Comparison of Small Molecule Drugs and Biologic Products
•
63 gefällt mir•8,927 views
Recent surveys indicate that physicians in the US and Europe are still unclear on what a biosimilar is – even those physicians who prescribe biologic medicines. See the infographic for a deeper look at the differences between biosimilar and generic medicines. Get the latest on Biosimilars at https://www.smallworldsocial.com/biolink/ Curated resources updated daily.
Melden
Teilen
Melden
Teilen
Downloaden Sie, um offline zu lesen
Empfohlen
Empfohlen
Weitere ähnliche Inhalte
Andere mochten auch
Andere mochten auch (12)
UX, ethnography and possibilities: for Libraries, Museums and Archives

UX, ethnography and possibilities: for Libraries, Museums and Archives
How Much Does Venture Capital Drive the U.S. Economy?

How Much Does Venture Capital Drive the U.S. Economy?
The Shift: UX Designer as Business Consultant (2016)

The Shift: UX Designer as Business Consultant (2016)
How do we see the healthcare's digital future and its impact on our lives?

How do we see the healthcare's digital future and its impact on our lives?
3 Things Every Sales Team Needs to Be Thinking About in 2017

3 Things Every Sales Team Needs to Be Thinking About in 2017
Mehr von Small World Social
Mehr von Small World Social (11)
5 Upcoming Biosimilar Events You Don't Want to Miss

5 Upcoming Biosimilar Events You Don't Want to Miss
The Complexities of Social Media, kids and the Classroom

The Complexities of Social Media, kids and the Classroom
Overcoming the camel's hump with digital literacy: how Web 2.0 is changing ed...

Overcoming the camel's hump with digital literacy: how Web 2.0 is changing ed...
Kürzlich hochgeladen
Kürzlich hochgeladen (20)
High Class Escorts in Hyderabad ₹7.5k Pick Up & Drop With Cash Payment 969456...

High Class Escorts in Hyderabad ₹7.5k Pick Up & Drop With Cash Payment 969456...
Kochi ❤CALL GIRL 84099*07087 ❤CALL GIRLS IN Kochi ESCORT SERVICE❤CALL GIRL

Kochi ❤CALL GIRL 84099*07087 ❤CALL GIRLS IN Kochi ESCORT SERVICE❤CALL GIRL
Vip profile Call Girls In Lonavala 9748763073 For Genuine Sex Service At Just...

Vip profile Call Girls In Lonavala 9748763073 For Genuine Sex Service At Just...
Justdial Call Girls In Indirapuram, Ghaziabad, 8800357707 Escorts Service

Justdial Call Girls In Indirapuram, Ghaziabad, 8800357707 Escorts Service
Formation of low mass protostars and their circumstellar disks

Formation of low mass protostars and their circumstellar disks
Pulmonary drug delivery system M.pharm -2nd sem P'ceutics

Pulmonary drug delivery system M.pharm -2nd sem P'ceutics
Pests of mustard_Identification_Management_Dr.UPR.pdf

Pests of mustard_Identification_Management_Dr.UPR.pdf
SAMASTIPUR CALL GIRL 7857803690 LOW PRICE ESCORT SERVICE

SAMASTIPUR CALL GIRL 7857803690 LOW PRICE ESCORT SERVICE
pumpkin fruit fly, water melon fruit fly, cucumber fruit fly

pumpkin fruit fly, water melon fruit fly, cucumber fruit fly
Asymmetry in the atmosphere of the ultra-hot Jupiter WASP-76 b

Asymmetry in the atmosphere of the ultra-hot Jupiter WASP-76 b
Chemical Tests; flame test, positive and negative ions test Edexcel Internati...

Chemical Tests; flame test, positive and negative ions test Edexcel Internati...
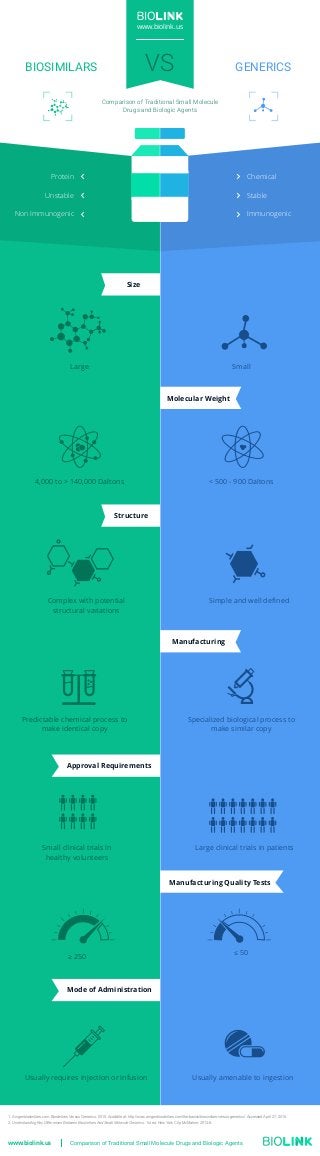
Comparison of Small Molecule Drugs and Biologic Products
- 1. Small Molecular Weight Manufacturing Size Structure Manufacturing Quality Tests Approval Requirements Mode of Administration Large 4,000 to > 140,000 Daltons < 500 - 900 Daltons Complex with potential structural variations Simple and well defined Specialized biological process to make similar copy Predictable chemical process to make identical copy ≥ 250 ≤ 50 Large clinical trials in patients Usually requires injection or infusion Small clinical trials in healthy volunteers Usually amenable to ingestion VSBIOSIMILARS GENERICS Comparison of Traditional Small Molecule Drugs and Biologic Agents www.biolink.us Immunogenic Stable Chemical Non Immunogenic Unstable Protein www.biolink.us Comparison of Traditional Small Molecule Drugs and Biologic Agents 1. Amgenbiosimilars.com. Biosimilars Versus Generics. 2015. Available at: http://www.amgenbiosimilars.com/the-basics/biosimilars-versus-generics/. Accessed April 27, 2015. 2. Understanding Key Differences Between Biosimilars And Small Molecule Generics. 1st ed. New York City: McMahon; 2013:8.
