2. diabetes mellitus
- 1. Diabetes Mellitus 11/1/2020 Prof. Dr. RS Mehta
- 2. Content • Review of Anatomy • Introduction • Epidemiology • Classification: (Type-I, Type-II, GDM and other) • Diagnostic criteria • Complications • Treatment • Nursing Management 21/1/2020 Prof. Dr. RS Mehta
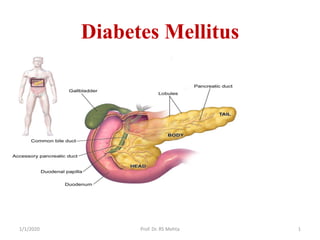
- 3. Anatomy and physiology of Pancrease • Pancreas is a narrow, 6 inch long gland that lies posterior and inferior to stomach on left side of abdominal cavity. • Pancreas extends laterally and superiorly across abdomen from the curve of duodenum to spleen. • The head of pancreas, which connects to the duodenum, is the widest region. • The glandular tissue surrounds many small ducts that drain into the central pancreatic duct. 31/1/2020 Prof. Dr. RS Mehta
- 4. Functions Pancreas is a dual-function gland, having features of both endocrine and exocrine glands. Endocrine • Pancreas with endocrine function is made of million cell called islets of Langerhans. • Four main cell types in the islets: α alpha cells- glucagon(increase glucose in blood), β beta cells- insulin (decrease glucose in blood), Δ delta cells- somatostatin (regulates/stops α and β cells) and PP cells , or γ (gamma) cells, secrete pancreatic polypeptide. 41/1/2020 Prof. Dr. RS Mehta
- 5. 51/1/2020 Prof. Dr. RS Mehta
- 6. Exocrine • Secretes pancreatic fluid that contains digestive enzymes that pass to small intestine. • These enzymes help to further break down carbohydrates, proteins and lipids (fats) in the chyme. • Digestive enzymes include trypsin, chymotrypsin, pancreatic lipase, and pancreatic amylase, and are produced and secreted by acinar cells of the exocrine pancreas. • Specific cells lining pancreatic ducts, called centroacinar cells, secrete bicarbonate- and salt-rich solution into the small intestine. 61/1/2020 Prof. Dr. RS Mehta
- 7. 71/1/2020 Prof. Dr. RS Mehta
- 8. 81/1/2020 Prof. Dr. RS Mehta
- 9. Introduction • Diabetes Mellitus (DM) refers to a group of common metabolic disorders that share the phenotype of hyperglycemia. • Caused by complex interaction of genetics and environmental factor. • Factors – reduced insulin secretion, decreased glucose utilization, and increased glucose production. 91/1/2020 Prof. Dr. RS Mehta
- 10. Epidemiology According to International Diabetes Federation, South East Asia: 382 million people have diabetes in the world and more than 72.1 million people in the South East Asia Region; by 2035 this will rise to 123 million. There were 674,120 cases of diabetes in Nepal in 2013. 101/1/2020 Prof. Dr. RS Mehta
- 11. Classification • Type 1 diabetes (due to β-cell destruction, usually leading to absolute insulin deficiency). • Type 2 diabetes (due to a progressive insulin secretory defect on the background of insulin resistance). • Gestational diabetes mellitus (diabetes diagnosed during pregnancy that is not clearly overt diabetes). • Diabetes mellitus associated with other conditions or causes. 111/1/2020 Prof. Dr. RS Mehta
- 12. Prediabetes Categories of increased risk for Diabetes (prediabetes)* FBG 100 mg/dL (5.6 mmol/L) to 125 mg/dL (6.9 mmol/L) (IFG) OR 2-h PG in the 75-g OGTT 140 mg/dL (7.8 mmol/L) to 199 mg/dL (11.0 mmol/L) (IGT) OR A1C 5.7–6.4% *For all three tests, risk is continuous, extending below the lower limit of the range and becoming disproportionately greater at higher ends of the range. 121/1/2020 Prof. Dr. RS Mehta
- 13. Diagnosis Criteria for the diagnosis of Diabetes Mellitus A1C ≥ 6.5%. The test should be performed in a laboratory using a method that is NGSP certified and standardized to the DCCT assay.* OR FPG ≥ 126 mg/dL (7.0 mmol/L). Fasting is defined as no caloric intake for at least 8 h.* OR Two-hour PG ≥ 200 mg/dL (11.1 mmol/L) during an OGTT. The test should be performed as described by the WHO, using a glucose load containing the equivalent of 75 g anhydrous glucose dissolved in water.* OR In a patient with classic symptoms of hyperglycemia or hyperglycemic crisis, a random plasma glucose ≥ 200 mg/dL (11.1 mmol/L). *In the absence of unequivocal hyperglycemia, result should be confirmed by repeat testing. National Glycohemoglobin Standardization Program (NGSP) Diabetes Control and Complications Trial (DCCT) 131/1/2020 Prof. Dr. RS Mehta
