Transforming the Management of Inflammatory Bowel Disease: A Closer Look at the Role of JAK Inhibitors and the Patient’s Perspective in Achieving Therapeutic Goals.
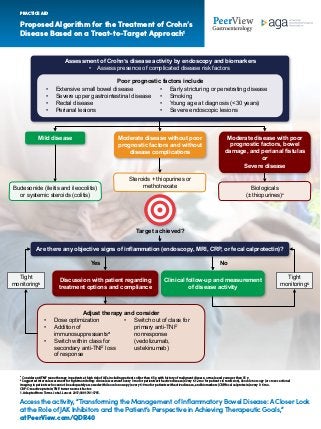
- 1. Access the activity, “Transforming the Management of Inflammatory Bowel Disease: A Closer Look at the Role of JAK Inhibitors and the Patient’s Perspective in Achieving Therapeutic Goals,” at PeerView.com/QDR40 Proposed Algorithm for the Treatment of Crohn’s Disease Based on a Treat-to-Target Approach1 PRACTICE AID a Consider anti-TNF monotherapy in patients at high risk of AEs, including patients older than 65 y, with history of malignant disease, or male and younger than 35 y. b Suggested interval assessment for tight monitoring: clinical assessment every 3 mo for patients with active disease (every 6-12 mo for patients in remission), ileocolonoscopy (or cross-sectional imaging in patients who cannot be adequately assessed with ileocolonoscopy) every 6-9 mo for patients with active disease, and biomarkers (CRP/fecal calprotecin) every 3-6 mo. CRP: C-reactive protein; TNF: tumor necrosis factor. 1. Adapted from Torres J et al. Lancet. 2017;389:1741-1755. Assessment of Crohn’s disease activity by endoscopy and biomarkers Assess presence of complicated disease risk factors Target achieved? Are there any objective signs of inflammation (endoscopy, MRI, CRP, or fecal calprotectin)? Yes No Tight monitoringb Discussion with patient regarding treatment options and compliance Poor prognostic factors include • Extensive small bowel disease • Severe upper gastrointestinal disease • Rectal disease • Perianal lesions • Early stricturing or penetrating disease • Smoking • Young age at diagnosis (<30 years) • Severe endoscopic lesions Mild disease Budesonide (ileitis and ileocolitis) or systemic steroids (colitis) Moderate disease without poor prognostic factors and without disease complications Steroids + thiopurines or methotrexate Moderate disease with poor prognostic factors, bowel damage, and perianal fistulas Severe disease Biologicals (± thiopurines)a Tight monitoringb Clinical follow-up and measurement of disease activity Adjust therapy and consider • Dose optimization • Addition of immunosuppressantsa • Switch within class for secondary anti-TNF loss of response • Switch out of class for primary anti-TNF nonresponse (vedolizumab, ustekinumab) • or
- 2. Access the activity, “Transforming the Management of Inflammatory Bowel Disease: A Closer Look at the Role of JAK Inhibitors and the Patient’s Perspective in Achieving Therapeutic Goals,” at PeerView.com/QDR40 AGA Institute Ulcerative Colitis Care Pathway1,a,b PRACTICE AID a Tofacitinib, an oral JAK inhibitor, was approved May 2018 for the treatment of adult patients with moderately to severely active ulcerative colitis who have had an inadequate response or who are intolerant to TNF blockers. b Ustekinumab, an interleukin-12 and -23 antagonist, was approved October 2019 for the treatment of adults with moderately to severely active ulcerative colitis. 5-ASA: 5-aminosalicylate; 6-TGN: 6-Thioguanine nucleotide; AGA: American Gastroenterological Association; CRP: C-reactive protein; ESR: erythrocyte sedimentation rate; JAK: Janus kinase; TNF: tumor necrosis factor. 1. Adapted from Dassopoulos T et al. Gastroenterology. 2015;149:238-245. Patient at Low Risk for Colectomy • Limited anatomic extent • Mild endoscopic disease Patient at Moderate/High Risk for Colectomy • Extensive colitis • Deep ulcers • Age <40 • High CRP and ESR • CMV infection • Steroid-requiring disease • History of hospitalization • C. difficile infection No remission Remission • Oral 5-ASA, and/or • Rectal 5-ASA, and/or • Oral budesonide or prednisone, and/or • Rectal corticosteroids • Short course of corticosteroids with initiation of thiopurine, or • Anti-TNF ± thiopurines • Vedolizumab ± immunomodulator • Maintenance with oral and/or rectal 5-ASA • Taper corticosteroid over 60 days Maintenance Options • Thiopurine and taper corticosteroids over 60 days • • Options • Anti-TNF ± thiopurine • Thiopurine (optimize 6-TGN concentrations) • Vedolizumab ± immunomodulator • Proctocolectomy Remission No remission Vedolizumab ± thiopurine or methotrexate Anti-TNF ± thiopurine
