Posters for Exhibition
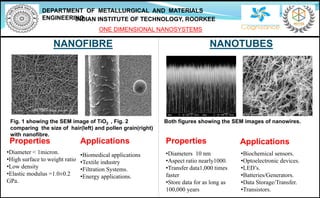
- 1. DEPARTMENT OF METALLURGICAL AND MATERIALS ENGINEERINGINDIAN INSTITUTE OF TECHNOLOGY, ROORKEE ONE DIMENSIONAL NANOSYSTEMS NANOFIBRE NANOTUBES Properties •Diameter < 1micron. •High surface to weight ratio •Low density •Elastic modulus =1.0±0.2 GPa. •Diameters 10 nm •Aspect ratio nearly1000. •Transfer data1,000 times faster •Store data for as long as 100,000 years Fig. 1 showing the SEM image of TiO2 , Fig. 2 comparing the size of hair(left) and pollen grain(right) with nanofibre. PropertiesApplications •Biomedical applications •Textile industry •Filtration Systems. •Energy applications. •Biochemical sensors. •Optoelectronic devices. •LED’s. •Batteries/Generators. •Data Storage/Transfer. •Transistors. Applications Both figures showing the SEM images of nanowires.
- 2. DEPARTMENT OF METALLURGICAL AND MATERIALS ENGINEERINGINDIAN INSTITUTE OF TECHNOLOGY, ROORKEE ONE DIMENSIONAL NANOSYSTEMS NANOFIBRES NANOWIRES Properties • Dimensions from 1-100 nm. •Aspect Ratio from 3-5. •Reflectivity changes by changing the orientation with an applied electric field. •Generate heat when excited with IR and many more. •Diameter of few nanometers. •NTs are hollow. •High Electrical Conductivity. •Very High Tensile Strength. •Highly Flexible- can be bent without damage. Fig. 1 showing the SEM image of ZnO Nanorods and Fig. 2 depicting gold Nanorods . PropertiesApplication •Display Technologies •Microelectromechanical Systems(MEMS) •Cancer Therapy •NRs from Semiconducting materials are used as LEDs. •Field Emission •Energy Storage •Thermal Materials •Biomedical Applications •Structural Applications •Molecular Electronics Application Fig. 3 showing Single and multi walled Carbon Nanotubes and Fig. 4 depicting the view of CNT at different magnifications.
- 3. SHAPE MEMORY ALLOYS Shape memory alloys (SMAs) are a particular group of metallic materials exhibiting unique properties, mainly represented by superelastic and shape memory effects. Superelasticity is the capability of recovering the original shape after large deformations (large strains can be undergone, until 8- 10%) induced by mechanical loading. Shape memory effect is the ability to recover the original shape from a deformed shape up on heating above austenite finish temperature. Superelasticity. When the alloy is mechanically loaded at a temperature higher than austenite finish temperature, superelasticity can be exhibited. If the value of applied stress is sufficiently high, austenite to martensite transformation is induced. Upon unloading, inverse martensite to austenite transformation occurs, because austenite is the stable phase at higher temperature, and original shape is recovered. DEPARTMENT OF METALLURGICAL AND MATERIALS ENGINEERINGINDIAN INSTITUTE OF TECHNOLOGY, ROORKEE SHAPE MEMORY ALLOYS
- 4. DEPARTMENT OF METALLURGICAL AND MATERIALS ENGINEERINGINDIAN INSTITUTE OF TECHNOLOGY , ROORKEEAPPLICATIONS OF SUPERCONDUCTORS 1. Superconducting Generators 2. Superconducting transmission cables 3. Magnetic Resonance Imaging 3. Superconducting Solar Water Heater 4. Superconducting Speakers 4. Magnetic Levitation Trains
- 5. DEPARTMENT OF METALLURGICAL AND MATERIALS ENGINEERING INDIAN INSTITUTE OF TECHNOLOGY, ROORKEE SUPERCONDUCTIVITY AND SUPERCONDUCTORS The Delhi iron pillar is testimony to the high level of skill achieved by ancient Indian iron smiths in the extraction and processing of iron. The iron pillar at Delhi has attracted the attention of archaeologists and corrosion technologists as it has withstood corrosion for the last 1600 years. Introduction Description : The height of the pillar, from the top of its capital to the bottom of its base, is 7.21 m (23 ft 8 in), 1.12 m (3 ft 8 in) of which is below ground. Its bell pattern capital is 1.07 m (3 ft 6 in) in height, and its bulb-shaped base is 0.71 m (2 ft 4 in) high. The base rests on a grid of iron bars soldered with lead into the upper layer of the dressed stone pavement. The pillar's lower diameter is 420 mm (17 in), and its upper diameter 306 mm (12 in). It is estimated to weigh more than six tonnes (13,228 lb).