- 14. Type 1 Diabetes • Type 1diabetes was previously called insulin-dependent diabetes or also called juvenile-onset diabetes, as it often begins in childhood. • Type 1 diabetes is characterized by destruction of the pancreatic beta cells, leading to absolute insulin deficiency. • It is thought that combined genetic, immunologic and possibly environmental (eg, viral) factors contribute to beta cell destruction. 141/1/2020 Prof. Dr. RS Mehta
- 15. DM Type 1 Pathology Genetic Factor • It is generally accepted that genetic susceptibility is a underlying factor in the development of type 1 diabetes. • People do not inherit type 1 diabetes itself; rather, they inherit a genetic predisposition or tendency, toward developing type 1 diabetes. • This genetic tendency has been found in people with certain HLA (human leukocyte antigen) types. 151/1/2020 Prof. Dr. RS Mehta
- 16. contd…. • One proposal is that reduced exposure to microorganisms in early childhood limits maturation of the immune system and increases susceptibility to autoimmune disease (the 'hygiene hypothesis'). • Stress may precipitate type 1 diabetes by stimulating the secretion of counter-regulatory hormones and possibly by modulating immune activity. • Dietary factors may also be important. • Various nitrosamines (found in smoked and cured meats) and coffee have been proposed as potentially diabetogenic toxins. 161/1/2020 Prof. Dr. RS Mehta
- 17. Clinical Features • Polyuria: Caused by osmotic diuresis secondary to hyperglycemia • Thirst: response to the hyperosmolar state and dehydration • Fatigue and weakness: may be caused by muscle wasting from the catabolic state of insulin deficiency, hypovolemia, and hypokalemia • Extreme hunger with unintended weight loss • Muscle cramps: caused by electrolyte imbalance • Blurred vision: Glucose and its metabolites cause osmotic swelling of the lens, altering its normal focal length • Feeling numbness or tingling in feet 171/1/2020 Prof. Dr. RS Mehta
- 18. Diagnosis Age: before the age of 30. The incidence is 12 to 14 cases per 1,00,000 people younger than 20 years and 1 case per 500 people younger than 16 years. Blood Tests: • Fasting blood test (126 mg/dL (7 mmol/L) or higher on two separate tests, is considered diabetes). • (HbA1c) test: 6.5% or higher • RBS level: 200 mg/dL (11.1 mmol/L) or higher 181/1/2020 Prof. Dr. RS Mehta
- 19. Contd… Immunologic Markers: • Islet cell autoantibodies (ICAs) are a composite of several different antibodies directed at pancreatic islet molecules such as glutamic acid decarboxylase(GAD), insulin and IA-2/ICA-512 and serve as a marker of the autoimmune process of type 1 DM. • Testing for ICAs can be useful in classifying the type of DM as type 1 and in identifying nondiabetic individuals at risk for developing type 1 DM. Urine test: Ketone bodies 191/1/2020 Prof. Dr. RS Mehta
- 20. Treatment Insulin Therapy 1.Rapid acting insulin • The rapid acting insulin is used as a bolus dosage. • The action onsets in 15 minutes with peak actions in 30 to 90 minutes. Drugs: Humalog or lispro, Novolog or aspart, Apidra or glulisine 2. Short acting insulin • Short acting insulin action onsets within 30 minutes with the peak action around 2 to 4 hours. Drugs: Regular (R) humulin or novolin, Velosulin (for use in the insulin pump) 3. Intermediate acting insulin I • Action onsets within 1 to 2 hours with peak action of 4 to 10 hours. • Drugs: NPH (N) 201/1/2020 Prof. Dr. RS Mehta
