Melden
Teilen
Downloaden Sie, um offline zu lesen
Empfohlen
Empfohlen
Presentation delivered at the University of Michigan Injury Center Opioid Overdose Summit, Ann Arbor, MI, December 1, 2015Non-medical Use and Medical Misuse of Opioids during Adolescence by Carol J B...

Non-medical Use and Medical Misuse of Opioids during Adolescence by Carol J B...University of Michigan Injury Center
Weitere ähnliche Inhalte
Was ist angesagt?
Presentation delivered at the University of Michigan Injury Center Opioid Overdose Summit, Ann Arbor, MI, December 1, 2015Non-medical Use and Medical Misuse of Opioids during Adolescence by Carol J B...

Non-medical Use and Medical Misuse of Opioids during Adolescence by Carol J B...University of Michigan Injury Center
Was ist angesagt? (20)
Ibalizumab - Journal Club Handout (Holden Young - Roseman University of Healt...

Ibalizumab - Journal Club Handout (Holden Young - Roseman University of Healt...
Chemotherapy+with+or+without+gefitinib+in+patients+with+advanced+non small-ce...

Chemotherapy+with+or+without+gefitinib+in+patients+with+advanced+non small-ce...
13 vol.-4-issue-2-feb-2013-ijpsr-ra-2131-paper-13 (1)

13 vol.-4-issue-2-feb-2013-ijpsr-ra-2131-paper-13 (1)
The gap between clinical practice and limited evidence of traditional Chinese...

The gap between clinical practice and limited evidence of traditional Chinese...
Non-medical Use and Medical Misuse of Opioids during Adolescence by Carol J B...

Non-medical Use and Medical Misuse of Opioids during Adolescence by Carol J B...
Journal of Clinical Pharmacology & Biopharmaceutics

Journal of Clinical Pharmacology & Biopharmaceutics
A Cross Sectional Study of Ethnic Differences in Occurrence and Severity of A...

A Cross Sectional Study of Ethnic Differences in Occurrence and Severity of A...
Prescriptions filled following an opioid-related hospitalization.

Prescriptions filled following an opioid-related hospitalization.
Preliminary study of Prescription audit for evaluation of prescribing pattern...

Preliminary study of Prescription audit for evaluation of prescribing pattern...
Inappropriate drug use in hospitalized elderly patients of medicine and cardi...

Inappropriate drug use in hospitalized elderly patients of medicine and cardi...
Andere mochten auch
Andere mochten auch (9)
Mark woodward smith news release | Stephen rayment

Mark woodward smith news release | Stephen rayment
Matteo cornali - Amministratore delegato, membro del Gruppo Restate

Matteo cornali - Amministratore delegato, membro del Gruppo Restate
Eri Digitaalinen valokuvaus vinkkejä - Tony Henrik Halttunen

Eri Digitaalinen valokuvaus vinkkejä - Tony Henrik Halttunen
Ähnlich wie Bupe vs No Bupe
Ähnlich wie Bupe vs No Bupe (20)
Homeopathic and conventional treatment for acute respiratory and ear complain...

Homeopathic and conventional treatment for acute respiratory and ear complain...
“The Value of Drug Monitoring in Chronic Opioid Therapy Patients”

“The Value of Drug Monitoring in Chronic Opioid Therapy Patients”
“The Value of Drug Monitoring in Chronic Opioid Therapy Patients”

“The Value of Drug Monitoring in Chronic Opioid Therapy Patients”
Outcomes in Long-term Opioid Tapering and Buprenorphine Transition: A Retrosp...

Outcomes in Long-term Opioid Tapering and Buprenorphine Transition: A Retrosp...
Proton Pump Inhibitors and Risk of Acute and Chronic Kidney Disease: A Retros...

Proton Pump Inhibitors and Risk of Acute and Chronic Kidney Disease: A Retros...
Journal Article Analysis: Medication Errors in Overweight and Obese Pediatric...

Journal Article Analysis: Medication Errors in Overweight and Obese Pediatric...
Concerns and Perceived Barriers Related to Treatment of Opioid Addiction with...

Concerns and Perceived Barriers Related to Treatment of Opioid Addiction with...
A Tool to Engage the Patient in Web-based Coordinated Treatment of Opioid Add...