- 6. DEPARTMENT OF METALLURGICAL AND MATERIALS ENGINEERING INDIAN INSTITUTE OF TECHNOLOGY, ROORKEE THE DELHI IRON PILLAR The Delhi iron pillar is testimony to the high level of skill achieved by ancient Indian iron smiths in the extraction and processing of iron. The iron pillar at Delhi has attracted the attention of archaeologists and corrosion technologists as it has withstood corrosion for the last 1600 years. Introduction : Description : The height of the pillar, from the top of its capital to the bottom of its base, is 7.21 m (23 ft 8 in), 1.12 m (3 ft 8 in) of which is below ground. Its bell pattern capital is 1.07 m (3 ft 6 in) in height, and its bulb-shaped base is 0.71 m (2 ft 4 in) high. The base rests on a grid of iron bars soldered with lead into the upper layer of the dressed stone pavement. The pillar's lower diameter is 420 mm (17 in), and its upper diameter 306 mm (12 in). It is estimated to weigh more than six tonnes (13,228 lb). The corrosion resistant property : The several theories which have been proposed to explain its superior corrosion resistance can be broadly classified into two categories: the environmental and the material theories. Proponents of the environmental theories state that the mild climate of Delhi is responsible for the corrosion resistance of the Delhi iron pillar. It is known that the relative humidity at Delhi does not exceed 70% for significant periods of time in the year, which therefore results in very mild corrosion of the pillar. On the other hand, several investigators have stressed the importance of the material of construction as the primary cause for the pillar's corrosion resistance. The ideas proposed in this regard are the relatively pure composition of the iron used, presence of Phosphorus (P) and absence of Sulphur/Magnesium in the iron, its slag-enveloped metal grain structure, and passivity enhancement in the presence of slag particles Microstructure : Fig. Showing the bell pattern at the top of Delhi Iron Pillar. Fig. Showing the complete view of Delhi Iron Pillar The microstructure of the iron of the Delhi iron pillar is typical of wrought iron. Iron was produced in ancient times by solid-state reduction of iron ore using charcoal and after the reduction process, the slag particles in iron were squeezed out by hammering. This invariably resulted in the presence of slag particles and unreduced iron oxide in the microstructure.. However, the role of P in the passivation process was not understood. The characterization of the Delhi iron pillar rust has provided clear ideas about the passive film formation process on the Delhi iron pillar. A detailed rust analysis using modern sophisticated characterization techniques like Mössbauer spectroscopy and Fourier transform infrared spectroscopy (FTIR) has been done. The FTIR spectrum proved the presence of g-FeOOH, a-FeOOH and d-FeOOH. The d-FeOOH was the major component of the rust as the peak was of relatively larger height compared to the others. An interesting result from the FTIR spectrum was that there was a distinct signal from the phase FePO4.2H2O and the shoulder from this phase was also identifiable. Therefore, the results of the FTIR study indicated that the constituents of the scale were g, a and d-FeOOH, in addition to a small amount of FePO4. In order to further understand the nature of the rust, the Mössbauer spectrum obtained from the rust in the transmission mode was analysed. The presence of g-FeOOH, a-FeOOH and d-FeOOH in superparamagnetic form was confirmed. The very fine particle size of these oxyhydroxides was also confirmed. The presence of iron phosphate was also confirmed. Finally, the rust was also composed of magnetite that was incorporated with some ions. The process of passive film formation on the ancient Indian iron can be visualized as follows. Initially, the corrosion of the metal leads to the formation of a- and g-FeOOH. However, the presence of slag particles accelerates the corrosion of iron thereby enhancing the P concentration on the surface. This enhancement of P on the surface catalyses the formation of amorphous d-FeOOH as a compact layer next to the surface and this results in atmospheric corrosion resistance of the Delhi iron pillar. With time, conversion of this d-FeOOH to a stable form of iron oxide, i.e., magnetite, is possible. The magnetite could be doped with ions. This would further enhance the corrosion resistance of the surface film on the surface. The FTIR and Mössbauer spectra indicate the presence of iron phosphates. The presence of these phosphates would provide further corrosion resistance to the passive film by lowering ionic diffusion in the oxide and also by blocking the pores in the oxide. The golden hue of the pillar when viewed in certain orientations is due to the presence of iron phosphates Process of Protective Rust Formation : Rust Analysis : Fig. Showing the Surface Film Characteristic of the Delhi Iron Pillar