- 21. 4. Long acting insulin • It is usually given around bedtime. • Action onset is roughly 1 to 2 hours with a sustained action of 24 hours. Drugs: Lantus (insulin glargine), Levemir (insulin detemir) 5. Premixed insulin • Combination of specific proportions of intermediate-acting and short-acting insulin in one bottle or insulin pen. • These products are generally taken two or three times a day before mealtime. • Action onset is 30minutes to 4hours with sustained action up to 24hours. Drugs: Humulin 70/30, Novolin 70/30, Novolog 70/30, Humulin 50/50, Humalog mix 75/25 211/1/2020 Prof. Dr. RS Mehta
- 22. Treatment • Dietary plan • Regular check up of blood sugar levels: Blood glucose to the near normal range, approximately 80– 140 mg/dl • Physically active or exercise • Controlling blood pressure • Monitoring cholesterol levels • Transplantation of Pancreas • Transplantation of Islet cells 221/1/2020 Prof. Dr. RS Mehta
- 23. Prevention Immunosuppressive drugs • Cyclosporine A • Anti-CD3 antibodies, including teplizumab and otelixizumab, had suggested evidence of preserving insulin production (as evidenced by sustained C-peptide production) in newly diagnosed type 1 diabetes patients. Diet • Some research has suggested breastfeeding decreases the risk in later life. • Giving children 2000 IU of Vitamin D during their first year of life is associated with reduced risk of type 1 diabetes, though the causal relationship is obscure. 231/1/2020 Prof. Dr. RS Mehta
- 24. Type 2 Diabetes • was previously referred to as non-insulin dependent diabetes mellitus or adult onset diabetes. • is a more complex condition than type 1 diabetes as there is a combination of resistance to the actions of insulin in liver and muscle together with impaired pancreatic β-cell function leading to 'relative' insulin deficiency. • Insulin resistance appears to come first, and leads to elevated insulin secretion in order to maintain normal blood glucose levels. • However, in susceptible individuals the pancreatic β cells are unable to sustain the increased demand for insulin and a slowly progressive insulin deficiency develops. 241/1/2020 Prof. Dr. RS Mehta
- 25. contd… Insulin resistance • refers to decreased tissue sensitivity to insulin. • Normally, insulin binds to special receptors on cell surfaces and initiates a series of reactions involved in glucose metabolism. • In type 2 diabetes, these intracellular reactions are diminished, thus rendering insulin less effective at stimulating glucose uptake by the tissues and at regulating glucose release by the liver. • The exact mechanisms that lead to insulin resistance and impaired insulin secretion in type 2 diabetes are unknown, although genetic factors are thought to play a role. 251/1/2020 Prof. Dr. RS Mehta
- 26. contd… Impaired pancreatic β-cell function • Insulin secretion and sensitivity are interrelated. • In type 2 DM, insulin secretion initially increases in response to insulin resistance to maintain normal glucose tolerance. • Initially, the insulin secretory defect is mild and selectively involves glucose-stimulated insulin secretion. • The response to other nonglucose secretagogues, such as arginine, is preserved. • Eventually, the insulin secretory defect progresses to a state of grossly inadequate insulin secretion. • The reason for the decline in insulin secretory capacity in type 2 DM is unclear. • The assumption is that a second genetic defect—superimposed on insulin resistance—leads to beta cell failure. 261/1/2020 Prof. Dr. RS Mehta
