment: I. RADIATION THERAPY: Radiation therapy is primarily used to control symptomatic disease such as bone metastases, metastatic brain lesions, and spinal cord compressions. II. HORMONAL THERAPY • goal of hormonal therapy is to reduce the stimulation of the tumor cells by estrogen. • Adjuvant hormonal therapy should be offered to any patient whose tumor overexpresses hormone receptors [either ER or progesterone (PgR)], regardless of patient age, nodal status, or menopausal status. In our case, patient had received Tamoxifen, a selective estro-‐ gen-‐receptor modulator (SERM), (adjuvant hor-‐ monal therapy most commonly used) for five years. • However, the benefits of tamoxifen must be weighed against the side effects of treatment, particularly when the drug is being used in the adjuvant setting. • The most common side effects of tamoxifen include hot flashes and vaginal discharge, but an increased risk of thromboembolic events and endometrial cancer can also occur. • Third-‐generation aromatase inhibitors have been extensively studied as first and second-‐line therapy for metastatic breast cancer. o The ATAC (Arimidex, Tamoxifen Alone or in Combination) Trialists’ Group found superior disease-‐ free survival for anastrozole as adjuvant therapy in post-‐ menopausal women with hormone-‐ sensitive disease when compared to tamoxifen or the combination of tamoxifen and anastrozole. As a result, anastrozole was granted accelerated approval as adjuvant therapy for breast cancer. • Fulvestrant, an injectable pure estrogen antagonist, has also shown activity in patients with hormone-‐ receptor-‐ positive disease progressing on hormonal therapy. • The choice of hormonal therapy is patient-‐specific and may be influenced by prior therapy in the adjuvant setting, toxicity profiles, cost, and ease of administration. *** Tamoxifen – acts like an anti-‐estrogen in breast cells, it acts like an estrogen in other tissues, like the uterus and the bones -‐ stop the growth and even shrink tumors in women with metastatic breast cancer. It can also be used to reduce the risk of developing breast cancer in women at high risk Aromatase inhibitors: cannot stop the ovaries from making estrogen, so they are only effective in women whose ovaries aren’t working (like after menopause) Fulvestrant -‐ first blocks the estrogen receptor and then also eliminates it temporarily; acts like an anti-‐estrogen throughout the body ***
- 3. Efficacy Safety Suitability Cost Anti-‐ estrogen Tamoxifen +++ selective estrogen receptor modulator or SERM ++ Disease flare, hot flashes; rare: thrombophlebitis, ocular abnormalities, endometrial cancer ++ premenopausal and postmenopausal women (and men) with ER-‐positive early-‐ stage breast cancer 1,400 Aromatase Inhibitors 3rd gen: anastrazole +++ Blocking aromatase in fat tissue that is responsible for making small amounts of estrogen in post-‐ menopausal women +++ Hot flashes, nausea, vomiting, headache, fatigue; rare: bone fractures, musculoskeletal disorders +++ No significant drug interactions initial therapy for metastatic hormone-‐sensitive breast cancer treat postmenopausal women with advanced breast cancer whose disease has worsened after treatment with tamoxifen +++ 2750 Pure Estrogen Antagonist Fulvestrant +++ +++ Hot flashes, headache, nausea, vomiting, injection site reactions ++ No significant drug interactions postmenopausal women with metastatic ER-‐positive breast cancer after treatment with other antiestrogens +++ 28,000 Median duration of response to the first attempt at hormonal manipulation is usually in the range of 9 to 12 mos. First-‐line hormonal therapy should be administered for at least 6 to 8 weeks before disease response is assessed. If a patient becomes refractory to hormonal therapy at any time, chemotherapy should be given. III. CHEMOTHERAPY: Chemotherapeutic drugs are most commonly used as palliative therapy in patients who would not be expected to respond to hormonal therapy 4 GROUPS OF CHEMOTHERAPEUTIC DRUGS 1. ALKYLATING AGENTS The major clinically useful alkylating agents have a structure containing a bis(chloroethyl)amine, ethyleneimine, or nitrosourea moiety, and they are classified in several different groups. Mechanism of Action • exert their cytotoxic effects via transfer of their alkyl groups to various cellular constituents. • Alkylations of DNA within the nucleus probably represent the major interactions that lead to cell death. • The general mechanism of action of these drugs involves intramolecular cyclization to form an ethyleneimonium ion that may directly or through formation of a carbonium ion transfer an alkyl group to a cellular constituent • a secondary mechanism that occurs with nitrosoureas involves carbamoylation of lysine residues of proteins through formation of isocyanates. SECTION 3E-‐ CLINICAL THERAPEUTICS CASE 12 BREAST CA 3
- 4. Adverse Effects • generally dose-‐related and occur primarily in rapidly growing tissues such as bone marrow, gastrointestinal tract, and reproductive system. • Nausea and vomiting can be a serious issue • potent vesicants and can damage tissues at the site of administration as well as produce systemic toxicity. • carcinogenic in nature, and there is an increased risk of secondary malignancies, especially acute myelogenous leukemia. Cyclophosphamide • is one of the most widely used alkylating agents. • One of the potential advantages: high oral bioavailability • oral and intravenous routes with equal clinical efficacy. • inactive in its parent form, and must be activated to cytotoxic forms by liver microsomal enzymes A. NITROSOUREAS • non-‐cross-‐resistant with other alkylating agents; all require biotransformation, which occurs by nonenzymatic decomposition, to metabolites with both alkylating and carbamoylating activities • highly lipid-‐soluble