Constructing Organic Compounds Lab (2)
•Download as DOC, PDF•
1 like•568 views
Report
Share
Report
Share
Recommended
More Related Content
What's hot
What's hot (20)
Chapter 3 water and the fitness of the environment

Chapter 3 water and the fitness of the environment
Chemistry Unit2 Part3 - Elements Compounds and Mixtures

Chemistry Unit2 Part3 - Elements Compounds and Mixtures
Viewers also liked
Viewers also liked (6)
Similar to Constructing Organic Compounds Lab (2)
Similar to Constructing Organic Compounds Lab (2) (20)
Chapter 4 carbon and the molecular diversity of life

Chapter 4 carbon and the molecular diversity of life
Essential Biology 03.2 Carbohydrates, Lipids and Proteins

Essential Biology 03.2 Carbohydrates, Lipids and Proteins
More from Toni Legg
More from Toni Legg (20)
Pre-AP Tie-in Objective 8.14A, B, and C Ecosystems and Pollution

Pre-AP Tie-in Objective 8.14A, B, and C Ecosystems and Pollution
Recently uploaded
Recently uploaded (20)
The Codex of Business Writing Software for Real-World Solutions 2.pptx

The Codex of Business Writing Software for Real-World Solutions 2.pptx
Exploring the Future Potential of AI-Enabled Smartphone Processors

Exploring the Future Potential of AI-Enabled Smartphone Processors
08448380779 Call Girls In Friends Colony Women Seeking Men

08448380779 Call Girls In Friends Colony Women Seeking Men
Handwritten Text Recognition for manuscripts and early printed texts

Handwritten Text Recognition for manuscripts and early printed texts
Workshop - Best of Both Worlds_ Combine KG and Vector search for enhanced R...

Workshop - Best of Both Worlds_ Combine KG and Vector search for enhanced R...
Mastering MySQL Database Architecture: Deep Dive into MySQL Shell and MySQL R...

Mastering MySQL Database Architecture: Deep Dive into MySQL Shell and MySQL R...
Axa Assurance Maroc - Insurer Innovation Award 2024

Axa Assurance Maroc - Insurer Innovation Award 2024
TrustArc Webinar - Stay Ahead of US State Data Privacy Law Developments

TrustArc Webinar - Stay Ahead of US State Data Privacy Law Developments
Automating Google Workspace (GWS) & more with Apps Script

Automating Google Workspace (GWS) & more with Apps Script
Strategies for Unlocking Knowledge Management in Microsoft 365 in the Copilot...

Strategies for Unlocking Knowledge Management in Microsoft 365 in the Copilot...
Bajaj Allianz Life Insurance Company - Insurer Innovation Award 2024

Bajaj Allianz Life Insurance Company - Insurer Innovation Award 2024
From Event to Action: Accelerate Your Decision Making with Real-Time Automation

From Event to Action: Accelerate Your Decision Making with Real-Time Automation
Factors to Consider When Choosing Accounts Payable Services Providers.pptx

Factors to Consider When Choosing Accounts Payable Services Providers.pptx
Constructing Organic Compounds Lab (2)
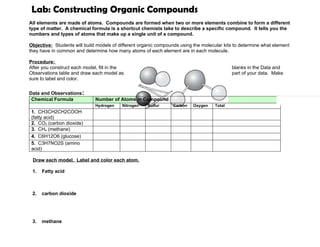
- 1. Lab: Constructing Organic Compounds All elements are made of atoms. Compounds are formed when two or more elements combine to form a different type of matter. A chemical formula is a shortcut chemists take to describe a specific compound. It tells you the numbers and types of atoms that make up a single unit of a compound. Objective: Students will build models of different organic compounds using the molecular kits to determine what element they have in common and determine how many atoms of each element are in each molecule. Procedure: After you construct each model, fill in the blanks in the Data and Observations table and draw each model as part of your data. Make sure to label and color. Data and Observations: Chemical Formula Number of Atoms in Compound Hydrogen Nitrogen Sulfur Carbon Oxygen Total 1. CH3CH2CH2COOH (fatty acid) 2. CO2 (carbon dioxide) 3. CH4 (methane) 4. C6H12O6 (glucose) 5. C3H7NO2S (amino acid) Draw each model. Label and color each atom. 1. Fatty acid 2. carbon dioxide 3. methane
- 2. 4. glucose 5. amino acid Questions and Conclusions: 1. Based on your data and observations, what elements do organic compounds have in common? 2. What are organic compounds? Explain. 3. Give examples of other organic compounds. 4. Distinguish the difference between organic foods in grocery stores and organic compounds? 5. Explain how organic compounds and living things are related.