- 27. Pathophysiology • Type 2 DM is characterized by impaired insulin secretion, insulin resistance, excessive hepatic glucose production, and abnormal fat metabolism. • Obesity (particularly visceral or central) is very common in type 2 DM. • In the early stages of the disorder, glucose tolerance remains near-normal, despite insulin resistance, because the pancreatic beta cells compensate by increasing insulin output. • As insulin resistance and compensatory hyperinsulinemia progress, the pancreatic islets in certain individuals are unable to sustain the hyperinsulinemic state. • A further decline in insulin secretion and an increase in hepatic glucose production lead to overt diabetes with fasting hyperglycemia. • Ultimately, beta cell failure may ensue. 271/1/2020 Prof. Dr. RS Mehta
- 28. Risk factors for DM type 2 • Family history of diabetes (i.e., parent or sibling with type 2 diabetes): Both parents have type 2 DM, the risk approaches 40%. • Obesity (BMI ≥25 kg/m2) • Habitual physical inactivity • Race/ethnicity (e.g., African American, Latino, Native American, Asian American, Pacific Islander) 281/1/2020 Prof. Dr. RS Mehta
- 29. contd… • History of GDM or delivery of baby >4 kg (>9 lb) • Hypertension (blood pressure ≥140/90 mmHg) • HDL cholesterol level <35 mg/dL (0.90 mmol/L) and/or a triglyceride level >250 mg/dL (2.82 mmol/L) • Polycystic ovary syndrome • History of vascular disease 291/1/2020 Prof. Dr. RS Mehta
- 30. Treatment of DM type 2 • Type 2 diabetes management should begin with Medical nutrition therapy (MNT). • An exercise regimen to increase insulin sensitivity and promote weight loss should also be instituted. • Pharmacologic approaches to the management of type 2 DM include oral glucose-lowering agents, insulin, and other agents that improve glucose control. • Type 2 DM is a progressive disorder, requires multiple therapeutic agents and often insulin 301/1/2020 Prof. Dr. RS Mehta
- 31. Glucose-Lowering Agents Sulfonylureas • Primary action is directly stimulating the pancreas to secrete insulin. • Therefore, a functioning pancreas is necessary for these agents to be effective, and they cannot be used in patients with type 1 diabetes. • These agents improve insulin action at the cellular level and may also directly decrease glucose production by the liver. • The sulfonylureas can be divided into first- and second-generation categories. 311/1/2020 Prof. Dr. RS Mehta
- 32. Contd… First-Generation Sulfonylureas • Acetohexamide (Dymelor) • Ahlorpropamide (Diabinese) • Tolazamide (Tolinase) • Tolbutamide (Orinase) Second-Generation Sulfonylureas • Glipizide (Glucatrol) • Glyburide (Micronase) • Glimepiride (Amaryl) 321/1/2020 Prof. Dr. RS Mehta
- 33. Biguanides • It produces antidiabetic effects by facilitating insulin’s action on peripheral receptor sites. • Therefore, it can be used only in the presence of insulin. • There is some evidence that it also impairs glucose absorption by the gut and inhibits hepatic gluconeogenesis. • Increases susceptibility to lactic acidosis. • Contraindicate in patients with impaired renal or hepatic function and in those who drink alcohol in excess. Drug: Metformin 331/1/2020 Prof. Dr. RS Mehta
- 34. contd… Alpha Glucosidase Inhibitors • work by delaying the absorption of glucose in the intestinal system, resulting in a lower postprandial blood glucose level. • must be taken immediately before a meal, making therapeutic adherence. • Side effects are diarrhea and flatulence. • Drug: Acarbose (Precose) 341/1/2020 Prof. Dr. RS Mehta
- 35. Thiazolidinediones • enhance insulin action at the receptor site without increasing insulin secretion from the beta cells of the pancreas. • may affect liver function; therefore, liver function studies must be performed (monthly for the first 12 months of treatment, and quarterly thereafter). Drug: Pioglitazone (Actos), rosiglitazone (Avandia) Meglitinides • lower the blood glucose level by stimulating insulin release from the pancreatic beta cells. • Its effectiveness depends on the presence of functioning beta cells. Drug: Repaglinide (Prandin), nateglinide (Starix) 351/1/2020 Prof. Dr. RS Mehta