and are able to cross the blood-‐brain barrier B. NONCLASSIC ALKYLATING AGENTS 1. Procarbazine 2. Dacarbazine 3. Bendamustine C. PLATINUM ANALOGS Three platinum analogs are currently used in clinical practice: cisplatin, carboplatin, and oxaliplatin. Cisplatin • • is an inorganic metal complex that was initially discovered through an observation that neutral platinum complexes inhibited division and filamentous growth of Escherichia coli. MOA: kill tumor cells in all stages of the cell cycle and bind DNA through the formation of intrastrand and interstrand cross-‐links, thereby leading to inhibition of DNA synthesis and function. -‐ Cisplatin and the other platinum analogs are extensively cleared by the kidneys and excreted in the urine. As a result, dose modification is required in patients with renal dysfunction. Carboplatin • is a second-‐generation platinum analog • MOA, mechanisms of resistance, and pharmacology are identical to cisplatin. • in contrast to cisplatin, it exhibits significantly less renal toxicity and GI toxicity • Its main dose-‐limiting toxicity is myelosuppression. • It has therefore been widely used in transplant regimens to treat refractory hematologic malignancies. Oxaliplatin • • third-‐generation diaminocyclohexane platinum analog. tumors that are resistant to cisplatin or carboplatin on the basis of mismatch repair defects are not cross-‐ resistant to oxaliplatin,
- 5. 2. ANTIMETABOLITES A. ANTIFOLATES Methotrexate is a folic acid analog that binds with high affinity to the active catalytic site of dihydrofolate reductase (DHFR) à inhibition of the synthesis of tetrahydrofolate (THF) Pemetrexed Pralatrexate • SECTION 3E-‐ CLINICAL THERAPEUTICS CASE 12 BREAST CA 5
- 6. B. FLUOROPYRIMIDINES 5-‐Fluorouracil inactive in its parent form; requires activation via a complex series of enzymatic reactions to ribosyl and deoxyribosyl nucleotide metabolites; cytotoxicity of 5-‐FU is thought to be the result of combined effects on both DNA-‐ and RNA-‐mediated events. Capecitabine • C. DEOXYCYTIDINE ANALOGS Cytarabine • (ara-‐C) is an S phase-‐specific antimetabolite that is converted by deoxycytidine kinase to the 5'-‐ mononucleotide (ara-‐CMP). Ara-‐CMP is further metabolized to the diphosphate and triphosphate metabolites, and the ara-‐CTP triphosphate is felt to be the main cytotoxic metabolite. Gemcitabine D. PURINE ANTAGONISTS 6-‐Thiopurines Fludarabine Cladribine
- 7. 3. NATURAL PRODUCT CANCER CHEMOTHERAPY DRUGS A. VINCA ALKALOIDS Vinblastine • inhibition of tubulin polymerization, which disrupts assembly of microtubules, an important part of the cytoskeleton and the mitotic spindle. This inhibitory effect results in mitotic arrest in metaphase, bringing cell division to a halt, which then leads to cell death. Vincristine • While myelosuppression occurs, it is generally milder and much less significant than with vinblastine. Vinorelbine B. TAXANES & RELATED DRUGS Paclitaxel • • • drug functions as a mitotic spindle poison through high-‐affinity binding to microtubules with enhancement of tubulin polymerization. This promotion of microtubule assembly by paclitaxel occurs in the absence of microtubule-‐associated proteins and guanosine triphosphate and results in inhibition of mitosis and cell division Hypersensitivity reactions may be observed in up to 5% of patients, but the incidence is significantly reduced by premedication with dexamethasone, diphenhydramine, and an H2 blocker. Abraxane • • A novel albumin-‐bound paclitaxel formulation is approved for use in metastatic breast cancer. In contrast to paclitaxel, this formulation is not associated with hypersensitivity reactions B. EPIPODOPHYLLOTOXINS Etoposide • The main site of action is inhibition of the DNA enzyme topoisomerase II C. CAMPTOTHECINS • • inhibit the activity of topoisomerase I, the key enzyme responsible for cutting and religating single DNA strands. Inhibition of this enzyme results in DNA damage Myelosuppression and diarrhea are the two most common adverse events SECTION 3E-‐ CLINICAL THERAPEUTICS CASE 12 BREAST CA 7
- 8. 4. ANTITUMOR ANTIBIOTICS Many of these antibiotics bind to DNA through intercalation between specific bases and block the synthesis of RNA, DNA, or both; cause DNA strand scission; and interfere with cell replication. All of the anticancer antibiotics now being used in clinical practice are products of various strains of the soil microbe Streptomyces. A. ANTHRACYCLINES among the most widely used cytotoxic anticancer drugs. The anthracyclines exert their cytotoxic action through four major mechanisms: (1) inhibition of topoisomerase II; (2) high-‐affinity binding to DNA through intercalation, with consequent blockade of the synthesis of DNA and RNA, and DNA strand scission; (3) generation of semiquinone free radicals and oxygen free radicals through an iron-‐dependent, enzyme-‐mediated reductive process; (4) binding to cellular membranes to alter fluidity and ion transport. Doxorubicin • is one of the most important anticancer drugs in clinical practice, with major clinical activity in cancers of the breast, endometrium, ovary, testicle, thyroid, stomach, bladder, liver, and lung Epirubicin • • is an anthracycline analog initially approved for use as a component of adjuvant therapy in early-‐stage, node-‐positive breast cancer but is also used in the treatment of metastatic breast cancer and gastroesophageal cancer. B. MITOMYCIN • undergoes metabolic activation through an enzyme-‐mediated reduction to generate an alkylating agent that cross-‐links DNA. C. BLEOMYCIN • small peptide that contains a DNA-‐binding region and an iron-‐binding domain at opposite ends of the molecule. It acts by binding to DNA, which results in single-‐ and double-‐ strand breaks following free radical formation, and inhibition of DNA biosynthesis.