A Tool to Engage the Patient in Web-based Coordinated Treatment of Opioid Add...
Trends in management of rheumatoid arthritis Dr.Neena Mehan

Trends in management of rheumatoid arthritis Dr.Neena Mehan
Mehr von Paul Coelho, MD
Mehr von Paul Coelho, MD (20)
Labeling Woefulness: The Social Construction of Fibromyalgia

Labeling Woefulness: The Social Construction of Fibromyalgia
Fibromyalgia & Somatic Symptom Disorder Patient Handout

Fibromyalgia & Somatic Symptom Disorder Patient Handout
Mortality quadrupled among opioid-driven hospitalizations notably within lowe...

Mortality quadrupled among opioid-driven hospitalizations notably within lowe...
Correlation of opioid mortality with prescriptions and social determinants -a...

Correlation of opioid mortality with prescriptions and social determinants -a...
The place-of-antipsychotics-in-the-therapy-of-anxiety-disorders-and-obsessive...

The place-of-antipsychotics-in-the-therapy-of-anxiety-disorders-and-obsessive...
Opioids for the Treatment of Chronic Pain: Mistakes Made, Lessons Learned, an...

Opioids for the Treatment of Chronic Pain: Mistakes Made, Lessons Learned, an...
The potential adverse influence of physicians’ words.

The potential adverse influence of physicians’ words.
The conundrum of opioid tapering in long term opioid therapy for chronic pain...

The conundrum of opioid tapering in long term opioid therapy for chronic pain...
Wolfe (2012) culture, science and the changing nature of fibromyalgia nrr s...

Wolfe (2012) culture, science and the changing nature of fibromyalgia nrr s...
Kürzlich hochgeladen
❤️ Call Girls service In Panchkula☎️9815457724☎️ Call Girl service in Panchkula☎️ Panchkula Call Girls Service ☎️ Call Girls In Panchkula BEST CALL GIRL ESCORTS SERVICE IN PANCHKULA CALL WATTSAPP 9815457724 THE MOST BEAUTIFUL INDEPENDENT ESCORT CALL GIRL SERVICE In Panchkula WE ARE PROVIDING GENUINE CALL GIRL SERVICE
I AM A a NATURAL BRUNETTES, SLIM BODY, NATURAL LONG HAIR AND ALL TYPE OF HAIR IS A NATURAL BRUNETTE IN THE MOST BEAUTIFUL MODELS INDEPENDENT ESCORT GIRL I AM A NATURAL BRUNETTE WITH ROOM AND HOTEL AND A NATURAL BRUNETTE WITH A BODY MADE FOR SIN AND ALL TYPE OF ME ALL THE TIME
I SEND YOU A HAIR, VERY SOCIABLE AND FUNNY, READY TO ENTERTAIN TO ENTERTAIN U AND MAKE FORGET ABOUT TO AGET ENTERTAINMENT YOU AND MAKE FORGET ABOUT ALL THE PROBLEMS. LET'S HAVE A WONDERFUL TIME TOGETHER AND FORGET ABOUT EVERYTHING ALL TYPE SERVICE ENJOYMENT SAFE AND SECURE IN CALL OUT CALL HOME AND HOTEL ANYTIME AVAILABLE
AND ALL TYPE SERVICE ENJOYMENTPANCHKULA INDEPENDENT BEST CALL GIRL ESCORTS SERVICE IN PANCHKULA INDEPENDENT CALL GIRLS❤️ Call Girls service In Panchkula☎️9815457724☎️ Call Girl service in Panchku...

❤️ Call Girls service In Panchkula☎️9815457724☎️ Call Girl service in Panchku...Rashmi Entertainment
❤️Chandigarh Escorts Service☎️9815457724☎️ Call Girl service in Chandigarh☎️ Chandigarh Call Girls Service ☎️ Call Girls In Chandigarh BEST Call Girls in CHANDIGARH Escort Service provide Cute Nice sweet and Sexy Models in beautiful CHANDIGARH city cash in hand to hand call girl in CHANDIGARH and CHANDIGARH escorts. HOT & SEXY MODELS // COLLEGE GIRLS IN CHANDIGARH AVAILABLE FOR COMPLETE ENJOYMENT WITH HIGH PROFILE INDIAN MODEL AVAILABLE HOTEL & HOME ★ SAFE AND SECURE HIGH CLASS SERVICE AFFORDABLE RATE ★ 100% SATISFACTION,UNLIMITED ENJOYMENT. ★ All Meetings are confidential and no information is provided to any one at any cost.
★ EXCLUSIVE Profiles Are Safe and Consensual with Most Limits Respected
★ Service Available In: - HOME & 24x7 :: 3 * 5 *7 *Star Hotel Service .In Call & Out call
Services :
★ A-Level (5 star escort)
★ Strip-tease
★ BBBJ (Bareback Blowjob)Receive advanced sexual techniques in different mode make their life more pleasurable.
★ Spending time in hotel rooms
★ BJ (Blowjob Without a Condom)
★ Completion (Oral to completion)
★ Covered (Covered blowjob Without a Condom)Chandigarh Call Girls❤️Chandigarh Escorts Service☎️9815457724☎️ Call Girl service in Chandigarh☎️ ...

