Lect 6
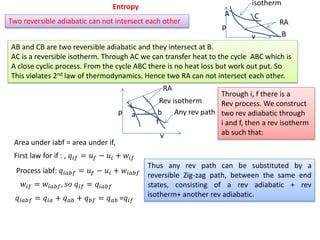
- 1. Entropy Two reversible adiabatic can not intersect each other CA B C isotherm RA AB and CB are two reversible adiabatic and they intersect at B. AC is a reversible isotherm. Through AC we can transfer heat to the cycle ABC which is A close cyclic process. From the cycle ABC there is no heat loss but work out put. So This violates 2nd law of thermodynamics. Hence two RA can not intersect each other. p v i f a b RA Rev isotherm Any rev path Through i, f there is a Rev process. We construct two rev adiabatic through i and f, then a rev isotherm ab such that: Area under iabf = area under if, First law for if : , 𝑞𝑖𝑓 = 𝑢 𝑓 − 𝑢𝑖 + 𝑤𝑖𝑓 Process iabf: 𝑞𝑖𝑎𝑏𝑓 = 𝑢 𝑓 − 𝑢𝑖 + 𝑤𝑖𝑎𝑏𝑓 𝑤𝑖𝑓 = 𝑤𝑖𝑎𝑏𝑓, 𝑠𝑜 𝑞𝑖𝑓 = 𝑞𝑖𝑎𝑏𝑓 p v 𝑞𝑖𝑎𝑏𝑓 = 𝑞𝑖𝑎 + 𝑞 𝑎𝑏 + 𝑞 𝑏𝑓 = 𝑞 𝑎𝑏 =𝑞𝑖𝑓 Thus any rev path can be substituted by a reversible Zig-zag path, between the same end states, consisting of a rev adiabatic + rev isotherm+ another rev adiabatic.
- 2. Clausius theorem p v Any rev cyclic process RA Rev isotherm T1 T2 Lets us consider a smooth close curve which represents a rev cycle. Then we take two Elemental parts from this cycle as shown in the picture (upper part and lower part). The upper part cab represented by an isotherm+ two RA and similarly the lower part of the curve. The upper isotherm is at a temp say T1 and the lower isotherm is at T2. Now this elementary process can be thought of as a Carnot engine which receives heat dq1 at T1 and rejects heat dq2 at T2. So for this elementary cycle 𝑑𝑞1 𝑇1 = 𝑑𝑞2 𝑇2 𝑜𝑟 𝑖𝑓 𝑤𝑒 𝑎𝑑𝑑 𝑤𝑖𝑡ℎ 𝑡ℎ𝑒 Signs of heat transfer then we get 𝑑𝑞1 𝑇1 + 𝑑𝑞2 𝑇2 = 0. If we add all the elementary cycles such that we cover the entire cyclic process then we get : dq1 dq2 𝑑𝑞1 𝑇1 + dq2 T2 + dq3 T3 + ⋯ . dqn Tn = 0 or 𝑑𝑞 𝑇 = 0 , hence 𝑑𝑞 𝑇 𝑖𝑠 𝑎 𝑝𝑟𝑜𝑝𝑒𝑟𝑡𝑦 𝑙𝑖𝑘𝑒 𝑢, 𝑇, 𝑣 𝑎𝑛𝑑 ℎ 𝑤ℎ𝑖𝑐ℎ is called entropy , ds. R1 R2 a b 𝑎 𝑏 𝑑𝑞 𝑇 𝑅1 + 𝑏 𝑎 𝑑𝑞 𝑇 𝑅2 = 0, 𝑎 𝑏 𝑑𝑞 𝑇 𝑅1 = 𝑎 𝑏 𝑑𝑞 𝑇 𝑅2 Hence entropy is a property, independent of path So, 𝑑𝑠 = 𝑑𝑞 𝑟𝑒𝑣 𝑇 or, 𝑎 𝑏 𝑑𝑞 𝑇 = 𝑎 𝑏 𝑑𝑠 = 𝑠 𝑏 −𝑠 𝑎 Clausius theorem