- 9. The American Society of Clinical Oncology (ASCO) breast cancer surveillance guidelines: • Women with a history of breast cancer should perform monthly BSE and undergo annual mammography of both the preserved and contralateral breast. • The patient should also have a complete history and physical examination every 3 to 6 months for the first 3 years after diagnosis, then every 6 to 12 months for 2 years, and then annually. SECTION 3E-‐ CLINICAL THERAPEUTICS CASE 12 BREAST CA 9
- 10. NATIONAL COMPREHENSIVE CANCER NETWORK SUMMARY: Since patient was diagnosed 6 years ago with Breast cancer: Stage IIB infiltrating ductal carcinoma of the right breast. Originally, the tumor was ER_/PR_ and did not overexpress HER-‐2/neu. The tumor was staged as T2N1M0. She received a lumpectomy with axillary lymph node dissection plus breast irradiation, 6 cycles of AC (A= ADRIAMYCIN an anthracycline; C=Cyclophosphamide), and tamoxifen for 5 years. NEXT step would be to change tamox to anastrazole and begin with chemotherapy preferably combination since patient had already a history of being treated with a combination chemo drugs –AC. Choice would depend on patient’s comorbidities and toxicities from chemo drugs.
- 11. PROBLEM 2: Bone Pain Zepeda Basis for diagnosis: • • • Chief Complaint: Severe (7 out of 10) hip pain Bone scan: multiple metastases to the right pelvis Medications: Ibuprofen 200 to 400 mg PO q4–6h PRN, calcium carbonate 1,000 mg PO TID with meals GOALS OF THERAPY: • • • Decrease the severity of pain from severe to moderate To minimize adverse reactions or intolerance to pain management therapies Improve the patient’s quality of life and optimize ability to perform activities of daily living Bone is the most common site of secondary breast cancer or breast cancer recurrence. Most commonly affected are the spine, skull, upper bones of the arms and legs and pelvis which is the one affected in our patient. Pain is defined as “an unpleasant sensory and emotional experience associated with actual or potential tissue damage, or described in terms of such damage”. It is the most common symptom that provokes people to seek medical attention Normally, the bone undergoes a continuous process of remodeling by the osteoclast and osteoblasts to maintain homeostasis. Disruption of this process, which occurs in cancer, will cause the bone cells to proliferate and hypertrophy causing the periosteum to stretch or affect the nerves thereby resulting to pain. The World Health Organization developed a stepladder for relief of pain management in adult cancer patient. It indicates the severity of pain which is rated in 1-‐10 scale and will dictate what type of medication is needed or used. Stage 1: Mild (Pain Scale: 1-‐3) Non-‐opioids are the first choice of treatment. Medications include are Acetaminophen or NSAIDS like Ibuprofen. Stage 2:Moderate (Pain Scale: 4-‐6) Those who are not responded to the first step should receive a weak opioid such as codeine, oxycodone, hydrocodone and Tramadol Stage 3: Severe (Pain Scale: 7-‐10) Those who have not been relieved by the previous recommendation will receive a stronger opioid such as Morphine, Methadone and Fentanyl. SECTION 3E-‐ CLINICAL THERAPEUTICS CASE 12 BREAST CA 11
- 12. Key Points: • Oral route is preferred unless contraindicated (parenteral therapy may be required for refractory pain or inability to take per orem) • Cancer pain is continuous. Relief of pain is only temporary and may return in a short time • Should be scheduled at regular intervals rather than prn • Adjuvant therapy is used to decrease anxiety and fear with chronic pain (e.g. antidepressants) • Non-‐opioids may be given in Step 2 and 3 Treatment: Opioids -‐ refers broadly to all compounds related to opium, a natural product derived from the poppy plant -‐ reduce moderate to severe pain, and are unique in their ability to do this without producing loss of consciousness -‐ produce analgesia, affect mood and rewarding behavior and alter respiratory, cardiovascular, GI, and neuroendocrine function -‐ All opioids have the potential for tolerance, habituation, and addiction The patient experiences a severe type of pain, therefore will be following the Step 3. Drug Morphine Efficacy ++++ Suitability +++ Safety ++ Hydromorphone ++++ 4-‐5x more potent than morphine +++ 100x +++ ++ ++ Only available in IV, buccal, spinal and patch + Not available in the Philippines ++ Fentanyl Methadone ++++ 0.3x ++ Cost ++++ Tab 60's (P1345.00/pack) ++ Tab 28's (P3640.00/pack) ++ Patch 5 × 1's (P2513.00/box) + Not available in the Philippines
