Cell injury-necrosis
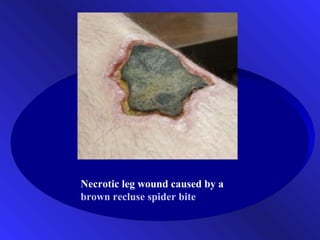
- 1. Necrotic leg wound caused by a brown recluse spider bite
- 2. IRREVERSIBLE CELL INJURY Necrosis NECROSIS Definition: Necrosis Necrosis ("death, the stage of dying, the act of killing") is the premature death of cells in living tissue. Necrosis is caused by factors external to the cell or tissue, such as infection, toxins, or trauma. This is in contrast to Apoptosis, which is a naturally occurring cause of cellular death. While apoptosis often provides beneficial effects to the organism, necrosis is almost always detrimental and can be fatal.
- 3. Causes of cell necrosis: See before, but the most common causes of cell death are viruses, ischaemia, bacterial toxins, hypersensitivity, and ionizing radiation. Morphologic change in necrosis: The changes don’t appear in the affected cells by light microscopy before 2-6 hours according to the type of the affected tissue.
- 4. Chang e s in the cyto plasm : i. Swelling and granularity of the cytoplasm due to (a) Imbibition of water (↓ATP in the cell) (b) Coagulation of the cytoplasm. ii. Loss of cellular membrane and release of intracellular enzymes in the blood e.g. Serum Transaminase(ST )& LDH where their detection is of diagnostic value in liver and heart diseases. iii. Fusion of cells forming homogeneous eosinophilic mass.
- 6. N ar chang e s: ucle Occur due to hydrolysis of nucleoproteins: i. Pyknosis i.e. the nucleus becomes shrunken condensed and deeply stained. ii. Karyorrhexis: rupture of nuclear membrane with fragmentation of the nucleus. iii. Karyolysis: the nucleus dissolves and disappears. Finally the affected tissue changes to homogeneous eosinophilic mass with nuclear debris.
- 7. NUCLEAR CHANGES IN NECROSIS
- 8. Ty of necrosis pes The variable types of necrosis differ as regards causes, gross and microscopic pictures. (1) Coagulative necrosis: It is mainly caused by sudden ischaemia e.g. infarction of heart, kidney and spleen. The protein of the affected tissue becomes denaturated. Grossly, it appears dry pale opaque. It is triangular ? subcapsular with the base towards the capsule of the affected organ. This is due to the fan like distribution of the supplying blood vessels. The infarct area is surrounded by narrow zone of inflammation and congestion. Microscopically, the structural outline of the affected tissue is preserved but the cellular details are lost.
- 9. GROSS AND MICROSCOPIC PICTURE OF NECROSIS
- 10. (2) Liquifactive necrosis The necrosed tissue undergoes rapid softening e.g. infarction of the nervous tissue which has abundant lysosomal enzymes. Also, this type of necrosis occurs in case of suppurative inflammation (Abscess) where liquefaction occurs under the effect of proteolytic enzymes of PNLs liquefaction of the amoebic abscess occurs due to the effect of strong proteolytic enzymes and hyaluronidase secreted by E. Histolytica. Grossly: the affected tissue appears as homogenous amorphous substance. Microscopically: it appears as homogenous eosinophilic structure.
- 12. (3) Caseous necrosis: • It is characteristic of tuberculosis. The necrotic tissue undergoes slow partial liquefaction forming yellow cheesy material. • Microscopically, it shows amorphous granular eosinophilic material lacking the cell outlines. • Unlike coagulative necrosis, the necrotic cells do not retain their cellular outlines, and do not disappear by lysis, as in liquifactive necrosi • Grossly, the caseous material resembles clumpy cheese, hence the name caseous necrosis. • The cause of necrosis in TB is hypersensitivity reaction caused by the tuberculoprotein
- 13. Caseous necrosis in LN. Amorphous ,granular ,eosinophilic ,necrotic center is surrounded by granulomatous inflammation.