- 3. Access the activity, “Transforming the Management of Inflammatory Bowel Disease: A Closer Look at the Role of JAK Inhibitors and the Patient’s Perspective in Achieving Therapeutic Goals,” at PeerView.com/QDR40 A Closer Look at the Clinical Potential of JAK Inhibitors for the Treatment of IBD PRACTICE AID CD: Crohn’s disease; GM-CSF: granulocyte-macrophage colony-stimulating factor; GP: glycoprotein; IL: interleukin; JAK: Janus kinase; NK: natural killer; STAT: signal transducer and activator of transcription; TYK: tyrosine kinase; UC: ulcerative colitis. 1. Flamant M et al. Drugs. 2017;77:1057-1068. 2. Danese S et al. Gut. 2019;68:1893-1899. Compound Tofacitinib Filgotinib Upadacitinib CD, stage of development UC, stage of development — FDA-approved Phase 3 Phase 3 Phase 3 Phase 3 Target JAK1, JAK2, JAK3 JAK1 JAK1 Gut selectivity — — — Hemoglobin level Lymphocyte number No change Neutrophil number Platelet count No data NK cell number No change HDL level LDL Level No change Liver transaminase level No change Creatinine level Creatine phosphokinase level No data JAK Inhibitors Currently Approved or in Development for IBD2 Ligand IL-6 IL-12/23 IL-9 IL-10 IL-22 GM-CSF IFN- Receptor IL-6R GP130R IL-12Rβ1 IL-12Rβ2 IL-23R IL-9Rα IL-2Rγ IL-10Rα IL-10Rβ IL-22R1 IL-10Rβ GM-CSFR IFNγR1 IFNγR2 JAKs JAK1 JAK2 TYK2 JAK2 TYK2 JAK1 JAK3 JAK1 TYK2 JAK1 TYK2 JAK2 JAK1 JAK2 STATs STAT1 STAT3 STAT3 STAT4 STAT1 STAT3 STAT5 STAT3 STAT1 STAT3 STAT5 STAT1 STAT3 STAT5 STAT1 JAK/STAT Pathways Involved in IBD1 Actions on intestinal epithelium Inflammation; wound healing Inflammation Negative impact on wound healing Anti- inflammatory Wound healing; defensin expression Epithelium protection Inflammation; epithelium protection Nucleus STAT STAT STAT STAT STAT STAT STAT STATSTAT STAT STAT STATSTAT STAT STAT STAT
- 4. Access the activity, “Transforming the Management of Inflammatory Bowel Disease: A Closer Look at the Role of JAK Inhibitors and the Patient’s Perspective in Achieving Therapeutic Goals,” at PeerView.com/QDR40 IBD Patient Education Materials From the Crohn's and Colitis Foundation1 PRACTICE AID Shared Decision-Making for IBD Treatment Decisions What is shared decision-making? A process in which patients, caregivers, and their healthcare team work together to make decisions about the patient’s treatment and healthcare plan What are the benefits of shared decision-making? • h confidence in treatment choice • h treatment satisfaction • May h treatment adherence Key Steps to Participating in Shared Decision-Making Support Share personal goals, values, preferences, and insurance coverage, and ask for support as you review all options Discuss Talk through options with your healthcare team and make a decision together based on medical evidence and personal needs Follow Through After making your decision, remain in contact with your healthcare team and ask any follow-up questions Information Request and gather all information about your treatment options, including the pros/cons and benefits/risks •
- 5. Access the activity, “Transforming the Management of Inflammatory Bowel Disease: A Closer Look at the Role of JAK Inhibitors and the Patient’s Perspective in Achieving Therapeutic Goals,” at PeerView.com/QDR40 IBD Patient Education Materials From the Crohn's and Colitis Foundation1 PRACTICE AID Are These Symptoms Related to My IBD? Abdominal pain, diarrhea, cramping, and weight loss are all very common symptoms of Crohn’s disease and ulcerative colitis You can also experience symptoms outside of your digestive tract. They can appear before you even know you have IBD Signs and Symptoms Beyond the Gut Arthritis Inflammation of joints Liver Disorders Hepatitis, gallstones, fatty liver disease, primary sclerosing cholangitis General Symptoms Dizziness, fatigue, shortness of breath Anemia Fewer red blood cells than normal Skin Disorders Erythema nodosum, skin tags, pyoderma gangrenosum Eye Disorders Uveitis, dry eyes, keratopathy, episcleritis
