Radiation
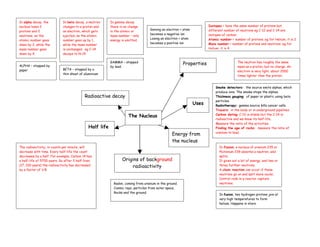
- 1. In alpha decay, the In beta decay, a neutron In gamma decay nucleus loses 2 changes to a proton and there is no change Isotopes = have the same number of protons but Gaining an electron = atom different number of neutrons eg C-12 and C-14 are protons and 2 an electron, which gets in the atomic or becomes a negative ion isotopes of carbon neutrons, so the ejected, so the atomic mass number – only Losing an electron = atom Atomic number = number of protons. eg for Helium, it is 2 atomic number goes number goes up by 1, energy is emitted. becomes a positive ion Mass number = number of protons and neutrons. eg for down by 2, while the while the mass number mass number goes is unchanged. eg C-14 Helium, it is 4 down by 4. decays to N-14 GAMMA – stopped ALPHA – stopped by Properties The neutron has roughly the same by lead. mass as a proton, but no charge. An paper BETA – stopped by a electron is very light, about 2000 thin sheet of aluminium times lighter than the proton. Smoke detectors: the source emits alphas, which produce ions. The smoke stops the alphas. Radioactive decay Thickness gauging: of paper or plastic using beta particles. Uses Radiotherapy: gamma source kills cancer cells. Tracers: in the body or in underground pipelines The Nucleus Carbon dating: C-12 is stable but the C-14 is radioactive and we know its half-life. Measure the ratio of the activities. Half life Finding the age of rocks: measure the ratio of uranium to lead. Energy from the nucleus The radioactivity, in counts per minute, will In fission, a nucleus of uranium 235 or decrease with time. Every half-life the count Plutonium 239 absorbs a neutron, and decreases by a half. For example, Carbon 14 has splits. a half-life of 5700 years. So after 3 half-lives Origins of background It gives out a lot of energy, and two or (17, 100 years) the radioactivity has decreased radioactivity three further neutrons. by a factor of 1/8 A chain reaction can occur if those neutrons go on and split more nuclei. Control rods in a reactor capture Radon, coming from uranium in the ground. neutrons. Cosmic rays, particles from outer space. Rocks and the ground. In fusion, two hydrogen protons join at very high temperatures to form helium. Happens in stars.