Quantum Pres
•Als PPT, PDF herunterladen•
0 gefällt mir•373 views
The document summarizes the quantum model of the atom. It describes how electrons exhibit wave-like properties and have quantized wavelengths. The Heisenberg uncertainty principle states that it is impossible to know both the velocity and position of an electron at the same time. Schrodinger's wave equation defines quantized energy levels and the probability of finding an electron. Electrons occupy orbitals defined by four quantum numbers that specify the electron's unique "address" within the atom.
Melden
Teilen
Melden
Teilen
Weitere ähnliche Inhalte
Was ist angesagt?
Was ist angesagt? (20)
Chapter 8 electron configuration and periodicity (1)

Chapter 8 electron configuration and periodicity (1)
Quantum numbers shells-subshells-orbitals-electrons

Quantum numbers shells-subshells-orbitals-electrons
Andere mochten auch
Andere mochten auch (20)
Sioux Hot-or-Not: Domain Driven Design (Edwin Van Dillen)

Sioux Hot-or-Not: Domain Driven Design (Edwin Van Dillen)
2014 Developers' Choice Awards Reveal Database Trends

2014 Developers' Choice Awards Reveal Database Trends
With Progress Pacific, The RAD Race Has Already Been Won!

With Progress Pacific, The RAD Race Has Already Been Won!
Ähnlich wie Quantum Pres
Ähnlich wie Quantum Pres (20)
Quantum Numbers and Atomic Orbitals By solving t.pdf

Quantum Numbers and Atomic Orbitals By solving t.pdf
Mehr von kitcoffeen
Mehr von kitcoffeen (20)
Kürzlich hochgeladen
Kürzlich hochgeladen (20)
Scanning the Internet for External Cloud Exposures via SSL Certs

Scanning the Internet for External Cloud Exposures via SSL Certs
"LLMs for Python Engineers: Advanced Data Analysis and Semantic Kernel",Oleks...

"LLMs for Python Engineers: Advanced Data Analysis and Semantic Kernel",Oleks...
Transcript: New from BookNet Canada for 2024: BNC CataList - Tech Forum 2024

Transcript: New from BookNet Canada for 2024: BNC CataList - Tech Forum 2024
Designing IA for AI - Information Architecture Conference 2024

Designing IA for AI - Information Architecture Conference 2024
Developer Data Modeling Mistakes: From Postgres to NoSQL

Developer Data Modeling Mistakes: From Postgres to NoSQL
Gen AI in Business - Global Trends Report 2024.pdf

Gen AI in Business - Global Trends Report 2024.pdf
DevoxxFR 2024 Reproducible Builds with Apache Maven

DevoxxFR 2024 Reproducible Builds with Apache Maven
Streamlining Python Development: A Guide to a Modern Project Setup

Streamlining Python Development: A Guide to a Modern Project Setup
Unraveling Multimodality with Large Language Models.pdf

Unraveling Multimodality with Large Language Models.pdf
WordPress Websites for Engineers: Elevate Your Brand

WordPress Websites for Engineers: Elevate Your Brand
What's New in Teams Calling, Meetings and Devices March 2024

What's New in Teams Calling, Meetings and Devices March 2024
How AI, OpenAI, and ChatGPT impact business and software.

How AI, OpenAI, and ChatGPT impact business and software.
Nell’iperspazio con Rocket: il Framework Web di Rust!

Nell’iperspazio con Rocket: il Framework Web di Rust!
Dev Dives: Streamline document processing with UiPath Studio Web

Dev Dives: Streamline document processing with UiPath Studio Web
Quantum Pres
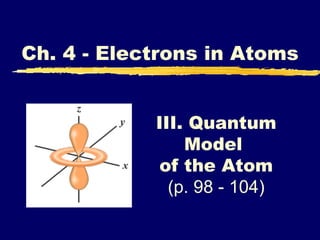
- 1. III. Quantum Model of the Atom (p. 98 - 104) Ch. 4 - Electrons in Atoms
- 3. A. Electrons as Waves QUANTIZED WAVELENGTHS
- 4. A. Electrons as Waves EVIDENCE: DIFFRACTION PATTERNS ELECTRONS VISIBLE LIGHT
- 13. C. Quantum Numbers p x p y p z
- 17. Feeling overwhelmed? Read Section 4-2!