Teaming Up to Reduce the Burden of Cancer-Associated VTE: Applying the Latest Guidelines and Evidence in Gastrointestinal and Hematologic Malignancies and Other Cancer Settings
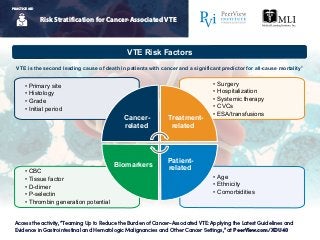
- 1. Access the activity, “Teaming Up to Reduce the Burden of Cancer-Associated VTE: Applying the Latest Guidelines and Evidence in Gastrointestinal and Hematologic Malignancies and Other Cancer Settings,” at PeerView.com/XDU40 PRACTICE AID Risk Stratification for Cancer-Associated VTE PRACTICE AID • Age • Ethnicity • Comorbidities • CBC • Tissue factor • D-dimer • P-selectin • Thrombin generation potential • Surgery • Hospitalization • Systemic therapy • CVCs • ESA/transfusions • Primary site • Histology • Grade • Initial period Cancer- related Treatment- related Patient- related Biomarkers VTE Risk Factors VTE is the second leading cause of death in patients with cancer and a significant predictor for all-cause mortality1
- 2. Access the activity, “Teaming Up to Reduce the Burden of Cancer-Associated VTE: Applying the Latest Guidelines and Evidence in Gastrointestinal and Hematologic Malignancies and Other Cancer Settings,” at PeerView.com/XDU40 PRACTICE AID Risk Stratification for Cancer-Associated VTE PRACTICE AID Khorana Risk Score2 Cancer type Stomach +2 Pancreas +2 Lung +1 Lymphoma +1 Gynecologic +1 Bladder +1 Testicular +1 Other 0 Prechemotherapy platelet count 350 x 109/L No 0 Yes +1 Hemoglobin level <10 g/dL or using RBC growth factors No 0 Yes +1 Prechemotherapy leukocyte count 11 x 109/L No 0 Yes +1 BMI 35 kg/m2 No 0 Yes +1 Result (total)
- 3. Access the activity, “Teaming Up to Reduce the Burden of Cancer-Associated VTE: Applying the Latest Guidelines and Evidence in Gastrointestinal and Hematologic Malignancies and Other Cancer Settings,” at PeerView.com/XDU40 PRACTICE AID CBC: complete blood count; CVC: central venous catheter; ESA: erythropoiesis-stimulating agent; RBC: red blood cell; VTE: venous thromboembolism. 1. Farge D et al. Lancet Oncol. 2016;17:e452-e466. 2. https://www.mdcalc.com/khorana-risk-score-venous-thromboembolism-cancer-patients. 3. Angelini D, Khorana AA. Semin Thromb Hemost. 2017;43:469-478. Risk Stratification for Cancer-Associated VTE PRACTICE AID Applying Risk Assessment3 Clinical applications of VTE risk-assessment tools Education Screening Prophylaxis Prognosis Increased awareness Early reporting of symptoms Identify occult VTE Early treatment of VTE Preventing VTE in high-risk patients Cancer progression Mortality
- 4. Decision Guide for Using Direct Oral Anticoagulants in Cancer-Associated VTE Access the activity, “Teaming Up to Reduce the Burden of Cancer-Associated VTE: Applying the Latest Guidelines and Evidence in Gastrointestinal and Hematologic Malignancies and Other Cancer Settings,” at PeerView.com/XDU40 PRACTICE AID Clinical Practice Guidelines for Cancer-Associated VTE1-4 Recommendations for Ambulatory Patients With Cancer (No Bleeding) ASCO • • NCCN • • • ITAC • • ISTH • Recommendations for Hospitalized Patients With Cancer Admitted for Medical Reasons (No Bleeding) ASCO NCCN ITAC Primary thromboprophylaxis – ONLY in High-risk patients (Khorana score 2) with apixaban, rivaroxaban, or LMWH Patients with myeloma on thalidomide- or lenalidomide-based regimen with aspirin or LMWH Primary thromboprophylaxis – ONLY in Intermediate-/high-risk patients (Khorana score 2) with apixaban or rivaroxaban Low-risk patients with myeloma on IMiDs with aspirin High-risk patients with myeloma on IMiDs with apixaban or LMWH or warfarin Primary thromboprophylaxis – ONLY in Locally advanced or metastatic pancreatic cancer treated with systemic anticancer therapy with LMWH Patients on systemic anticancer therapy, at intermediate-high risk of VTE (Khorana score 2 or cancer type [eg, pancreatic]) with DOACs (apixaban or rivaroxaban) Primary thromboprophylaxis – ONLY in Cancer patients with Khorana score 2 with DOACs (apixaban or rivaroxaban) Yes Yes Yes • • • • • • • •