- 36. • DDP 4 Inhibitors Drugs: sitagliptin, Saxagliptin, Vildagliptin, Linagliptin • GLP-1 analogues: GLP-1 analogues have been found to be particularly effective in helping to improve blood glucose levels by stimulating insulin secretion and helping with weight loss. Drugs: • Dulaglutide (Trulicity) • Exenatide (Byetta) • Liraglutide (Victoza) • Lixisenatide (Lyxumia) 361/1/2020 Prof. Dr. RS Mehta
- 37. Prevention of DM type 2 • Lifestyle modifications and pharmacologic agents prevent or delay the onset of DM. • Weight loss of 7% of body weight and increasing physical activity to at least 150 min/week of moderate activity such as walking. • The Diabetes Prevention Program (DPP) demonstrated that intensive changes in lifestyle (diet and exercise for 30 min/d five times per week) in individuals with delayed the development of type 2 DM by 58% and • Metformin prevented or delayed diabetes by 31%. 371/1/2020 Prof. Dr. RS Mehta
- 38. Gestational Diabetes • Refers to hyperglycaemia occurring for the first time during pregnancy. • During normal pregnancy, insulin sensitivity is reduced through the action of placental hormones (human placental lactogen, estrogen, and cortisol) and this affects glucose tolerance. • GDM occurs in ͠ 4% of pregnancies in the United States • Most women revert to normal glucose tolerance post-partum but have a substantial risk (30–60%) of developing DM later in life. 381/1/2020 Prof. Dr. RS Mehta
- 39. DETECTION & DIAGNOSIS OF GESTATIONAL DIABETES MELLITUS (GDM) Recommendations for diabetes in Pregnancy Screen for undiagnosed type 2 diabetes at the first prenatal visit in those with risk factors, using standard diagnostic criteria. Screen for GDM at 24–28 weeks of gestation in pregnant women not previously known to have diabetes. Screen women with GDM for persistent diabetes at 6–12 weeks postpartum, using the OGTT and nonpregnancy diagnostic criteria. Women with a history of GDM should have lifelong screening for the development of diabetes or prediabetes at least every 3 years. Further research is needed to establish a uniform approach to diagnosing GDM. 391/1/2020 Prof. Dr. RS Mehta
- 40. contd… Treatment • Dietary and life style modification • Insulin therapy 401/1/2020 Prof. Dr. RS Mehta
- 41. Other conditions or causes • Genetic defects of β-cell function • Genetic defects of insulin action (e.g. leprechaunism, lipodystrophies) • Pancreatic disease (e.g. pancreatitis, pancreatectomy, neoplastic disease, cystic fibrosis, fibrocalculous pancreatopathy) • Excess endogenous production of hormonal antagonists to insulin (e.g. growth hormone- acromegaly; glucocorticoids- Cushing's syndrome; catecholamines- phaeochromocytoma; thyroid hormones-thyrotoxicosis) 411/1/2020 Prof. Dr. RS Mehta
- 42. Contd….. • Drug-induced (e.g. corticosteroids, thiazide diuretics, phenytoin) • Viral infections (e.g. congenital rubella, mumps) • Uncommon forms of immune-mediated diabetes • Associated with genetic syndromes (e.g. Down's syndrome; Klinefelter's syndrome; Turner's syndrome 421/1/2020 Prof. Dr. RS Mehta
- 43. Complications Hypoglycemia (insulin reaction): • occurs when the blood glucose falls to less than 50 to 60 mg/dL (2.7 to 3.3 mmol/L). • caused by too much insulin or oral hypoglycemic agents, too little food, or excessive physical activity. 431/1/2020 Prof. Dr. RS Mehta
- 44. Clinical Manifestations • grouped into two categories: adrenergic symptoms and central nervous system (CNS) symptoms. In mild hypoglycemia, as the blood glucose level falls, the sympathetic nervous system is stimulated, resulting in a surge of epinephrine and norepinephrine. This causes symptoms such as sweating, tremor, tachycardia, palpitation, nervousness, and hunger. In moderate hypoglycemia • Fall in blood glucose level deprives the brain cells of needed fuel for functioning. • Signs of impaired function of the CNS may include inability to concentrate, headache, lightheadedness, confusion, memory lapses, numbness of the lips and tongue, slurred speech, impaired coordination, emotional changes, double vision, and drowsiness. • Any combination of these symptoms (in addition to adrenergic symptoms) may occur with moderate hypoglycemia. 441/1/2020 Prof. Dr. RS Mehta