- 7. DEPARTMENT OF METALLURGICAL AND MATERIALS ENGINEERING INDIAN INSTITUTE OF TECHNOLOGY, ROORKEE SHAPE MEMORY ALLOYS AND THEIR APPLICATIONS SHAPE MEMORY ALLOYS Shape memory alloys (SMAs) are a particular group of metallic materials exhibiting unique properties, mainly represented by superelastic and shape memory effects. Superelasticity is the capability of recovering the original shape after large deformations (large strains can be undergone, until 8-10%) induced by mechanical loading. Superelasticity. When the alloy is mechanically loaded at a temperature higher than austenite finish temperature, superelasticity can be exhibited. If the value of applied stress is sufficiently high (i.e. over the value σAM in the graph), austenite to martensite transformation is induced. Upon unloading, inverse martensite to austenite transformation occurs, because austenite is the stable phase at higher temperature, and original shape is recovered. Shape memory effect is the ability to maintain a deformed shape up to heat induced recovery of the original shape. Shape memory effect. When the material is mechanically deformed at a temperature lower than martensite finish temperature, a transformation from multi-variant to single-variant martensite occurs, if the stress is higher than a determinate value (σMM in the figure). Upon unloading, a residual deformation remains. When the material, in an unloaded state, is heated above austenite finish temperature, the transformation from martensite to austenite allows shape recovery (shape memory effect). Upon cooling, most SMAs show no macroscopic deformation and the original shape is maintained. SMA particular characteristics allow a variety of innovative applications in different engineering fields, such as biomedical (e.g. stents, surgery tools, active implants), structural (e.g. damping devices), automotive (e.g. actuators), aeronautical (e.g. the Variable Geometry Chevron), mechanical (e.g. heat engines, coupling devices) and robotic (e.g. SMA actuated microrobots) engineering. Applications of Shape Memory Alloys 1. SMA Microgripper 2. SMA helical spring actuator 3. 3. SM eyeglass frame 4. SM Actuator Shape memory eyeglass frame. Shape memory eyeglass frame is a real life example of superelastic behavior. It can undergo severe deformation and still recover original shape. 5. Coupling for Tubing 6. Flexible Nitinol wires Wires have the ability to flex the robotic muscles according to electric pulses sent through the wire. Use of memory alloys for coupling tubing: A memory alloy coupling is expanded (a) so it fits over the tubing (b). When the coupling is reheated, it shrinks back to its original diameter (c), squeezing the tubing for a tight fit Spring actuator. A) initial geometry; B)deformed shape due to the weight application at T=-25 C; C) spring shape recovery and weight lifting due to heating to T=100 C; D) spring stretching due to cooling to T=-25 C Shape memory actuator vs. EM actuator. Few centimeters of SMA wire can replace bulky electromagnetic (EM) actuators for automotive applications. Simulation of micro-gripper with a sample. a) starting configuration; b) memorization step; c) heating of linear actuator and sample gripping; d) heating of rotary actuator with consequent re- opening. von Mises stress [MPa] distribution in the micro-gripper are depicted in the contour plot.
- 8. DEPARTMENT OF METALLURGICAL AND MATERIALS ENGINEERINGINDIAN INSTITUTE OF TECHNOLOGY, ROORKEESUPERCONDUCTIVITY AND SUPERCONDUCTORS Superconductivity is the ability of certain materials to conduct electric current having practically zero resistance below a certain temperature (Tc), magnetic field (Bc), and applied current(Jc). Those materials are called as superconductors. Definition: Properties of Superconductors : • Meissner Effect: Exhibiting diamagnetic properties to the total exclusion of all magnetic fields. This is a classic hallmark of superconductivity and can actually be used to levitate a strong rare-earth magnet. • Divided into two categories: Type-I superconductors repel a magnetic field (the Meissner effect). If the strength of the magnetic field increases, they lose their superconductivity. This does not happen with type-II superconductors, which accommodate strong magnetic fields by letting the magnetic field in. • Virtually zero electrical resistance. • Perfect diamagnetic property. • Critical field depends upon temperature of superconducting material. • At very high pressure Tc is directly proportional to pressure. • Tc is inversely proportional to square root of At.wt of the isotope of single superconductor. History : • In 1911 superconductivity was first observed in mercury by Dutch physicist Heike Kamerlingh Onnes of Leiden University. When he cooled it to the temperature of liquid helium, 4 degrees Kelvin, its resistance suddenly disappeared! • In 1933 Walter Meissner and Robert Ochsenfeld discovered that a superconducting material will repel a magnetic field. This phenomenon is known as perfect diamagnetism and is often referred to as the Meissner effect. Example : YBa2Cu307 • Discovered: 1987 by Paul Chu • Tc: 90-95K • Bc2: 100 Tesla at 77 K • Jc: 1.0x109 A/m2 at 77 K • Referred to as “1-2-3” superconductor because of the because of the ratio of the three metallic elements. • Type: Type II Ceramic Structure of YBa2Cu307 Type 1 and 2 Superconductors The Meissner EffectDifference between SC and non-SC Applications of Superconductors 1. Superconducting Generators 2. Superconducting transmission cables 3. Superconducting Magnetic Energy Storage System (SMES) 3. Superconducting Tube Direct-Plug Solar Water Heater 3. Superconducting Speakers 4. Magnetic Levitation Trains