- 14. T.B LUNG : (Large Area Of Caseous Necrosis) AREA ,YELLOW-WHITE AND CHESSY
- 15. (4) Fat ne cro sis it is necrosis of adipose tissue including two types: a) Traumatic: caused by trauma to adipose tissue e.g. breast and subcutaneous tissue. b) Enzymatic: which occurs in case of acute haemorrhagic pancreatitis. Obstruction of the pancreatic duct leads to release of lipase which splits the fat cells of the omentum into fatty acid (combine with Ca giving chalky white calcification) and to glycerol which is absorbed in the circulation.
- 16. (5) Fibrino id ne cro sis This is characterized by swelling, fragmentation, increased eosinophilia of collagen fibers and accumulation of mucopolysaccharides and fibrin due to vascular exudation of fibrinogen at the site of lesion, e.g.: a) Collagen diseases (Rheumatic fever, Rheumatoid, Sclerodermia, Lupus erythematosus and Polyarteritis nodosa). b) In the wall of blood vessels in malignant hypertension
- 18. (6 ) Ze nke r’s ne cro sis: Of the rectus abdominus muscle and diaphragm as a complication of : bacterial infection particularly typhoid fever. The striated muscles lose its striation, swell and fuse together in homogeneous structureless mass.
- 19. (7 ) Gang re no us ne cro sis: The tissue in this case have undergone ischaemic cell death and coagulative necrosis followed by liquifactive action of putrefactive organisms. When coagulative pattern is dominant the process is termed dry gangrene. When the liquifactive action of the bacteria is more pronounced it is called wet gangrene.
- 20. Obtraction of the bloodsupplytoto bowelis almost followed by Obstruction of blood supply the bowel is alrmost followed by gangrene. Gangrene
- 21. Fate and local effects OF NECROSIS : 1. A small area undergoes repair: A) The products of the necrotic cells irritate the surrounding tissue forming a zone of inflammation. B) The accumulated neutrophils in the zone of inflammation soften the necrotic tissue and make its removal by macrophages and blood stream easy and help the process of healing. C) Repair by regeneration or fibrosis depends upon the type of cells affected (labile-stable- permanent).
- 22. 2. If the necrotic area is wide, its products can’t be removed and a fibrous capsule form around it in order to separate it from the living tissue. Areas of necrotic softening in the brain become surrounded by proliferated neuroglia (gliosis). 3. Old unabsorbed caseous lesions and fat necrosis usually becomes heavily calcified (dystrophic calcification). 4-when the necrotic tissue is infected with putrefactive Organism------Gangrine
- 23. General effects of necrosis 1. Release of enzymes from the breakdown tissue into the blood forms the basis of clinical tests for diagnosis e.g. detection of transamenase in myocardial infarction and liver necrosis in hepatitis. 2. Absorption of dead products into
- 25. N.B.: I. Necrosis of: 1) Of small groups of cells is called focal necrosis. 2) Of large groups of cells is called confluent necrosis. 3) Of extensive areas of an organ is called massive necrosis. II. Somatic death means death of the individual. III. Post-mortum autolysis of the tissue occurring after death can be differentiated from necrosis by the absence of inflammatory zone around the affected tissue (inflammation
- 26. A pto sis po Definition: • It is programmed death of cells in living tissues. It is an active process differing from necrosis by the following points: • Occurs in both physiological and pathological conditions. • Starts by nuclear changes in the form of chromatin condensation and fragmentation followed by cytoplasmic budding and then phagocytosis of the extruded apoptotic bodies. • Plasma membrane are thought to remain intact during apoptosis until the last stage so does not initiate inflammatory reaction around it.
- 28. Death by Committing Suicide AP T OP OSIS While committing suicide cells: Shrink Chromatin Degraded Mitochondria Break Down Break into Membrane-bound Fragments Phosphatidylserine Exposed Phagocytic Receptors
- 29. Microscopically: In the tissue stained with H & E apoptosis affects single or small clusters of cells and apoptotic cell appears as round mass of intensely eosinophilic cytoplasm with dense nuclear chromatin fragments.