- 5. Decision Guide for Using Direct Oral Anticoagulants in Cancer-Associated VTE Access the activity, “Teaming Up to Reduce the Burden of Cancer-Associated VTE: Applying the Latest Guidelines and Evidence in Gastrointestinal and Hematologic Malignancies and Other Cancer Settings,” at PeerView.com/XDU40 PRACTICE AID Clinical Practice Guidelines for Cancer-Associated VTE1-3 (Cont’d) ASCO • NCCN • • • ITAC • Recommendations for Patients With Cancer Undergoing Major Surgery (No Contraindications) ASCO • NCCN • ITAC • Recommendations for DOAC Therapy for Acute VTE Yes Initial treatment with LMWH followed by edoxaban, rivaroxaban Yes DOACs (apixaban, edoxaban, rivaroxaban) preferred for nongastric or nongastroesophageal cancers LMWH (dalteparin, enoxaparin) preferred for gastric or gastroesophageal cancers DOACs (dabigatran) if above regimens not appropriate/available Yes Initial treatment with LMWH, DOAC, unfractionated heparin, or fondaparinux followed by LMWH or DOACs Yes Continue 7-10 d or up to 4 wk post dismissal in high-risk patients Yes Continue for 4 wk post dismissal in high-risk patients Yes Continue 7-10 d or up to 4 wk post dismissal in high-risk patients • • • • • • • •
- 6. Decision Guide for Using Direct Oral Anticoagulants in Cancer-Associated VTE Access the activity, “Teaming Up to Reduce the Burden of Cancer-Associated VTE: Applying the Latest Guidelines and Evidence in Gastrointestinal and Hematologic Malignancies and Other Cancer Settings,” at PeerView.com/XDU40 PRACTICE AID ASCO: American Society of Clinical Oncology; DOAC: direct oral anticoagulant; IMiD: immunomodulatory drug; ISTH: International Society on Thrombosis and Haemostasis; ITAC: International Initiative on Thrombosis and Cancer; LMWH: low-molecular-weight heparin; NCCN: National Comprehensive Cancer Network; PE: pulmonary embolism; VTE: venous thromboembolism. 1. Key NS et al. JClinOncol. 2020;38:496-520. 2. NCCN Clinical Practice Guidelines in Oncology. Cancer-Associated Venous Thromboembolic Disease. Version 2.2020. https://www.nccn.org/professionals/physician_ gls/pdf/vte.pdf. 3. Farge D et al; International Initiative on Thrombosis and Cancer (ITAC) advisory panel. LancetOncol. 2019;20:e566-e581. 4. Wang et al. JThrombHaemost. 2019;17:1772–1778. 5. Kraaijpoel N, Carrier M. Blood. 2019;133:291-298. Treatment Algorithm for Cancer-Associated VTE5 Cancer patient with symptomatic or incidental DVT or PE Does the patient have 1. Drug-drug interactions with DOACS, or 2. A high risk of bleeding? Initiate anticoagulant treatment with DOAC (apixaban, edoxaban, rivaroxaban) Initiate anticoagulant treatment with LMWH Consider extended anticoagulant treatment beyond 6 months if the cancer is still active No Yes
- 7. PRACTICE AID Direct Oral Anticoagulants for Cancer-Associated VTE PRACTICE AID Access the activity, “Teaming Up to Reduce the Burden of Cancer-Associated VTE: Applying the Latest Guidelines and Evidence in Gastrointestinal and Hematologic Malignancies and Other Cancer Settings,” at PeerView.com/XDU40 FDA-Approved DOACs Available in the United States Apixaban Rivaroxaban Edoxaban Betrixaban X VIIIa Dabigatran Va Prothrombin Fibrinogen Fibrin Thrombin Xa