- 45. Contd… In severe hypoglycemia • CNS function is so impaired that the patient needs the assistance. • Symptoms may include disoriented behavior, seizures, difficulty arousing from sleep, or loss of consciousness. • Also can cause a coma and even death. 451/1/2020 Prof. Dr. RS Mehta
- 46. Management of Hypoglycemia • Immediate treatment must be given when hypoglycemia occurs. The usual recommendation is 15 g of a fast- acting concentrated source of carbohydrate . • The equivalency of 15 grams of glucose (approximate servings) are: Three or four commercially prepared glucose tablets 4 to 6 glass of fruit juice or regular soda 6 to 10 Life Savers or other hard candies 2 to 3 teaspoons of sugar or honey • Injection of glucagon or intravenous glucose. 461/1/2020 Prof. Dr. RS Mehta
- 47. Diabetic Ketoacidosis • It is sometimes the first indication that a person has type 1 diabetes, and can be a serious complication of lack of insulin. • It occurs when the body cannot use sugar (glucose) as a fuel source because there is no insulin or not enough insulin. • Fat is used for fuel instead. When fat breaks down, waste products called ketones build up in the body. 471/1/2020 Prof. Dr. RS Mehta
- 48. Signs and symptoms of DKA • Frequent urination • Extreme thirstiness • Abdominal pain • Weight loss • Deep, rapid breathing • Fruity smell on breath (Smell of ketones being released from body) • Confusion • Weakness- Muscle stiffness or aches • Nausea and vomiting 481/1/2020 Prof. Dr. RS Mehta
- 49. Chronic or Long-term Diabetes Complications Microvascular Complications • Eye disease • Retinopathy (nonproliferative/proliferative) • Macular edema • Neuropathy • Sensory and motor (mono- and polyneuropathy) • Autonomic • Nephropathy 491/1/2020 Prof. Dr. RS Mehta
- 50. contd….. Macrovascular Complications • Coronary artery disease • Peripheral arterial disease • Cerebrovascular disease Others • Gastrointestinal (diarrhea) • Genitourinary (uropathy/sexual dysfunction) • Dermatologic- protracted wound healing and skin ulcerations • Infectious • Cataracts • Glaucoma • Periodontal disease 501/1/2020 Prof. Dr. RS Mehta
- 51. Criteria for testing for diabetes in asymptomatic adult individuals Testing should be considered in all adults who are overweight (BMI ≥25 kg/m2) and have additional risk factors: • physical inactivity • first-degree relative with diabetes • high-risk race/ethnicity (e.g., African American, Latino, Native American, Asian American) • women who delivered a baby weighing >9 lb or were diagnosed with GDM • hypertension (≥140/90 mmHg or on therapy for hypertension) • HDL cholesterol level <35 mg/dL and/or a triglyceride level >250 mg/dL • women with polycystic ovarian syndrome • other clinical conditions associated with insulin resistance (e.g., severe obesity, acanthosis nigricans) • history of CVD In the absence of the above criteria, testing for diabetes should begin at age 45 years. FPG ≥126 mg/dL (7.0 mmol/L). Fasting is defined as no caloric intake for at least 8 h. If results are normal, testing should be repeated at least at 3-year intervals, with consideration of more frequent testing depending on initial results (e.g., those with prediabetes should be tested yearly) and risk status. 511/1/2020 Prof. Dr. RS Mehta