- 30. Major criteria of Apoptosis 1- Morphological changes 2- Chromatin condensation 3- DNA fargmentation 4- Cell death
- 32. Initiation for apoptosis Factors predisposes to apoptosis 1- Nontoxic stimuli can lead to apoptosis . 2- loss of growth factors. 3- Direct action of cytokines (e.g., tumor necrosis factor) 4- Immune system action (e.g., natural killer cells or cytotoxic T lymphocytes). 5- Viral infection. 6- Adult tissue homeostasis . 7- Sublethal damage to the cells (e.g., by ionizing radiation, hyperthermia, toxins.) 8- Loss of cell-cell or cell-matrix attachments.
- 33. Examples of physiologic and pathologic cases accompanied with apoptosis: 1. Programmed cell death during embryogenesis. 2. Hormone dependent cell involution in case of endometrial cell break down during menstrual cycle. 3. Cell death in tumours during regression induced by cytotoxic drugs or irradiation. 4. In some viral disease e.g. viral hepatitis in which apoptotic cells are known as councilman bodies.
- 34. ROL (VAL )OF AP T E UE OP OSIS 1-Defense: Against nonself multicellular organisms (cell commits suicide when infected by a virus may protect other cells from further spread of the virus) 2-Digestion: The cellular DNA destroy the genetic information of any external stimulus. 3-Protecting: Protection of the organism from unregulated
- 35. Apoptosis vs. Necrosis Apoptosis Necrosis • Cells shrink and condense • Cells swell and burst, • Release small membrane releasing their intracellular bound bodies contents • Small fragments are • Damaging to surrounding engulfed by surrounding cells cells • Causes inflammation
- 36. RE RSIB E CE L INJ VE L L URY Hydropic swelling Hydropic swelling is an acute stress cell injury caused by a variety of agents leading to swelling in the cells. Pathogenesis: Hydropic swelling results from impairment of the process controlling ionic sodium concentration in the cytoplasm. This regulation is controlled by: (i) Plasma membrane itself, (ii) Plasma membrane sodium pump, (iii) The supply of ATP. Injurious agents may interfere with one of these factors leading to intracellular accumulation of sodium and increase water to maintain isosmotic condition of the cell. The result is
- 37. Structural changes: Grossly, the affected organ increases in size becomes pale, bloodless, having sharp edge which bulge over the capsule on cut section of that organ. Microscopically, the cell becomes large with pale cytoplasm and normally located nucleus. Examples of hydropic swelling:Ballooning of hepatocytes in cases of acute viral hepatitis, epidermal cells in burns, Mickulicz cell(Histiocytes) in Rhinoscleroma .
- 39. Viral hepatitis The hepatocytes adjacent to the portal tract (right) are very swollen and hydropic (severe ballooning degeneration)
- 40. CE L AR ADAP AT L UL T ION DIFINITION: Cellular adaptation is a state that lies intermediate between the normal unstressed cell and the injured over stressed cells. The major most important adaptive changes in the cells are: Hyperplasia, Hypertrophy, Atrophy, Metaplasia, Dysplasia and intracellular storage.
- 41. Hyperplasia: Definition: It is an increase in the size of tissue or organ due to increase in the number of its specialized cells. This can result from: 1. Increased functional demand: • Physiological hyperplasia of the breast in pregnancy and lactation. • Hyperplasia of the bone marrow in haemolytic anaemia, Fe, B12 or folic acid deficiency anaemias. • Hyperplasia of the lining epithelia in the process of regeneration and repair of an ulcer
- 42. 2. Increased hormonal stimulation A) Hyperplasia of endocrine glands: * Pituitary gland → excess growth hormone: • Before puberty → gigantism. • After puberty → acromegaly. * Thyroid gland → thyrtoxicosis. * Parathyroid gland → hypercalcaemia → metastatic calcification → osteitis fibrosa cystica. * Adrenal cortex → Cushing’s syndrome.
- 43. THYROID HYPERFUNCTION Exophthalmos
- 44. B) Hyperplasia of endocrine-target organs: *Breast → mammary cystic hyperplasia (Fibrocystic disease). * Endometrium → endometrial hyperplasia. * Prostate → senile nodular hyperplasia. They result from increased oestrogenic stimulation.
