Blood pH balance
- 1. Lab 11: Acid - Base Balance of the Arterial Plasma
- 2. Lab 11 - Carbonic Acid Buffering System Important to maintain blood pH at intermediate set point (pH = 7.4) - For metabolic processes and systems to work well - Lot of the proteins are pH-dependent : Homeostatic mechanism in which the lungs and/or kidneys will maintain arterial blood pH approximately at 7.4.
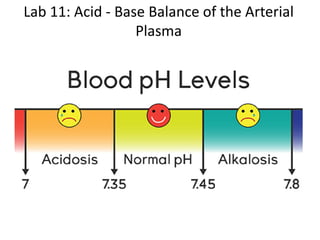
- 3. Lab 11 - Causes of Blood pH Imbalance (in arterial plasma) Blood pH is mainly determined by the in the plasma.
- 4. Lab 11 - Acidosis and Alkalosis Disorders Respiratory acidosis: a disorder where the respiratory system fails to eliminate CO2 as fast as it is produced thus H+ concentration in the arterial plasma is increased above normal (pH < 7.35). Respiratory alkalosis: a disorder where the respiratory system eliminates CO2 faster than it is produced thus H+ concentration in the arterial plasma is decreased below normal (pH > 7.45). Metabolic acidosis: a disorder where there is an increase of arterial H+ concentration due to some cause other than a primary change in PCO2 (pH < 7.35). Metabolic alkalosis: a disorder where there is a decrease of arterial H+ concentration due to some cause other than a primary change in PCO2 (pH > 7.45).
- 5. Lab 11 - Respiratory Acid-Base Disorders – Cause and Compensatory Mechanism Cause: Respiratory system (PCO2) • • Increase CO2 production to alveolar ventilation (Net gain of plasma CO2) • • Decrease CO2 production to alveolar ventilation (Net loss of plasma CO2) Compensatory: Kidneys slowly adjust HCO3 - • via secretion of H+ (No net gain of plasma HCO3 -) • (Net gain of plasma HCO3 -) • Binding to filtered nonbicarbonate buffer via secretion of excess H+ • Glutamate breakdown and secretion of NH4 + • (Net loss of plasma HCO3 -) • Insufficient secretion of H+ Respiratory Acidosis Respiratory Alkalosis
- 6. Lab 11 - Metabolic Acid-Base Disorders – Cause and Compensatory Mechanism • Respiratory rate • Tidal volume Influence net gain or loss of plasma CO2 Cause: Any situation other than the respiratory system • Excessive production of lactic acid • Excessive diarrhea • Persistent vomiting Compensatory: Secondary: Kidneys slowly adjust HCO3 - Primary: Lungs quickly adjust PCO2 • “Reabsorption” of filtered HCO3 - • New HCO3 - • Metabolic acidosis Metabolic alkalosis • • Excretion of HCO3 -
- 7. Lab 11 – Goals (aka Learning Outcomes) • Explain the homeostatic regulation of arterial pH using the carbonic acid buffering system. • Examine respiratory and metabolic acidosis and alkalosis under various conditions. o Predict and justify the cause of the acid-base disorder. o Predict and justify the compensatory system and mechanisms to correct pH imbalances.
- 8. Lab 11 – Experimental Setup for Part 1 Animal model: None, but there is an inanimate model Experimental Setup: PhysioEx simulation Renal Responses to Respiratory Acidosis and Alkalosis
- 9. Lab 11 – Experimental Setup for Part 2 Metabolic Acidosis and Alkalosis Animal model: Unknown Experimental Setup: Case study Initial pH change Values before compensation Compensated state (steady state) pH Arterial PCO2 (mmHg) Alveolar ventilation (ml/min) Arterial HCO3 - (mMol/L) pH (log molar) Arterial CO2 (mmHg) Alveolar ventilation (breaths/min) Arterial HCO3 - (mMol/L) How these variables change 5 minutes after compensation relative to before compensation