- 13. Methadone: (Diphenylheptanes) -‐ long-‐acting mu-‐receptor agonist with properties qualitatively similar to those of morphine. -‐ relief of chronic pain, treatment of opioid abstinence syndromes, and treatment of heroin users. -‐ roughly equivalent in potency to morphine on a single dose basis; however, with repeated administration accumulation in CNS and lipid tissues occurs Fentanyl: (Phenylpiperidines) -‐ is a synthetic opioid derivative of the 4-‐anilinophenyl-‐piperidine class -‐ approximately 100 times more potent than morphine -‐ used clinically as an analgesic; administered intraspinally or intravenously and as a preoperative anesthetic agent because of its potency, rapid onset, and short duration of action -‐ Not suitable for rapid dose filtration. Should be used for relatively stable analgesic requirement. -‐ Also available as a transdermal patch which can be given every 8 days Hydromorphone: (Phenanthrenes) -‐ Semisyntheticopioid that xerts major pharmacodynamic effects on mu-‐receptors and kappa-‐ receptors -‐ less potential to produce nausea, vomiting, constipation, sedation, or euphoria and has a more rapid onset and shorter duration of action than morphine -‐ can be used as a substitute when these adverse effects warrant a therapeutic alternative Morphine: (Phenanthrenes) -‐ prototype strong opioid agonist (the gold standard given for cancer patients with moderate-‐severe pain) -‐ Exert major pharmacodynamic effects on mu-‐receptors (strong) and kappa-‐receptors -‐ Interact w/ opioid receptors in the CNS and GIT causing hyperpolarization of nerve cells, inhibition of nerve firing and presynaptic inhibition of transmitter release -‐ Acts at κ receptors in lamina I and II of the substantia gelatinosa of the SC which then decreases the release of substance P -‐ Main indication is for preoperative pain and chronic malignant pain * All are efficacious but have different potency. Methadone is not available in the Philippines. All opioids have produce these side/adverse effects: constipation (most common), nausea, vomiting, somnolence, mood changes like euphoria, dysphoria, addiction, physical dependence and respiratory depression (most dreaded complication). Drug of Choice: Morphine Sulphate Drug interactions: Paroxetine and Morphine: Opioids may enhance effect of SSRI. Additive effect to sedation. Metformin and Morphine: increase effects of Metformin Lisinopril and Morphine: may have additive effect causing hypotension *Therefore it is important to take the medication as prescribed and strictly monitor compliance. Plan of Action • Initiate Morphine Sulphate immediate release 15mg PO q3-‐4hours • If the opiate requirement is determined, switch to a sustained release formulation SECTION 3E-‐ CLINICAL THERAPEUTICS CASE 12 BREAST CA 13
- 14. • • • • Start with: Senna 1 tablet PO BID (stool softener) Docusate sodium 100 mg PO BID (laxative) * All these adverse events (nausea, vomiting, sedation, confusion, constipation, or itching) except constipation will be gone. Should take these two medications every day to prevent constipation from morphine) Ibuprofen 800mg q8h with food Pamidronate 90 mg IV over 2 hours every 4 weeks (Check SCr prior to each dose) Monitor Efficacy (decrease pain scale and opiate requirement) and Toxicity (increase in pain, opiate requirement, nausea, vomiting, itching, BP, constipation, confusion, sedation, respiratory rate, renal function, platelets, Hct/Hgb, signs and symptoms of bleeding, calcium, magnesium, phosphate Report any prolonged adverse events, severe confusion/lightheadedness, or difficulty breathing Important to take the pain medication around the clock to prevent the pain from recurring Non-‐Pharmacologic Intervention: • Relaxation Techniques, massage therapy, and exercise can be done • Counsel KF that the pain may not completely resolve but that it should substantially decrease and she should notice an improvement in mobility Problem 3: Hypercalcemia of Malignancy Secondary to Bone Metastases Villanueva Hypercalcemia in patients with cancer is primarily due to increased bone resorption and release of calcium from bone. There are three major mechanisms by which this can occur: osteolytic metastases with local release of cytokines (including osteoclast activating factors); tumor secretion of parathyroid hormone-‐related protein (PTHrP); and tumor production of 1,25-‐dihydroxyvitamin D (calcitriol). In this case, I. Basis for diagnosis • • • • • • • • Breast cancer: commonly associated with hypercalcemia Pain on right hip Decreased appetite Increasing fatigue Constipation More forgetful Confusion Ca level: 12.5 (N:8.5-‐10.2) II. Treatment objectives a. To reduce serum calcium level b. To reverse signs and symptoms of hypercalcemia c. avoid exacerbation of hypercalcemia d. Reduce gastrointestinal calcium absorption