❤️Chandigarh Escorts Service☎️9815457724☎️ Call Girl service in Chandigarh☎️ ...Rashmi Entertainment
Kürzlich hochgeladen (20)
Low Rate Call Girls Jaipur {9521753030} ❤️VVIP NISHA CCall Girls in Jaipur Es...

Low Rate Call Girls Jaipur {9521753030} ❤️VVIP NISHA CCall Girls in Jaipur Es...
💞 Safe And Secure Call Girls Mysore 🧿 9332606886 🧿 High Class Call Girl Servi...

💞 Safe And Secure Call Girls Mysore 🧿 9332606886 🧿 High Class Call Girl Servi...
Low Rate Call Girls Pune {9142599079} ❤️VVIP NISHA Call Girls in Pune Maharas...

Low Rate Call Girls Pune {9142599079} ❤️VVIP NISHA Call Girls in Pune Maharas...
💞 Safe And Secure Call Girls Prayagraj 🧿 9332606886 🧿 High Class Call Girl Se...

💞 Safe And Secure Call Girls Prayagraj 🧿 9332606886 🧿 High Class Call Girl Se...
💞 Safe And Secure Call Girls gaya 🧿 9332606886 🧿 High Class Call Girl Service...

💞 Safe And Secure Call Girls gaya 🧿 9332606886 🧿 High Class Call Girl Service...
Independent Call Girls Bangalore {7304373326} ❤️VVIP POOJA Call Girls in Bang...

Independent Call Girls Bangalore {7304373326} ❤️VVIP POOJA Call Girls in Bang...
💚 Low Rate Call Girls In Chandigarh 💯Lucky 📲🔝8868886958🔝Call Girl In Chandig...

💚 Low Rate Call Girls In Chandigarh 💯Lucky 📲🔝8868886958🔝Call Girl In Chandig...
science quiz bee questions.doc FOR ELEMENTARY SCIENCE

science quiz bee questions.doc FOR ELEMENTARY SCIENCE
❤️Zirakpur Escorts☎️7837612180☎️ Call Girl service in Zirakpur☎️ Zirakpur Cal...

❤️Zirakpur Escorts☎️7837612180☎️ Call Girl service in Zirakpur☎️ Zirakpur Cal...
Call Girls Service Amritsar Just Call 9352988975 Top Class Call Girl Service ...

Call Girls Service Amritsar Just Call 9352988975 Top Class Call Girl Service ...
Premium Call Girls Bangalore {9179660964} ❤️VVIP POOJA Call Girls in Bangalor...

Premium Call Girls Bangalore {9179660964} ❤️VVIP POOJA Call Girls in Bangalor...
❤️Chandigarh Escorts☎️9814379184☎️ Call Girl service in Chandigarh☎️ Chandiga...

❤️Chandigarh Escorts☎️9814379184☎️ Call Girl service in Chandigarh☎️ Chandiga...
Erotic Call Girls Bangalore {7304373326} ❤️VVIP SIYA Call Girls in Bangalore ...

Erotic Call Girls Bangalore {7304373326} ❤️VVIP SIYA Call Girls in Bangalore ...
❤️ Call Girls service In Panchkula☎️9815457724☎️ Call Girl service in Panchku...

❤️ Call Girls service In Panchkula☎️9815457724☎️ Call Girl service in Panchku...
💞 Safe And Secure Call Girls Coimbatore 🧿 9332606886 🧿 High Class Call Girl S...

💞 Safe And Secure Call Girls Coimbatore 🧿 9332606886 🧿 High Class Call Girl S...
Indore Call Girl Service 📞9235973566📞Just Call Inaaya📲 Call Girls In Indore N...

Indore Call Girl Service 📞9235973566📞Just Call Inaaya📲 Call Girls In Indore N...
💸Cash Payment No Advance Call Girls Surat 🧿 9332606886 🧿 High Class Call Girl...