- 8. PRACTICE AID a In combination with aspirin. Afib: atrial fibrillation; CAD: coronary artery disease; CV: cardiovascular; DOAC: direct oral anticoagulant; PAD: peripheral artery disease; VTE: venous thromboembolism. 1. https://natfonline.org/wp-content/uploads/2020/05/NATF-ACC-Patients-May-2020-.pdf. 2. Eliquis (apixaban) Prescribing Information. https://packageinserts.bms.com/pi/pi_eliquis.pdf. 3. Bevyxxa (betrixaban) Prescribing Information. https://bevyxxa.com/wp-content/ uploads/2020/04/BevyxxaPI.pdf. 4. Savaysa (edoxaban) Prescribing Information. https://dsi.com/prescribing-information-portlet/getPIContent?productName=Savaysa&inline=true. 5. Pradaxa (dabigatran etexilate mesylate) Prescribing Information. https://docs.boehringer- ingelheim.com/Prescribing%20Information/PIs/Pradaxa/Pradaxa.pdf. 6. Xarelto (rivaroxaban) Prescribing Information. http://www.janssenlabels.com/package-insert/product-monograph/prescribing-information/XARELTO-pi.pdf. Direct Oral Anticoagulants for Cancer-Associated VTE PRACTICE AID Access the activity, “Teaming Up to Reduce the Burden of Cancer-Associated VTE: Applying the Latest Guidelines and Evidence in Gastrointestinal and Hematologic Malignancies and Other Cancer Settings,” at PeerView.com/XDU40 Apixaban1,2 2.5-mg and 5-mg tablets Initial US Approval: 2012 Indication • Stroke prevention in nonvalvular Afib • VTE treatment • VTE prevention in patients already treated for a blood clot • VTE prevention after hip and knee replacement surgery Adverse Events • Increased risk of thrombotic events after premature discontinuation • Risk of bleeding • Spinal/epidural anesthesia or puncture Betrixaban1,3 40-mg or 80-mg capsule Initial US Approval: 2017 Indication • VTE prevention in hospitalized acute medically ill patients Adverse Events • Risk of bleeding • Spinal/epidural anesthesia or puncture Dabigatran1,5 75-mg, 110-mg,or 150-mg capsule Initial US Approval: 2010 Indication • Stroke prevention in nonvalvular Afib • VTE treatment • VTE prevention in patients already treated for a blood clot • VTE prevention after hip replacement surgery Adverse Events • Increased risk of thrombotic events after premature discontinuation • Risk of bleeding • Spinal/epidural anesthesia or puncture • Thromboembolic and bleeding events in patients with prosthetic heart valves • Increased risk of thrombosis in patients with triple-positive antiphospholipid syndrome Edoxaban1,4 15-mg, 30-mg, or 60-mg tablet Initial US Approval: 2015 Indication • Stroke prevention in nonvalvular Afib • VTE treatment Adverse Events • Increased risk of stroke with discontinuation in patients with nonvalvular Afib • Risk of bleeding • Spinal/epidural anesthesia or puncture Rivaroxaban1,6 2.5-mg, 10-mg, 15-mg, or 20-mg tablet Initial US Approval: 2011 Indication • Stroke prevention in nonvalvular Afib • VTE treatment • VTE prevention in patients already treated for a blood clot • VTE prevention after hip and knee replacement surgery • VTE prevention during hospitalization and after discharge in adults with acute medical illness • Prevention of major CV events in patients with CAD and PADa Adverse Events • Increased risk of stroke after discontinuation in nonvalvular Afib • Risk of bleeding • Spinal/epidural hematoma