- 15. III. Management A. Therapeutic B. Non pharmacologic Therapeutic Loop diuretic Bisphosphonat es Calcitonin Mechanism of action enhances urine flow but also inhibits calcium reabsorption in the ascending limb of the loop of Henle Mimic pyrophosphat e's structure, inhibiting activation of enzymes that utilize pyrophosphat e -‐ binding and blocking the enzyme farnesyldipho sphate synthase (FPPS) in the HMG-‐CoA reductase pathway Calcitonin lowers plasma Ca2+ and phosphate concentration s thereby blocking bone resorption, increases urinary calcium excretion by Indications Adverse effects acute pulmonary edema, other edematous conditions, acute hypercalcemia . ototoxicity, hypovolemia, K wasting, hyperuricemia Oral, IV , hypomagnese mia osteoclast-‐ mediated bone resorption, including osteoporosis, steroid-‐ induced osteoporosis, Paget's disease, tumor-‐ associated osteolysis, breast and prostate cancer, and hypercalcemia . upset stomach and inflammation and erosions of the esophagus, IV: can give fever and flu-‐like symptoms after the first infusion, rareosteonecr osis of the jaw Oral, IV 50% is excreted unchanged by the kidney. The remainder has a very high affinity for bone tissue, and is rapidly adsorbed onto the bone surface nasuea, vomitting effect on serum calcium is observed within 4–6 hours and lasts for 6–10 hours, subcutaneous, intranasal, oral Paget’s diasease, osteoporosis Pharmocokine tics SECTION 3E-‐ CLINICAL THERAPEUTICS CASE 12 BREAST CA 15
- 16. inhibiting renal calcium reabsorption reducing serum calcium nephrotoxicity Oral, t1/2: 1 in cancer hr patients thrombocytop decreases enia, Reduction in plasma Ca2+ hemorrhage, plasma Ca2+ concentration hepatic and concentration hypercalcemia s by inhibiting renal toxicity s occurs bone hypocalcemia, within 24 to resorption. nausea, and 48 hours vomiting IV: short-‐ hypocalcemia, termcalcemic ectopic control of calcification, some patients acute renal Binds to Ca with primary failure, and Oral and IV ions hyperparathyr hypotension. oidism who Oral: ectopic are awaiting calcification surgery. and renal failure inhibiting Gallium Nitrate bone resorption Plicamycin (Mithramycin) Phosphate Rapid reduction of serum calcium is required. The first steps include rehydration with saline and diuresis with furosemide. Saline rehydration is used to dilute serum calcium and promote calciuresis. Most patients presenting with severe hypercalcemia have a substantial component of prerenal azotemia owing to dehydration, which prevents the kidney from compensating for the rise in serum calcium by excreting more calcium in the urine. Therefore, the initial infusion of 500–1000 mL/h of saline to reverse the dehydration and restore urine flow can by itself substantially lower serum calcium. The addition of a loop diuretic such as furosemide following rehydration not only enhances urine flow but also inhibits calcium reabsorption in the ascending limb of the loop of Henle. Monitoring central venous pressure is important to forestall the development of heart failure and pulmonary edema in predisposed subjects. Calcitonin Calcitonin has proved useful as ancillary treatment in a large number of patients. Calcitonin by itself seldom restores serum calcium to normal, and refractoriness frequently develops. However, its lack of toxicity permits frequent administration at high doses (200 MRC units or more). An effect on serum calcium is observed within 4–6 hours and lasts for 6–10 hours. The drug has its greatest effect on spine and is most effective in patients who have high bone turnover rates. Calcitonin also has a significant analgesic effect on acute pain from vertebral fracture that is independent of its effects on bone metabolism.Given by injection or intranasal spray. Recommended injectable dosage is 100IU (SQ or IM) and the intranasal dosage is 200IU (one spray) per day in alternate nostrils. Oral formulation is under investigation. Side effects of injectable calcitonin include nausea and GI discomfort. This may be
- 17. minimized by bedtime administration. Pruritus at the injection site is also problematic. To minimize these side effects, patients should be instructed to administer calcitonin SQ rather than IM. Intranasal formulation appears to be better tolerated; rhinitis is the most commonly reported side effect. Gallium Nitrate Gallium nitrate is approved by the FDA for the management of hypercalcemia of malignancy. This drug acts by inhibiting bone resorption. Given as continuous intravenous infusion in 5% dextrose for 5 days, gallium nitrate proved superior to calcitonin in reducing serum calcium in cancer patients. Because of potential nephrotoxicity, patients should be well hydrated and have good renal output before starting the infusion. Plicamycin (Mithramycin) Because of its toxicity, plicamycin (mithramycin) is not the drug of first choice for the treatment of hypercalcemia. However, when other forms of therapy fail, 25–50 mcg/kg given intravenously usually lowers serum calcium substantially within 24–48 hours. This effect can last several days. This dose can be repeated as necessary. The most dangerous toxic effect is sudden thrombocytopenia followed by hemorrhage. Hepatic and renal toxicity can also occur. Hypocalcemia, nausea, and vomiting may limit therapy. Use of this drug must be accompanied by careful monitoring of platelet counts, liver