💸Cash Payment No Advance Call Girls Surat 🧿 9332606886 🧿 High Class Call Girl...
Ulhasnagar Call girl escort *88638//40496* Call me monika call girls 24*

Ulhasnagar Call girl escort *88638//40496* Call me monika call girls 24*
Low Rate Call Girls Udaipur {9xx000xx09} ❤️VVIP NISHA CCall Girls in Udaipur ...

Low Rate Call Girls Udaipur {9xx000xx09} ❤️VVIP NISHA CCall Girls in Udaipur ...
❤️Chandigarh Escorts Service☎️9815457724☎️ Call Girl service in Chandigarh☎️ ...

❤️Chandigarh Escorts Service☎️9815457724☎️ Call Girl service in Chandigarh☎️ ...
Bupe vs No Bupe
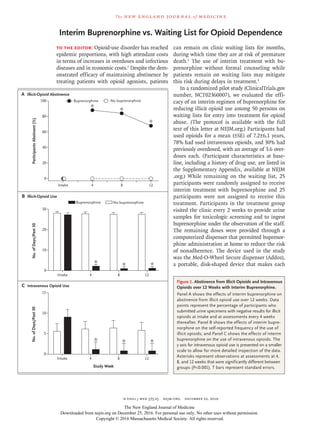
- 1. The new engl and jour nal of medicine n engl j med 375;25 nejm.org December 22, 2016 Interim Buprenorphine vs. Waiting List for Opioid Dependence To the Editor: Opioid-use disorder has reached epidemic proportions, with high attendant costs in terms of increases in overdoses and infectious diseases and in economic costs.1 Despite the dem- onstrated efficacy of maintaining abstinence by treating patients with opioid agonists, patients can remain on clinic waiting lists for months, during which time they are at risk of premature death.2 The use of interim treatment with bu- prenorphine without formal counseling while patients remain on waiting lists may mitigate this risk during delays in treatment.3 In a randomized pilot study (ClinicalTrials.gov number, NCT02360007), we evaluated the effi- cacy of an interim regimen of buprenorphine for reducing illicit opioid use among 50 persons on waiting lists for entry into treatment for opioid abuse. (The protocol is available with the full text of this letter at NEJM.org.) Participants had used opioids for a mean (±SE) of 7.2±6.1 years, 78% had used intravenous opioids, and 30% had previously overdosed, with an average of 3.6 over- doses each. (Participant characteristics at base- line, including a history of drug use, are listed in the Supplementary Appendix, available at NEJM .org.) While remaining on the waiting list, 25 participants were randomly assigned to receive interim treatment with buprenorphine and 25 participants were not assigned to receive this treatment. Participants in the treatment group visited the clinic every 2 weeks to provide urine samples for toxicologic screening and to ingest buprenorphine under the observation of the staff. The remaining doses were provided through a computerized dispenser that permitted buprenor- phine administration at home to reduce the risk of nonadherence. The device used in the study was the Med-O-Wheel Secure dispenser (Addoz), a portable, disk-shaped device that makes each Figure 1. Abstinence from Illicit Opioids and Intravenous Opioids over 12 Weeks with Interim Buprenorphine. Panel A shows the effects of interim buprenorphine on abstinence from illicit opioid use over 12 weeks. Data points represent the percentage of participants who submitted urine specimens with negative results for illicit opioids at intake and at assessments every 4 weeks thereafter. Panel B shows the effects of interim bupre norphine on the self-reported frequency of the use of illicit opioids, and Panel C shows the effects of interim buprenorphine on the use of intravenous opioids. The y axis for intravenous opioid use is presented on a smaller scale to allow for more detailed inspection of the data. Asterisks represent observations at assessments at 4, 8, and 12 weeks that were significantly different between groups (P<0.001). T bars represent standard errors.Study Week * ** * ** Buprenorphine No buprenorphine Buprenorphine No buprenorphine * * * ParticipantsAbstinent(%) 100 80 60 40 20 0 Intake 4 8 12 No.ofDays/Past30 30 20 10 0 Intake 4 8 12 No.ofDays/Past30 15 10 5 0 Intake 4 8 12 B Illicit-Opioid Use A Illicit-Opioid Abstinence C Intravenous Opioid Use The New England Journal of Medicine Downloaded from nejm.org on December 25, 2016. For personal use only. No other uses without permission. Copyright © 2016 Massachusetts Medical Society. All rights reserved.
