20 Apr 9 Nuclear, Strong Force, Fission With Brainstorming
•Als PPT, PDF herunterladen•
3 gefällt mir•688 views
The strong force (nuclear force), fission
Melden
Teilen
Melden
Teilen
Empfohlen
Weitere ähnliche Inhalte
Was ist angesagt?
Was ist angesagt? (20)
Prof Steve F King 'The standard models in particle physics'

Prof Steve F King 'The standard models in particle physics'
Lattice Energy LLC - Many body collective magnetic mechanism creates ultrahig...

Lattice Energy LLC - Many body collective magnetic mechanism creates ultrahig...
New Energy Part 3: The Science - Quantum Electrodynamics

New Energy Part 3: The Science - Quantum Electrodynamics
Andere mochten auch
Andere mochten auch (7)
19 Apr 7 Fundamental Forces, Radiation Doses, Medical Applications Actual Pre...

19 Apr 7 Fundamental Forces, Radiation Doses, Medical Applications Actual Pre...
23 Apr 21 Assorted Topics, Mechanics Intro Actual Presented

23 Apr 21 Assorted Topics, Mechanics Intro Actual Presented
Osmotic stress and water isotope effects in kinesin-1 gliding motility assays

Osmotic stress and water isotope effects in kinesin-1 gliding motility assays
Ähnlich wie 20 Apr 9 Nuclear, Strong Force, Fission With Brainstorming
Ähnlich wie 20 Apr 9 Nuclear, Strong Force, Fission With Brainstorming (20)
Radioactivity ( Tajuk : Astronomi & Fizik Moden_Tugasan Kumpulan Sem 1_UTHM)

Radioactivity ( Tajuk : Astronomi & Fizik Moden_Tugasan Kumpulan Sem 1_UTHM)
Mehr von Steve Koch
Mehr von Steve Koch (20)
2011 NSF CAREER_Steve Koch Full Project Description 

2011 NSF CAREER_Steve Koch Full Project Description
24 Apr 28 Newtons Laws, Linear Angular Momentum Presented

24 Apr 28 Newtons Laws, Linear Angular Momentum Presented
21 Apr 14 Fission, Fusion, Nuclear Energy, Nw With Brainstorming

21 Apr 14 Fission, Fusion, Nuclear Energy, Nw With Brainstorming
18 Apr 2 Bg Radiation, Shielding, Inverse Square For Slideshare

18 Apr 2 Bg Radiation, Shielding, Inverse Square For Slideshare
16 Mar 26 Soap Bubble, Photoelectric, Wave Particle Duality Actual

16 Mar 26 Soap Bubble, Photoelectric, Wave Particle Duality Actual
14 Mar 10 Diffraction, Interference Actual Presented For Slideshare

14 Mar 10 Diffraction, Interference Actual Presented For Slideshare
10 Feb 24 Eye, Scattering, Fluorescence, Phosphorescence Slideshare

10 Feb 24 Eye, Scattering, Fluorescence, Phosphorescence Slideshare
08 Feb 17 Light, Electron E Levels Actual Presented

08 Feb 17 Light, Electron E Levels Actual Presented
06 Feb 5 Sound, Interference, Pitch, Beats After Class

06 Feb 5 Sound, Interference, Pitch, Beats After Class
Kürzlich hochgeladen
Mattingly "AI & Prompt Design: The Basics of Prompt Design"

Mattingly "AI & Prompt Design: The Basics of Prompt Design"National Information Standards Organization (NISO)
Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"

Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"National Information Standards Organization (NISO)
Kürzlich hochgeladen (20)
Mattingly "AI & Prompt Design: The Basics of Prompt Design"

Mattingly "AI & Prompt Design: The Basics of Prompt Design"
Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"

Mattingly "AI & Prompt Design: Structured Data, Assistants, & RAG"
The byproduct of sericulture in different industries.pptx

The byproduct of sericulture in different industries.pptx
Z Score,T Score, Percential Rank and Box Plot Graph

Z Score,T Score, Percential Rank and Box Plot Graph
Russian Call Girls in Andheri Airport Mumbai WhatsApp 9167673311 💞 Full Nigh...

Russian Call Girls in Andheri Airport Mumbai WhatsApp 9167673311 💞 Full Nigh...
BAG TECHNIQUE Bag technique-a tool making use of public health bag through wh...

BAG TECHNIQUE Bag technique-a tool making use of public health bag through wh...
9548086042 for call girls in Indira Nagar with room service

9548086042 for call girls in Indira Nagar with room service
Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...

Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...
JAPAN: ORGANISATION OF PMDA, PHARMACEUTICAL LAWS & REGULATIONS, TYPES OF REGI...

JAPAN: ORGANISATION OF PMDA, PHARMACEUTICAL LAWS & REGULATIONS, TYPES OF REGI...
Separation of Lanthanides/ Lanthanides and Actinides

Separation of Lanthanides/ Lanthanides and Actinides
20 Apr 9 Nuclear, Strong Force, Fission With Brainstorming
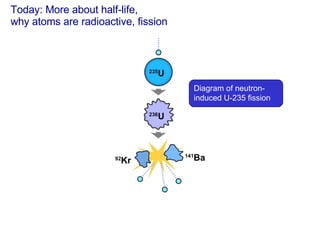
- 1. Today: More about half-life, why atoms are radioactive, fission Diagram of neutron-induced U-235 fission
- 2. Half-life is the average amount of time for ½ of the atoms to decay 100% remaining 50% remaining 25% remaining 12.5% remaining Time Isotope with long half-life Isotope with short half-life “ Exponential Decay”
- 5. Why are some atoms radioactive? First: Some brainstorming Form small groups, with a spokesperson How many different forces in nature can you think of? Can you classify them into similar groups?
- 14. Why are some atoms radioactive? The nuclear (residual strong) force presents an activation barrier Quantum mechanics allows some probability that an alpha particle can “tunnel” through the barrier Once through, electrostatic force dominates (imagine ripping apart velcro)
- 17. As nuclei get larger, electrostatic force “wins” Strong force large between close protons. Strong force much lower between distal protons But electrostatic force still very high! (Nucleons on the surface feel less strong attraction.)
- 18. You can imagine large nuclei like large soap bubbles…very floppy Small Medium Large