- 46. Hyperplasia of Endocrine target organes HYPERPLASIA OF ENDOCRIN TARGET ORGAN
- 47. 3. Chronic inflammation or irritation Pressure from ill fitting shoes causes hyperplasia of the skin (calluses). Chronic cystitis of the bladder commonly causes hyperplasia of the bladder epithelium (Bilharziasis & stones). Chronic inflammatory lesions of the skin → hyperplasia.
- 48. 4. Hyperplasia of connective tissue cells in wound healing (proliferating fibroblasts and blood vessels. 5. Compensatory hyperplasia in the liver after partial hepatectomy.
- 49. 6. Pseudoneoplastic hyperplasia: a) Pseudomalignant connective tissue hyperplasia e.g. pseudolymphoma of the orbit and pseudosarcoma in fibrous tissue. b) Pseudomalignant epithelial hyperplasia e.g. keratoacanthoma and hyperplasia of the skin around chronic ulcer. Cell proliferation depends on the action of: a) Some growth factors and cytokines e.g. epidermal growth factor (EGF)-alfa transforming growth factor (TGF-α), Her- 2 neu-and interleukin-6 (IL-6) and tumour necrosis factor (TNF-α).
- 50. It Differs From Neoplasia By The Following : It occurs in tissue made up of labile or stable cells that have the power of regeneration under normal or pathologic conditions. Occurs in response to a stimulus, continues as the stimulus continues and disappears when it is removed. Usually performs a function e.g. Lactating breast. It is a reversible non-neoplastic process, nevertheless sometimes malignant tumours do arise on top of abnormal or atypical hyperplasia (Endometrial hypeplasia).
- 51. HY ERTROP P HY DIFINITION: It is the increase in the size of the organ or tissue due to increase in the size of it specialized cells In a pure form, it is found in muscles: 1. Occurs in response to an increased demand for overwork: a) Skeletal muscle in athelets
- 52. Smooth muscles The uterus in pregnancy. Stomach in pyloric stenosis. Alimentary tract proximal to an obstruction. Urinary bladder with obstruction to urine outflow e.g. prostatic enlargement or urethral stricture.
- 53. With hypertrophy of the muscle wall
- 54. c) Cardiac muscle: * Right ventricle: in MS, Tl, PS, chronic lung diseases. * Left ventricle in MI, AS, AI, systemic hypertension.
- 55. Chronic lung diseases: RS Hypertrophy MS MI RSH hypertrophy LS Hypertrophy
- 57. • 2. Physiologic (hormonal) hypertrophy: occurs during maturation under the effects of hormones. Sex hormones at puterty lead to hypertrophy of juvenile sex organs, and breast tissue in lactating women under the effect of prolactin. • 3. Compensatory hypertrophy of one kidney due to removal of the other
- 58. Blood supply Nerve Supply Hormonal Stimulation Does A Function
- 59. Mechanisms include: 1. Loss of innervation. 2. Reduced nutrient and oxygen supply. 3. Reduced functional demand. 4. Reduced hormonal stimulation. These can occur under physiologic or pathologic conditions.
- 60. A) Physiological atrophy Ductus arteriosus and umbilical vessels,after birth. Thymus gland after puberty. Lymphoid tissue in adenoid and tonsils. Postmenopausal atrophy of the breast, uterus and ovaries. Aging process in the skin, brown atrophy of the heart and brain atrophy.
