Electronegativity Difference
•Als PPT, PDF herunterladen•
1 gefällt mir•379 views
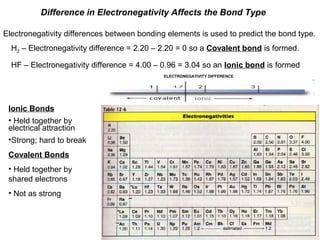
Chart depicting the electronegativity differences of atoms, including definitions and examples of ionic and covalent bonding in the context of electronegativity.
Melden
Teilen
Melden
Teilen
Empfohlen
Weitere ähnliche Inhalte
Was ist angesagt?
Was ist angesagt? (20)
Mehr von Emma Wise
Mehr von Emma Wise (20)
Cleveland's Got It All CD and the Greater Cleveland Food Bank 

Cleveland's Got It All CD and the Greater Cleveland Food Bank
How to Help the Humane Society of Central Illinois

How to Help the Humane Society of Central Illinois
Kürzlich hochgeladen
USPS® Forced Meter Migration - How to Know if Your Postage Meter Will Soon be...

USPS® Forced Meter Migration - How to Know if Your Postage Meter Will Soon be...Postal Advocate Inc.
Kürzlich hochgeladen (20)
Global Lehigh Strategic Initiatives (without descriptions)

Global Lehigh Strategic Initiatives (without descriptions)
Barangay Council for the Protection of Children (BCPC) Orientation.pptx

Barangay Council for the Protection of Children (BCPC) Orientation.pptx
How to do quick user assign in kanban in Odoo 17 ERP

How to do quick user assign in kanban in Odoo 17 ERP
Difference Between Search & Browse Methods in Odoo 17

Difference Between Search & Browse Methods in Odoo 17
ENG 5 Q4 WEEk 1 DAY 1 Restate sentences heard in one’s own words. Use appropr...

ENG 5 Q4 WEEk 1 DAY 1 Restate sentences heard in one’s own words. Use appropr...
AUDIENCE THEORY -CULTIVATION THEORY - GERBNER.pptx

AUDIENCE THEORY -CULTIVATION THEORY - GERBNER.pptx
ENGLISH 7_Q4_LESSON 2_ Employing a Variety of Strategies for Effective Interp...

ENGLISH 7_Q4_LESSON 2_ Employing a Variety of Strategies for Effective Interp...
MULTIDISCIPLINRY NATURE OF THE ENVIRONMENTAL STUDIES.pptx

MULTIDISCIPLINRY NATURE OF THE ENVIRONMENTAL STUDIES.pptx
USPS® Forced Meter Migration - How to Know if Your Postage Meter Will Soon be...

USPS® Forced Meter Migration - How to Know if Your Postage Meter Will Soon be...
Electronegativity Difference
- 1. Difference in Electronegativity Affects the Bond Type Electronegativity differences between bonding elements is used to predict the bond type. H2 – Electronegativity difference = 2.20 – 2.20 = 0 so a Covalent bond is formed. HF – Electronegativity difference = 4.00 – 0.96 = 3.04 so an Ionic bond is formed ELECTRONEGATIVITY DIFFERENCE Ionic Bonds • Held together by electrical attraction •Strong; hard to break Covalent Bonds • Held together by shared electrons • Not as strong