and kidney function, and serum calcium levels. Phosphate Giving intravenous phosphate is probably the fastest and surest way to reduce serum calcium, but it is a hazardous procedure if not done properly. Intravenous phosphate should be used only after other methods of treatment (bisphosphonates, calcitonin, and saline diuresis) have failed to control symptomatic hypercalcemia. The risks of intravenous phosphate therapy include sudden hypocalcemia, ectopic calcification, acute renal failure, and hypotension. Oral phosphate can also lead to ectopic calcification and renal failure if serum calcium and phosphate levels are not carefully monitored, but the risk is less and the time of onset much longer Biphosphonates Alendronate Risedronate Ibandronate Zoledronate Pamidronate Efficacy +++ +++ +++ ++++ ++++ Safety +++ +++ ++ ++ +++ Suitability ++++ +++ +++ +++ +++ Cost +++ P1100 ++ P1,800 + P17,000 + P24,000 ++ P1700 First-‐generation bisphosphonates contain minimally modified side chains (R1, R2) (medronate, clodronate, and etidronate) or contain a chlorophenyl group (tiludronate). They are the least potent and in some instances cause bone demineralization. Second-‐generation aminobisphosphonates (e.g., alendronate and pamidronate) contain a nitrogen group in the side chain. They are 10 to 100 times more potent than first-‐generation compounds. SECTION 3E-‐ CLINICAL THERAPEUTICS CASE 12 BREAST CA 17
- 18. Third-‐generation bisphosphonates (e.g., risedronate and zoledronate) contain a nitrogen atom within a heterocyclic ring and are up to 10,000 times more potent than first-‐generation agents Alendronate and ibandronate directly inhibit multiple steps in the pathway from mevalonate to cholesterol and isoprenoid lipids, such as geranylgeranyldiphosphate, that are required for the prenylation of proteins that are important for osteoclast function. The potency of inhibiting farnesyl synthase correlates directly with their antiresorptive activity. They should not be taken with iron supplements, vitamins with minerals, or antacids containing calcium, magnesium, or aluminum because they reduce absorption of bisphosphonates. Pamidronate is approved for management of hypercalcemia but also is effective in other skeletal disorders. Pamidronate is available only for parenteral administration. For treatment of hypercalcemia, pamidronate may be given as an intravenous infusion of 60 to 90 mg over 4 to 24 hours. Electrolyte imbalances may occur with pamidronate use. Pamidronate overdose could manifest with a low blood calcium level. Twitching, anxiety, muscle weakness or seizures could result.Onset: 24-‐48 hr. Duration: Peak effect: max 5-‐7 days. Absorption: Poor absorption. Excretion: Elimination half-‐life: 21-‐35 hr. Excretion: Biphasic; urine (approx 50% as unchanged drug) within 120 hr. Zoledronate has been associated with renal toxicity, deterioration of renal function, and potential renal failure. Thus, the infusion should be given over at least 15 minutes, and the dose should be 4 mg. Patients who receive zoledronate should have standard laboratory and clinical parameters of renal function assessed prior to treatment and periodically after treatment to monitor for deterioration in renal function. It can be administered at home rather than in hospital. With monitoring of Ca level, albumin, phosphate level, K level, Mg level, Na level, hydration status (BUN, SCr, BP, HR). Distribution: Protein binding: Low (22-‐56%). Excretion: Excreted unchanged in urine (23-‐55%), the rest sequestered to bone and eliminated very slowly.The total time between reconstitution, dilution, storage in a refrigerator at 2-‐ 8°C and end of administration must not exceed 24 hrs. First-‐generation bisphosphonate etidronate was associated with osteomalacia. Alendronate and risedronate were well tolerated in clinical trials, some patients experience symptoms of esophagitis. If symptoms persist despite precautions, use a proton pump inhibitor at. Both drugs may be better tolerated on a once-‐weekly regimen with no reduction of efficacy. Patients with active upper gastrointestinal disease should not be given oral bisphosphonates. Mild fever and aches may attend the first parenteral infusion of pamidronate, likely owing to cytokine release. These symptoms are short-‐lived and generally do not recur with subsequent administration. All oral bisphosphonates are very poorly absorbed from the intestine and have remarkably limited bioavailability [<1% (alendronate, risedronate) to 6% (etidronate, tiludronate)]. Thus these drugs should be administered with a full glass of water following an overnight fast and at least 30 minutes before breakfast. Oral bisphosphonates have not been used widely in children or adolescents because of uncertainty of long-‐term effects of bisphosphonates on the growing skeleton. Bisphosphonates are excreted primarily by the kidneys. Adjusted doses for patients with diminished renal function have not been determined; bisphosphonates currently are not recommended for patients with a creatinine clearance of less than 30 ml/min. Non pharmacologic 1. Hold calcium supplement Patient education 1. Confusion, decreased appetite, constipation are due to high calcium level 2. Nausea and vomiting are side effects of pamidronate 3. Eat small frequent meals to help with the nausea and vomiting