- 2. Correspondence n engl j med 375;25 nejm.org December 22, 2016 day’s dose available during a preprogrammed 3-hour window. Participants in the treatment group also received daily calls to assess drug use, craving, and withdrawal by means of an inter- active voice-response telephone system as well as system-generated random callbacks. Participants in the control group remained on the waiting list of their local clinic and did not receive these services. At 4, 8, and 12 weeks, all participants com- pleted assessments that included the provision of urine specimens that were collected under staff observation and the completion of a struc- tured clinical interview based on the Addiction Severity Index (ASI),4 which addresses problem severity in seven areas commonly affected in sub- stance abusers: drug use, alcohol use, employ- ment, legal issues, family and social issues, psychiatric issues, and medical issues. (Patient scores are available in the Supplementary Appen- dix.) The primary outcome was the percentage of specimens with negative results for illicit opioids obtained at assessments at 4, 8, and 12 weeks; missing urine specimens were considered to be positive. Secondary outcomes included in- travenous drug use, ASI scores, adherence to the treatment regimen, and patient satisfaction. Participants assigned to receive interim treat- ment with buprenorphine submitted a higher percentage of specimens that were negative for illicit opioids than those in the control group at 4 weeks (88% vs. 0%), 8 weeks (84% vs. 0%), and 12 weeks (68% vs. 0%), which was the primary outcome (P<0.001 for all comparisons) (Fig. 1). These participants also had greater reductions in the frequency of use of any intravenous drug (P<0.001) and in scores on the drug (P<0.001) and psychiatric (P = 0.02) subscales of the ASI. Adherence to the regimens for buprenorphine administration (99%), daily monitoring calls (96%), and random callbacks (96%) was high, as were ratings of treatment satisfaction (4.6±0.7 on a 5-point scale). Among patients on a waiting list to receive comprehensive treatment, interim dosing with buprenorphine, paired with technology-assisted components intended to support adherence, was associated with a statistically significant reduc- tion in the use of illicit opioids and intravenous drugs as compared with remaining on the wait- ing list alone over 12 weeks. These results suggest that interim buprenorphine dosing could reduce drug-related risks and consequences when com- prehensive treatment is unavailable. Interim treat- ment with buprenorphine may be suitable for patients in rural areas where there are limited treatment options.5 Further trials with larger sample sizes and longer durations are needed to replicate these preliminary findings. Stacey C. Sigmon, Ph.D. Taylor A. Ochalek, B.A. Andrew C. Meyer, Ph.D. Bryce Hruska, Ph.D. Sarah H. Heil, Ph.D. Gary J. Badger, M.S. Gail Rose, Ph.D. John R. Brooklyn, M.D. University of Vermont Burlington, VT stacey.sigmon@uvm.edu Robert P. Schwartz, M.D. Friends Research Institute Baltimore, MD Brent A. Moore, Ph.D. Yale University School of Medicine New Haven, CT Stephen T. Higgins, Ph.D. University of Vermont Burlington, VT Supported by grants from the National Institutes of Health (R34DA037385, to Dr. Sigmon) and (T32DA007242, to Dr. Hig- gins) and the National Institute of General Medical Sciences (P20GM103644, to Dr. Higgins). Disclosure forms provided by the authors are available with the full text of this letter at NEJM.org. 1. Jones CM, Mack KA, Paulozzi LJ. Pharmaceutical overdose deaths, United States, 2010. JAMA 2013;309:657-9. 2. Peles E, Schreiber S, Adelson M. Opiate-dependent patients on a waiting list for methadone maintenance treatment are at high risk for mortality until treatment entry. J Addict Med 2013; 7:177-82. 3. Sigmon SC, C Meyer A, Hruska B, et al. Bridging waitlist delays with interim buprenorphine treatment: initial feasibility. Addict Behav 2015;51:136-42. 4. McLellan AT, Luborsky L, Cacciola J, et al. New data from the Addiction Severity Index: reliability and validity in three cen- ters. J Nerv Ment Dis 1985;173:412-23. 5. Paulozzi LJ, Xi Y. Recent changes in drug poisoning mortal- ity in the United States by urban-rural status and by drug type. Pharmacoepidemiol Drug Saf 2008;17:997-1005. DOI: 10.1056/NEJMc1610047 Correspondence Copyright © 2016 Massachusetts Medical Society. The New England Journal of Medicine Downloaded from nejm.org on December 25, 2016. For personal use only. No other uses without permission. Copyright © 2016 Massachusetts Medical Society. All rights reserved.