- 52. Testing for type 2 diabetes in asymptomatic children* Criteria • Overweight (BMI >85th percentile for age and sex, weight for height >85th percentile, or weight >120% of ideal for height) Plus any two of the following risk factors: • Family history of type 2 diabetes in first- or second-degree relative • Race/ethnicity (Native American, African American, Latino, Asian American, Pacific Islander) • Signs of insulin resistance or conditions associated with insulin resistance ( hypertension, dyslipidemia, polycystic ovarian syndrome, or small-for- gestational-age birth weight) • Maternal history of diabetes or GDM during the child’s gestation Age of initiation: age 10 years or at onset of puberty, if puberty occurs at a younger age Frequency: every 3 years *Persons aged 18 years and younger. 521/1/2020 Prof. Dr. RS Mehta
- 53. Patient Education 1. Diabetes Education • Self-monitoring of blood glucose • Urine ketone monitoring (type 1 DM) • Insulin administration: Insulin Therapy and insulin preparations, types, time course of action, selecting and rotating the injection site, complications of insulin therapy (insulin lipodystrophy, local and systemic allergic reaction) • Guidelines for diabetes management during illnesses • Management of hypoglycemia • Foot and skin care • Diabetes management before, during, and after exercise and • Risk factor modifying activities. 531/1/2020 Prof. Dr. RS Mehta
- 54. contd… 2. Nutrition • Medical nutrition therapy (MNT) is a term used by the ADA to describe the optimal coordination of caloric intake with other aspects of diabetes therapy (insulin, exercise, weight loss). The ADA has issued recommendations for three types of MNT. • Primary prevention measures of MNT are directed at preventing or delaying the onset of type 2 DM in high-risk individuals (obese or with pre- diabetes) by promoting weight reduction. • Secondary prevention measures of MNT are directed at preventing or delaying diabetes related complications in diabetic individuals by improving glycemic control. • Tertiary prevention measures of MNT are directed at managing diabetes- related complications (cardiovascular disease, nephropathy) in diabetic individuals. For example, in individuals with diabetes and chronic kidney disease, protein intake should be limited to 0.8 g/kg of body weight per day. 541/1/2020 Prof. Dr. RS Mehta
- 55. Nutritional Recommendation for Adults with Diabetes Fat • 20–35% of total caloric intake • Saturated fat <7% of total calories • <200 mg/d of dietary cholesterol • Two or more servings of fish per week provide omega-3 polyunsaturated fatty acids • Minimal trans fat consumption 551/1/2020 Prof. Dr. RS Mehta
- 56. Contd… Carbohydrate • 45–65% of total caloric intake (low-carbohydrate diets are not recommended) • Amount and type of carbohydrate important (amount of carbohydrate determined by estimating grams of carbohydrate in diet; glycemic index reflects how consumption of a particular food affects the blood glucose). • Sucrose-containing foods may be consumed with adjustments in insulin dose 561/1/2020 Prof. Dr. RS Mehta
- 57. Contd… Protein • 10–35% of total caloric intake (high-protein diets are not recommended) Other components • Fiber-containing foods may reduce postprandial glucose • Non nutrient sweeteners Alcohol 571/1/2020 Prof. Dr. RS Mehta
- 58. Contd… 3. Exercise • For individuals with type 1 or type 2 DM, exercise is also useful for lowering plasma glucose (during and following exercise) and increasing insulin sensitivity. • In patients with diabetes, the ADA recommends 150 min/ week (distributed over at least 3 days) of aerobic physical activity. • In patients with type 2 DM, the exercise regimen should also include resistance training (weight lifting) 581/1/2020 Prof. Dr. RS Mehta
- 59. Guidelines for ongoing medical care for patients with diabetes • Self-monitoring of blood glucose (individualized frequency) • A1C testing (two to four times per year) • Patient education in diabetes management (annual) • Medical nutrition therapy and education (annual) • Eye examination (annual) • Foot examination (one to two times per year by physician; daily by patient) • Screening for diabetic nephropathy (annual) • Blood pressure measurement (quarterly) • Lipid profile and serum creatinine (estimate GFR) (annual) • Influenza/pneumococcal immunizations • Consider antiplatelet therapy 591/1/2020 Prof. Dr. RS Mehta
