Phase changes
•Als PPT, PDF herunterladen•
5 gefällt mir•18,150 views
Melden
Teilen
Melden
Teilen
Weitere ähnliche Inhalte
Was ist angesagt?
Was ist angesagt? (20)
Ähnlich wie Phase changes
Ähnlich wie Phase changes (20)
Redesign- Make a prediction relating heat to the movement of molecules.docx

Redesign- Make a prediction relating heat to the movement of molecules.docx
PHYSICAL CHANGES IN TERMS OF THE arrangement and.pptx

PHYSICAL CHANGES IN TERMS OF THE arrangement and.pptx
0708_matter and classification,states, phases of matter.ppt

0708_matter and classification,states, phases of matter.ppt
2.1 form 3 simple kinetic molecular model of matter

2.1 form 3 simple kinetic molecular model of matter
ICSE Class VIII Chemistry Matter Around Us - TopperLearning

ICSE Class VIII Chemistry Matter Around Us - TopperLearning
Mehr von Grover Cleveland Middle School
Mehr von Grover Cleveland Middle School (20)
Ocean acidification will-the_reef_survive-non-narrated[1]![Ocean acidification will-the_reef_survive-non-narrated[1]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Ocean acidification will-the_reef_survive-non-narrated[1]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Ocean acidification will-the_reef_survive-non-narrated[1]
Ocean acidification will-the_reef_survive-non-narrated[1]![Ocean acidification will-the_reef_survive-non-narrated[1]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Ocean acidification will-the_reef_survive-non-narrated[1]](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Ocean acidification will-the_reef_survive-non-narrated[1]
Kürzlich hochgeladen
Kürzlich hochgeladen (20)
A Journey Into the Emotions of Software Developers

A Journey Into the Emotions of Software Developers
Why device, WIFI, and ISP insights are crucial to supporting remote Microsoft...

Why device, WIFI, and ISP insights are crucial to supporting remote Microsoft...
Testing tools and AI - ideas what to try with some tool examples

Testing tools and AI - ideas what to try with some tool examples
Assure Ecommerce and Retail Operations Uptime with ThousandEyes

Assure Ecommerce and Retail Operations Uptime with ThousandEyes
How to Effectively Monitor SD-WAN and SASE Environments with ThousandEyes

How to Effectively Monitor SD-WAN and SASE Environments with ThousandEyes
Genislab builds better products and faster go-to-market with Lean project man...

Genislab builds better products and faster go-to-market with Lean project man...
Transcript: New from BookNet Canada for 2024: Loan Stars - Tech Forum 2024

Transcript: New from BookNet Canada for 2024: Loan Stars - Tech Forum 2024
The Ultimate Guide to Choosing WordPress Pros and Cons

The Ultimate Guide to Choosing WordPress Pros and Cons
Use of FIDO in the Payments and Identity Landscape: FIDO Paris Seminar.pptx

Use of FIDO in the Payments and Identity Landscape: FIDO Paris Seminar.pptx
The Future Roadmap for the Composable Data Stack - Wes McKinney - Data Counci...

The Future Roadmap for the Composable Data Stack - Wes McKinney - Data Counci...
Passkey Providers and Enabling Portability: FIDO Paris Seminar.pptx

Passkey Providers and Enabling Portability: FIDO Paris Seminar.pptx
Time Series Foundation Models - current state and future directions

Time Series Foundation Models - current state and future directions
Long journey of Ruby standard library at RubyConf AU 2024

Long journey of Ruby standard library at RubyConf AU 2024
TeamStation AI System Report LATAM IT Salaries 2024

TeamStation AI System Report LATAM IT Salaries 2024
Phase changes
- 2. Do Now: What are the names for the phases that matter goes through when changing from solid to liquid to gas, and then back again?
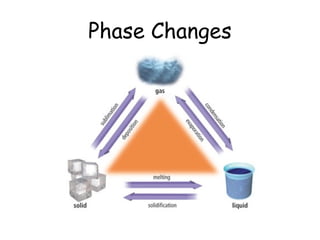
- 3. States of Matter - Review By adding energy, usually in the form of heat, the bonds that hold a solid together can slowly break apart. The object goes from one state, or phase, to another. These changes are called PHASE CHANGES.
- 4. Solids A solid is an ordered arrangement of particles that have very little movement. The particles vibrate back and forth but remain closely attracted to each other.
- 5. Liquids A liquid is an arrangement of less ordered particles that have gained energy and can move about more freely. Their attraction is less than a solid.
- 6. Compare the two states:
- 7. Gases By adding energy, solids can change from an ordered arrangement to a less ordered arrangement (liquid), and finally to a very random arrangement of the particles (the gas phase).
- 8. Each phase change has a specific name, based on what is happening to the particles of matter.
- 9. Melting and Freezing The solid, ordered molecules gain energy, collide more, and become less ordered. The less ordered liquid molecules loose energy, slow down, and become more ordered.
- 10. Condensation and Vaporization The less ordered gas molecules loose energy, slow down, and become more ordered. The more ordered liquid molecules gain energy, speed up, and become less ordered. Evaporation only occurs at the surface of a liquid. Boiling occurs throughout the liquid.
- 11. Sublimation and Deposition The more ordered solid gains energy, the molecules speed up and become less ordered gas. Liquid phase is bypassed. The less ordered gas looses energy, molecules slow down, and become a solid (frost). Liquid phase is bypassed
- 12. Phase changes are classified as one of two types: 1. ENDOTHERMIC – The object absorbs energy from the surroundings. Think of ENDO as “entering”: energy comes into the object. What is happening to the particles? 2. EXOTHERMIC – The object releases energy to the surroundings. Think of EXO as “exiting”: energy goes out of the object. What is happening to the particles now?
- 13. Now we can complete the remaining two columns of the study guide.