- 6. Access the activity, “Transforming the Management of Inflammatory Bowel Disease: A Closer Look at the Role of JAK Inhibitors and the Patient’s Perspective in Achieving Therapeutic Goals,” at PeerView.com/QDR40 IBD Patient Education Materials From the Crohn's and Colitis Foundation1 PRACTICE AID 1. http://www.crohnscolitisfoundation.org. Accessed January 16, 2020. I’m Still Experiencing IBD Symptoms. What Now? If you are taking IBD medication(s) and still experiencing symptoms, it is important to share the following information with your healthcare team: Ask questions Inquire about other possible treatment options, risks and benefits of medications, and opportunities to participate in clinical trials Any changes in your symptoms before or during treatment Have your symptoms improved, deteriorated, or stayed the same? The severity of abdominal pain Share pain level on a 0-10 scale Quality of life issues Share your ability/inability to attend work/school, eat, socialize, and exercise Your symptoms in detail Some examples: Number of bowel movements/day, amount of weight lost/gained
- 7. Ongoing Clinical Trials With JAK Inhibitors for the Treatment of Crohn’s Disease and Ulcerative Colitis1 PRACTICE AID Access the activity, “Transforming the Management of Inflammatory Bowel Disease: A Closer Look at the Role of JAK Inhibitors and the Patient’s Perspective in Achieving Therapeutic Goals,” at PeerView.com/QDR40 Trial Patient Population Enrollment Treatment Arms ClinicalTrials.gov Identifier SELECTION1 (Phase 3) Moderate to severe UC; biologic-naïve and biologic-experienced 1,351 Filgotinib vs placebo NCT02914522 SELECTIONLTE (Phase 3) Long-term safety in patients with UC who completed or discontinued a prior filgotinib trial 1,000 Filgotinib vs placebo NCT02914535 DIVERSITY1 (Phase 3) Moderate to severe CD; biologic-naïve and biologic-experienced 1,320 Filgotinib vs placebo NCT02914561 DIVERSITYLTE (Phase 3) Long-term safety in CD patients who completed or discontinued a prior filgotinib trial 1,000 Filgotinib vs placebo NCT02914600 DIVERGENCE2 (Phase 2) Perianal fistulizing CD 75 Filgotinib vs placebo NCT03077412 Phase 2 Small bowel CD 100 Filgotinib vs placebo NCT03046056 Filgotinib: Ongoing Clinical Trials
- 8. Ongoing Clinical Trials With JAK Inhibitors for the Treatment of Crohn’s Disease and Ulcerative Colitis1 CD: Crohn’s disease; JAK: Janus kinase; UC: ulcerative colitis. 1. https://clinicaltrials.gov. Accessed February 10, 2020. PRACTICE AID Access the activity, “Transforming the Management of Inflammatory Bowel Disease: A Closer Look at the Role of JAK Inhibitors and the Patient’s Perspective in Achieving Therapeutic Goals,” at PeerView.com/QDR40 Trial Patient Population Estimated Enrollment Treatment Arms ClinicalTrials.gov Identifier U-ACCOMPLISH (Phase 3) Moderate to severe UC 462 Upadacitinib vs placebo NCT03653026 U-ACHIEVE (Phase 3) Moderate to severe UC 844 Upadacitinib vs placebo NCT02819635 Phase 3 Long-term efficacy and safety in UC patients who participated in U-ACHIEVE 950 Upadacitinib vs placebo NCT03006068 M14-431 (Phase 3) Moderate to severe CD; inadequate response or intolerance to biologics 645 Upadacitinib vs placebo NCT03345836 M14-433 (Phase 3) Moderate to severe CD; inadequate response or intolerance to conventional and/or biologics 501 Upadacitinib vs placebo NCT03345849 Phase 3 Long-term efficacy and safety in CD patients who completed M14-431 or M14-433 studies 738 Upadacitinib vs placebo NCT03345823 Upadacitinib: Ongoing Clinical Trials