- 60. Nursing Management Assessment 1. Obtain a history of current problems, family history, and general health history. • Has the patient experienced polyuria, polydipsia, polyphagia, and any other symptoms? – Number of years since diagnosis of diabetes – Family members diagnosed with diabetes, their subsequent treatment, and complications 601/1/2020 Prof. Dr. RS Mehta
- 61. Contd… 2. Perform a review of systems and physical examination to assess for signs and symptoms of diabetes, general health of patient, and presence of complications. – General: recent weight loss or gain, increased fatigue, tiredness, anxiety – Skin: skin lesions, infections, dehydration, evidence of poor wound healing – Eyes: changes in vision- floaters, halos, blurred vision, dry or burning eyes, cataracts, glaucoma – Mouth: gingivitis, periodontal disease – Cardiovascular: orthostatic hypotension, cold extremities, weak pedal pulses, leg claudication – GI: diarrhea, constipation, early satiety, bloating, increased flatulence, hunger or thirst – Genitourinary (GU): increased urination, nocturia, impotence, vaginal discharge – Neurologic: numbness and tingling of the extremities, decreased pain and temperature perception, changes in gait and balance 611/1/2020 Prof. Dr. RS Mehta
- 62. Nursing Diagnosis • Imbalanced Nutrition: More than Body Requirements related to intake in excess of activity expenditures • Fear related to insulin injection • Risk for Injury (hypoglycemia) related to effects of insulin, inability to eat • Activity Intolerance related to poor glucose control • Deficient Knowledge related to use of oral hypoglycemic agents • Risk for Impaired Skin Integrity related to decreased sensation and circulation to lower extremities • Ineffective Coping related to chronic disease and complex self-care regimen 621/1/2020 Prof. Dr. RS Mehta
- 63. Nursing Interventions Improving Nutrition • Assess current timing and content of meals. • Advise patient on the importance of an individualized meal plan in meeting weight-loss goals. • Discuss the goals of dietary therapy for the patient. • Setting a goal of a 10% (of patient's actual body weight) weight loss over several months, reducing blood sugar and other metabolic parameters. • Explain the importance of exercise in maintaining/reducing body weight. 631/1/2020 Prof. Dr. RS Mehta
- 64. Teaching about insulin • Assist patient to reduce fear of injection by encouraging verbalization of fears regarding insulin injection, and identifying supportive coping techniques. • Demonstrate and explain thoroughly the procedure for insulin self-injection. • Help patient to master technique by taking a step-by-step approach. – Allow patient time to handle insulin and syringe to become familiar with the equipment. – Teach self-injection first to alleviate fear of pain from injection • Review dosage and time of injections in relation to meals, activity, and bedtime based on patient's individualized insulin regimen 641/1/2020 Prof. Dr. RS Mehta
- 65. Improving activity tolerance • Advise patient to assess blood glucose level before and after strenuous exercise. • Instruct patient to plan exercises on a regular basis each day. • Encourage patient to eat a carbohydrate snack before exercising to avoid hypoglycemia. • Advise patient that prolonged strenuous exercise may require increased food at bedtime to avoid nocturnal hypoglycemia. • Instruct patient to avoid exercise whenever blood glucose levels exceed 250 mg/day and urine ketones are present. Patient should contact health care provider if levels remain elevated. 651/1/2020 Prof. Dr. RS Mehta
- 66. 661/1/2020 Prof. Dr. RS Mehta