- 61. B) Pathologic atrophy: 1. Ischaemic atrophy: usually due to partial and gradual occlusion of the arterial blood supply by atherosclerosis, in the heart (atherosclerotic heart disease), brain or kidney etc. 2. Disuse atrophy: due to forced inactivity of muscle e.g. after prolonged immobilization of a limb in plaster (Cast). 3. Neuropathic atrophy: following lower motor neuron lesions e.g. poliomyelitis. 4. Pressure atrophy upon a localized area or group of cells, interfering with its blood and nutrient supply. * Pressure by growing tumour. * Prolonged pressure of a pulsating aortic aneurysm may cause pressure atrophy of the undersurface of the sternum anteriorly or of the bodies of the vertebrae
- 62. Gradualsupply & cellular in supply supply nutrients lead to Reduction in Grodual diminution in blood oxygen diminution atrophy; blood a nd and nutrients Lead to reduction in oxygen supply &cellular atrophy Normal Accumulation 0 f lipofucsin around nucleus
- 63. Preasure Atrophy on renal tissue by distended pelvis Increasing / pressure Pelvis greatly distended Obstruction incomplete
- 64. Hormonal atrophy: cessation of pituitary activity results in atrophic changes in the thyroid, adrenals, ovaries and other organs that are influenced by pituitary hormones. Secondary to immunologic injuries: the resulting tissue damage is accompanied by fibrosis and atrophy of the affected organ e.g. primary Addison’s disease due to autoimmune bilateral atrophy of adrenal gland, atrophic gastritis, atrophic thyroiditis, testicular
- 65. METAPLASIA Definition : It is the transformation of one type of differentiated tissue into another type of the same kind. It may occur in either epithelial or connective tissue. Pathogenesis: Metaplasia is thought to arise from reprogramming of stem cells to differentiate along new pathway under the effects of mixture of cytokines and growth factors. The most common is the replacement of a glandular epithelium by a squamous one due to prolonged chronic irritation, replacing the thin delicate epithelium with the tougher and more resistant squamous epithelium.
- 67. A) Epitlielium metaplasia 1. Squamous metaplasia: 2. Columnar metaplasia ( 1) Squamous metaplasia a) From pseudo-stratified columnar: * Trachea and bronchi in chronic bronchitis, cigarette smoking and bronchiectesis. * Nasal sinuses in chronic sinusitis and hypovitaminosis A. B) From transitional epithelium in bilharziasis of U.B. c) From simple columnar epithelium: * Endocervical mucosa and glands in cervical erosion. * Gall bladder with stones. d) From mesothelium of the pleura and peritoneum.
- 68. (2) Columnar metaplasia (A) From squamous: in the lower oesophagus e.g. Barrett oesophagitis (Precancerus). (B) Intestinal metaplasia of the specialized gastric mucosa in chronic atrophic gastritis. (C) Apocrine, pink cell, hyperplasia seen in fibrocystic disease of the breast. (D) In mesothelium of pleura, peritoneum and synovium.
- 70. (B) Connective tissue metaplasia • - It is the formation of cartilage, bone or adipose tissue, in tissues that normally do not contain these elements. • Osseous metaplasia: occurs in: (a) Sites of dystrophic calcification e.g. in scars, old T.B. (b) In muscles, in post-traumatic myositis ossificans.
- 71. Formation of bone in fibrous tissue In case of healing of a wound Scar Bone
- 72. Dysplasia (Intraepithelial neoplasia) Definition: It is partial loss of differentiation. 1. The involved epithelium shows evidence of cellular atypia: Pleomorphism of cells (variation in size and shape). Hyperchromatic nuclei with increased nucleo- cytoplasmic ratio and increased mitotic activity. Loss of polarity (orientation) of cells. Disordered maturation with impaired function. No invasion of basement membrane. 2. It represents reaction to underlying inflammation or to chronic irritation. 3. Mild and moderate degrees of dysplasia are
- 74. Carcinoma in situ. A section of the uterine cervix shows neoplastic squamous cells occupying the full thickness of the epithelium and confined to the mucosa by the underlying basement membrane.
- 75. 4. Examples of dysplasia: • 1. Occurs in the cervix in chronic cervicitis. • 2. In urothelium of urinary bladder in case of bilharziasis. 5. The most severe form, when the changes occupy the whole thickness of the epithelium indicates the diagnosis of intraepithelial carcinoma or carcinoma in situ (pre-invasive carcinoma). Carcinoma in situ characterized by diffuse cellular atypia involving the whole thickness of the affected epithelium without invasion of the basement membrane. The commonest sites of IEN are cervix uetri, bronchial epithelium, buccal mucosa and skin.