- 3. 𝑑𝑠 = 𝑑𝑞 𝑟𝑒𝑣 𝑇 ; If 𝑑𝑞 𝑟𝑒𝑣 = 0, then the process is called rev adiabatic and then ds=0 Hence s=Const; T S A B CD 𝑑𝑞 𝑑𝑞2 RA AB is a general process Either rev or irreversible, All other processes are rev. Consider an elemental cycle, as shown in the picture. 𝑑𝑞 = heat supplied at 𝑇1, 𝑑𝑞2 = ℎ𝑒𝑎𝑡 𝑟𝑒𝑗𝑒𝑐𝑡𝑒𝑑 𝑎𝑡 𝑇2 1 − 𝑑𝑞2 𝑑𝑞 𝑎𝑛𝑦 ≤ 1 − 𝑑𝑞2 𝑑𝑞 𝑟𝑒𝑣 𝑑𝑞2 𝑑𝑞 𝑎𝑛𝑦 ≥ 𝑑𝑞2 𝑑𝑞 𝑟𝑒𝑣 𝑑𝑞 𝑎𝑛𝑦 𝑑𝑞2 ≤ 𝑑𝑞 𝑟𝑒𝑣 𝑑𝑞2 𝑑𝑞 𝑎𝑛𝑦 𝑑𝑞2 ≤ 𝑇 𝑇2 ; 𝑠𝑖𝑛𝑐𝑒 𝑑𝑞 𝑟𝑒𝑣 𝑑𝑞2 = 𝑇 𝑇2 Or, Or, Or, Or, 𝑑𝑞 𝑎𝑛𝑦 𝑇 ≤ 𝑑𝑞2 𝑇2 𝑑𝑞 𝑎𝑛𝑦 𝑇 ≤ 𝑑𝑠, 𝑓𝑜𝑟 𝑎𝑛𝑦 𝑝𝑟𝑜𝑐𝑒𝑠𝑠 For a reversible process: 𝑑𝑞 𝑇 = 𝑑𝑠 For any other process : 𝑑𝑠 ≥ 𝑑𝑞 𝑇 For any cyclic process: 𝑑𝑞 𝑇 ≤ 𝑑𝑠 𝑑𝑞 𝑇 ≤ 0 𝐼𝑓 𝑑𝑞 𝑇 = 0, cycle is reversible, 𝑑𝑞 𝑇 < 0, cycle is irreversible and feasible 𝑑𝑞 𝑇 > 0, cycle is impossible Clausius Inequality
- 4. Entropy principle For any process undergone by the system, 𝑑𝑠 ≥ 𝑑𝑞 𝑇 , if the process is reversible then 𝑑𝑠 = 𝑑𝑞 𝑇 For isolated system, dq=0, so, 𝑑𝑠 ≥ 0, if isolated process is reversible, 𝑡ℎ𝑒𝑛 𝑑𝑠 = 0, 𝑠 = 𝑐 For irreversible process, 𝑑𝑠 > 0, 𝑠𝑜 𝑒𝑛𝑡𝑟𝑜𝑝𝑦 𝑎𝑙𝑤𝑎𝑦𝑠 𝑖𝑛𝑐𝑟𝑒𝑎𝑠𝑒𝑠 Causes of entropy increase: for a closed system entropy increases due to (a) External interaction, (b) internal irreversibility 𝑑𝑠 = 𝑑𝑠 𝑒𝑥𝑡 + 𝑑𝑠𝑖𝑛𝑡 = 𝑑𝑞 𝑇 + 𝑑𝑠𝑖𝑛𝑡; so, 𝑑𝑠 > 𝑑𝑞 𝑇 ; 𝑜𝑛𝑙𝑦 𝑓𝑜𝑟 𝑟𝑒𝑣𝑒𝑟𝑠𝑖𝑏𝑙𝑒 𝑝𝑟𝑜𝑐𝑒𝑠𝑠 𝑑𝑠𝑖𝑛𝑡 = 0 𝑑𝑠 = 𝑑𝑞 𝑇 + 𝑑𝑠𝑔𝑒𝑛 𝑠2 − 𝑠1 = 1 2 𝑑𝑞 𝑇 + 𝑠𝑔𝑒𝑛1−2 𝑑𝑠 𝑑𝑡 = 𝑞 𝑐𝑣 𝑇 + 𝑠 𝑔𝑒𝑛 Entropy change Entropy transfer Entropy generation dq T 𝑠 𝑔𝑒𝑛 𝑠2 − 𝑠1 For an open system 𝑚𝑖 𝑞 𝑠 𝑔𝑒𝑛 𝑚 𝑒 𝑚𝑖 𝑠𝑖 + 𝑞 𝑐𝑣 𝑇 + 𝑠 𝑔𝑒𝑛 = 𝑚 𝑒 𝑠 𝑒 + 𝑑𝑠 𝑐𝑣 𝑑𝑡 , for steady state 𝑑𝑠 𝑐𝑣 𝑑𝑡 = 0 𝑚𝑖 𝑠𝑖 + 𝑞 𝑐𝑣 𝑇 + 𝑠 𝑔𝑒𝑛 = 𝑚 𝑒 𝑠 𝑒 + 𝑚2 𝑠2 − 𝑚1 𝑠1 Integral form of the equation Change of entropy within the CV system
- 5. 𝑻 𝒅𝒔 relation First law and second law combined: 𝑑𝑞 = 𝑑𝑢 + 𝑝𝑑𝑣 𝑇𝑑𝑠 = 𝑑𝑢 + 𝑝𝑑𝑣 𝑑𝑠 = 𝑑𝑢 𝑇 + 𝑝 𝑇 𝑑𝑣 𝑑𝑠 = 𝑑𝑢 𝑇 + 𝑅 𝑣 𝑑𝑣 𝑑𝑞 𝑟𝑒𝑣 = 𝑇𝑑𝑠 𝑠2 − 𝑠1 = 𝑐 𝑣 𝑑𝑇 𝑇 + 𝑅 𝑣 𝑑𝑣 𝑠2 − 𝑠1 = 𝑐 𝑣 ln 𝑇2 𝑇1 + 𝑅 ln 𝑣2 𝑣1 1st Tds relation Only for ideal gases ℎ = 𝑢 + 𝑝𝑣 𝑑ℎ = 𝑑𝑢 + 𝑝𝑑𝑣 + 𝑣𝑑𝑝 𝑑ℎ = 𝑇𝑑𝑠 + 𝑣𝑑𝑝 T𝑑𝑠 = 𝑑ℎ − 𝑣𝑑𝑝 2nd Tds relation 𝑑𝑠 = 𝑑ℎ 𝑇 − 𝑣 𝑇 𝑑𝑝 𝑠2 − 𝑠1 = 𝑐 𝑝 𝑑𝑇 𝑇 − 𝑅 𝑝 𝑑𝑝 𝑐 𝑝 ln 𝑇2 𝑇1 − 𝑅 ln 𝑝2 𝑝1 Only for ideal gases