- 19. Edward Philip I. Villanueva FEU-‐NRMF Medical Center Regalado Avenue, West Fairview, Quezon City Room 416 MWF – 10:00am-‐11:00am Tel no: (02) 632-‐1234 Patient: Kay Floyd January 30, 2014 62 years old, female Address: #4 Iris St., West Fairview, Quezon City Description: D:FEU-‐NRMFSY 12-‐13 2nd semClinical Therapeutics 3ACase 4 REPORTRx.jpg Pamidronate 90 mg Sig: initiate pamidronateintravenouslyover 2 hours Edward Philip I. Villanueva,MD Lic. No. 123456 PTR No. 78910 PROBLEM 4: DIABETES MELLITUS TYPE 2 Zagada Basis for diagnosis: • Type 2 diabetes mellitus for 7 years • 20 packs per year tobacco history • Overweight • HbA1c=7 Type 2 diabetes is characterized by tissue resistance to the action of insulin combined with a relative deficiency in insulin secretion GOALS OF THERAPY: • • Continue control of blood sugar by maintaining normal or near-‐normal ranges o Keep HbA1C of <7 Prevent disease and drug related complications The major goal of pharmacologic therapy for diabetes is to normalize metabolic parameters, such as blood sugar, in order to reduce the risk of long-‐term complications. Treatment: The treatment of Type II diabetes is multifaceted. First, obese patients should endeavor to reduce body weight and increase exercise in order to improve insulin sensitivity. Some Type II patients can achieve good control of their diabetes by modifying their diet and exercise habits. Pharmacologically, treatments include orally available agents that act to slow glucose absorption from the gut (a-‐glucosidase inhibitors), to increase insulin secretion by ß cells (sulfonylureas, meglitinides, and GLP-‐1 mimetics), or to increase insulin sensitivity at target tissues (thiazolidinediones and biguanides). These agents are generally ineffective for patients with Type I diabetes. Patients with Type II diabetes are frequently treated with combinations of these drugs and are therefore utilizing multiple strategies. SECTION 3E-‐ CLINICAL THERAPEUTICS CASE 12 BREAST CA 19
- 20. Class of Drugs used for Diabetes Drug Class Action SULFONYLUREA AND MEGLITINIDES Insulin secretagogue BIGUANIDES Clinical Application Effects Insulin Sensitizer • • THIAZOLIDINEDIONES Insulin Sensitizer Competitive inhibitors • ALPHA-‐GLUCDIDASE of the intestinal INHIBITOR α-‐glucosidases • • Glucagon-‐like peptide-‐ • GLP-‐1 AGONISTS 1 (GLP-‐1) receptor • agonist reduce circulating glucose increase glycogen,fat, and protein formation Decreased endogenous glucose production Reduces insulin resistance Reduce conversion of starch and disaccharides to monosaccharides reduce postprandial hyperglycemia enhances glucose-‐dependent insulin secretion inhibits glucagon secretion delays gastric emptying, and decreases appetite DM type 2 DM type 2 DM type 2 DM type 2 DM type 2 Class THIAZOLIDINEDIONES (TZDs) BIGUANIDES GLP-‐1 AGONISTS Sulfonylureas A-‐glucosidase inhibitors +++ Efficacy +++ Safety Suitability ++++ ++ Cost ++++ ++++ +++ +++ ++++ +++ +++ +++ ++++ ++++ +++ +++ ++++ ++ +++ ++ 1. SULFONYLUREAS AND MEGLITINIDES -‐inhibit the ß cell K+/ATP channel at the SUR1 subunit, thereby stimulating insulin release from pancreatic ß cells and increasing circulating insulin to levels sufficient to overcome insulin resistance. First-‐generation sulfonylureas: Second-‐generation sulfonylureas: Acetohexamide Glimepiride Chlorpropamide Glipizide Tolazamide Glibenclamide (Glyburide) Tolbutamide Gliclazide Gliquidone Sulfonylureas are the mainstay of treatment for Type II diabetes; orally available and metabolized by the liver. The major adverse effect is hypoglycemia resulting from oversecretion of insulin; Thus, these medications should be used cautiously in patients who are unable to recognize or respond appropriately to hypoglycemia, such as those with impaired sympathetic function, mental status changes (our patient has depression), or advanced age. However this agents can cause weight gain secondary to increased insulin activity in adipose tissue; therefore, are better suited for nonobese patients (wherein our patient is already overweight). The adverese effect of hypoglycemia and weight gain makes this drugs less suitable for our patient. As with sulfonylureas, meglitinides stimulate insulin release by binding to SUR1 and inhibiting the ß cell K+/ATP channel. Although both sulfonylureas and meglitinides act on the SUR1 subunit, these two classes of drugs bind to distinct regions of the SUR1 molecule. The absorption, metabolism, and adverse effect profiles of meglitinides are similar to those of sulfonylureas.
- 21. 2. BIGUANIDES (METFORMIN) -‐ activates AMP-‐dependent protein kinase (AMPPK) to block breakdown of fatty acids and to inhibit hepatic gluconeogenesis and glycogenolysis; increases insulin receptor activity and metabolic responsiveness in liver and skeletal muscle. The most common adverse effect is mild gastrointestinal distress, which is usually transient and can be minimized by slow titration of the dose. A potentially more serious adverse effect is lactic acidosis. Because biguanides decrease the flux of metabolic acids through gluconeogenic pathways, lactic acid can accumulate to dangerous levels in biguanide-‐treated patients. This drug is currently being taken by the patient. 3. THIAZOLIDINEDIONES (TZDS) -‐ bind and stimulate the nuclear hormone receptor peroxisome proliferator activated receptor-‐γ (PPARγ), thereby increasing insulin sensitivity in adipose tissue, liver, and muscle. The TZDs do not affect insulin secretion, but rather enhance the action of insulin at target tissues. Two thiazolidinediones are currently available: pioglitazone and rosiglitazone. An adverse effect common to both Tzds is fluid retention, which presents as a mild anemia and peripheral edema, especially when the drugs are used in combination with insulin or insulin secretagogues. Both drugs increase the risk of heart failure. Many users have a dose-‐related weight gain (average 1–3 kg), which may be fluid related. This is drug (Rosiglitazone) is currently being taken by the patient but its adverse effect profile may warrant its discontinuation. 4. ALPHA-‐GLUCOSIDASE INHIBITORS are carbohydrate analogues that bind avidly to intestinal brush border a-‐glucosidase enzymes, slowing breakdown and absorption of dietary carbohydrates such as starch, dextrin, and disaccharides. Flatulence, bloating, abdominal discomfort, and diarrhea are common adverse effects, all of which result from gas released by bacteria acting on undigested carbohydrates that reach the large intestine. The patient is currently taking metformin which can ossibly cause GI distress and lactic acidosis making this drug less favorable addition to the patients treatment. Examples of this drugs are Acarbose, Miglitol and Voglibose SECTION 3E-‐ CLINICAL THERAPEUTICS CASE 12 BREAST CA 21
- 22. 5. GLP-‐1 (GLUCAGON-‐LIKE PEPTIDE-‐1) MIMETICS -‐ are the newest class of drugs developed for the treatment of diabetes. (Ex; Exanitide and Sitagliptin). Exenatide is Glucagon-‐like peptide-‐1 (GLP-‐1) receptor agonist is not orally available and must be injected while Sitagliptin (available orally) is a dipeptidyl peptidase-‐IV (DPP IV) inhibitor that slows the proteolytic inactivation of GLP-‐1 and other incretin hormones. This agents can be used as monotherapy or in combination with a TZD or metformin. The known physiological functions of GLP-‐1 include: • Increases insulin secretion from the pancreas in a glucose-‐dependent manner. • Decreases glucagon secretion from the pancreas by engagement of a specific G protein-‐coupled receptor. • increases insulin-‐sensitivity in both alpha cells and beta cells • Increases beta cells mass and insulin gene expression, post-‐translational processing and incretion. • Inhibits acid secretion and gastric emptying in the stomach. • Decreases food intake by increasing satiety in brain • Promotes insulin sensitivity. Dose adjustment is necessary in patients with moderate or severe kidney disease. This agents may ay cause hypoglycemia in combination with sulfonylureas and insulin. In this case, taking into consideration the patients condition, we chose to give a combination therapy of Sitagliptin and Metformin. Drug of choice: Sitagliptin + Metformin (Janumet) maintenance 50 mg/500 mg tab twice a day. Non pharmacologic Intervention: • Counsel KF to; – continue diabetes medications and self-‐monitoring. – Remind her of the importance of diet/exercise in the treatment of diabetes. – Remind her to maintain all follow-‐up appointments for diabetes. – Report any shortness of breath or swelling in the legs to the physician.
- 23. PROBLEM 5: DEPPRESSION Basis for diagnosis: • Present in patients medical history • Use of SSRI-‐paroxetine (controlled under current regiment) • Decreased appetite over the past few weeks and increasing fatigue. • Slightly confused Selectively inhibit reuptake of serotonin – increase synaptic serotonin levels – also cause increased 5HT receptor activation and enhanced postsynaptic responses. At present, SSRIs are the most commonly prescribed first-‐line agents in the treatment of both MDD and anxiety disorders. Their popularity comes from their ease of use, tolerability, and safety in overdose. GOALS OF THERAPY: • • Continue monitoring for signs and symptoms of depression Continue therapy to avoid future episodes • Continue current regimen – controlled with current regimen – Paroxetine, 20 mg PO daily Treatment: Non-‐Pharmacologic Intervention: • Counsel KF to continue depression medication unless otherwise directed by her physician. • She should seek a psychologist to discuss her new diagnosis. She should report any new/worsened depression symptoms to her physician. SUMMARY: To address the patients diabetes, we chose a combination therapy of Sitagliptin and metformin taking into account the patients present condition. Sulfonylureas can cause hypoglycemia and weight gain which is not favorable since the patient is already overweight. Alpha glucosidase inhibitors causes abdominal distention and flatulence. TZD’s can cause fluid retention and edema and is also known to worsen CVD’s. Therefore we chose to retain Metformin, a Biguanide which is currently used by the patient and replace Rosiglitazone (TZD’s) with GLP-‐1 mimetics which is a newer class of drug with multiple effects mechanism in promoting euglycemia. There were no changes in the patient’s medications for Depression because it is currently controlled by the current regimen thus, paroxetine was retained. SECTION 3E-‐ CLINICAL THERAPEUTICS CASE 12 BREAST CA 23
- 24. CLINICAL THERAPEUTICS CASE 12 BREAST CANCER Proctor: Dr. Zenaida Maglaya Reporters: Villanueva, Edward Phillip Yang, Sheryl Ray Zagada, Timothy Zepeda, Monina Mae 3E
