Hitting the BCMA Target in Multiple Myeloma: Insights on CAR-T Therapy and Innovative Antibody Options
•
0 likes•75 views
This document summarizes BCMA antibody and CAR-T cell therapies for multiple myeloma. It provides regulatory status, dosing details, and safety considerations for four BCMA antibody therapies - elranatamab, linvoseltamab, teclistamab, and ABBV-383. It also reviews dosing ranges and general safety principles for the two FDA-approved CAR-T cell therapies ciltacabtagene autoleucel and idecabtagene vicleucel. Both CAR-T therapies require administration through a Risk Evaluation and Mitigation Strategy program due to risks of cytokine release syndrome and neurologic toxicities.
Report
Share
Report
Share
Download to read offline
Recommended
Chair, Anthony R. Mato, MD, MSCE, Kristen Battiato, MSN, RN, AGNP-C, Dipti Gupta, MD, MPH, and Amber C. King, PharmD, BCOP, prepared useful Practice Aids pertaining to B-cell cancers for this CME/MOC/NCPD activity titled “Interprofessional Perspectives on Safety Management With Targeted Therapy for B-Cell Malignancies.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD information, and to apply for credit, please visit us at http://bit.ly/3cHX3zm. CME/MOC/NCPD credit will be available until October 3, 2023.Interprofessional Perspectives on Safety Management With Targeted Therapy for...

Interprofessional Perspectives on Safety Management With Targeted Therapy for...PVI, PeerView Institute for Medical Education
Chair & Presenter, Beth Faiman, PhD, MSN, APN-BC, AOCN, BMTCN, FAAN, FAPO, Donna D. Catamero, ANP-BC, OCN, CCRC, and Tiffany A. Richards, PhD, ANP-BC, AOCNP, prepared useful Practice Aids pertaining to multiple myeloma for this NCPD/ILNA activity titled “Betting on BCMA in Multiple Myeloma: Oncology Nurse Principles for Delivering Effective Care With BCMA Antibodies and Cellular Therapy.” For the full presentation, downloadable Practice Aids, and complete NCPD/ILNA information, and to apply for credit, please visit us at https://bit.ly/3ZSymre. NCPD/ILNA credit will be available until May 27, 2024.Betting on BCMA in Multiple Myeloma: Oncology Nurse Principles for Delivering...

Betting on BCMA in Multiple Myeloma: Oncology Nurse Principles for Delivering...PVI, PeerView Institute for Medical Education
Recommended
Chair, Anthony R. Mato, MD, MSCE, Kristen Battiato, MSN, RN, AGNP-C, Dipti Gupta, MD, MPH, and Amber C. King, PharmD, BCOP, prepared useful Practice Aids pertaining to B-cell cancers for this CME/MOC/NCPD activity titled “Interprofessional Perspectives on Safety Management With Targeted Therapy for B-Cell Malignancies.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD information, and to apply for credit, please visit us at http://bit.ly/3cHX3zm. CME/MOC/NCPD credit will be available until October 3, 2023.Interprofessional Perspectives on Safety Management With Targeted Therapy for...

Interprofessional Perspectives on Safety Management With Targeted Therapy for...PVI, PeerView Institute for Medical Education
Chair & Presenter, Beth Faiman, PhD, MSN, APN-BC, AOCN, BMTCN, FAAN, FAPO, Donna D. Catamero, ANP-BC, OCN, CCRC, and Tiffany A. Richards, PhD, ANP-BC, AOCNP, prepared useful Practice Aids pertaining to multiple myeloma for this NCPD/ILNA activity titled “Betting on BCMA in Multiple Myeloma: Oncology Nurse Principles for Delivering Effective Care With BCMA Antibodies and Cellular Therapy.” For the full presentation, downloadable Practice Aids, and complete NCPD/ILNA information, and to apply for credit, please visit us at https://bit.ly/3ZSymre. NCPD/ILNA credit will be available until May 27, 2024.Betting on BCMA in Multiple Myeloma: Oncology Nurse Principles for Delivering...

Betting on BCMA in Multiple Myeloma: Oncology Nurse Principles for Delivering...PVI, PeerView Institute for Medical Education
Chair & Presenter, Robert Z. Orlowski, MD, PhD, Noa Biran, MD, and Ajay K. Nooka, MD, MPH, FACP, prepared useful Practice Aids pertaining to multiple myeloma for this CME/MOC/AAPA activity titled “The New ABCs of Myeloma Care: Enhancing Outcomes With CD38 Antibodies, BCMA Immunotherapy, and Unique Bispecific Platforms.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/AAPA information, and to apply for credit, please visit us at https://bit.ly/3MPq140. CME/MOC/AAPA credit will be available until July 3, 2024.The New ABCs of Myeloma Care: Enhancing Outcomes With CD38 Antibodies, BCMA I...

The New ABCs of Myeloma Care: Enhancing Outcomes With CD38 Antibodies, BCMA I...PVI, PeerView Institute for Medical Education
Chair, Amit Singal, MD, MS, Anthony El-Khoueiry, MD, Ahmed Omar Kaseb, MD, CMQ, and Anjana Pillai, MD, prepared useful Practice Aids pertaining to hepatocellular carcinoma for this CME/MOC activity titled “It Takes a Team for HCC: Improving Outcomes Through Multidisciplinary Collaboration & Modern Therapeutics.” For the full presentation, downloadable Practice Aids, and complete CME/MOC information, and to apply for credit, please visit us at https://bit.ly/3AbNtC5. CME/MOC credit will be available until December 4, 2023.
It Takes a Team for HCC: Improving Outcomes Through Multidisciplinary Collabo...

It Takes a Team for HCC: Improving Outcomes Through Multidisciplinary Collabo...PVI, PeerView Institute for Medical Education
Dr. Cathy Eng delivers updates on current trials and studies impacting the treatment of colon and rectal cancer. Colorectal Cancer Research & Treatment News - recap from the May 2014 ASCO co...

Colorectal Cancer Research & Treatment News - recap from the May 2014 ASCO co...Fight Colorectal Cancer
Chair and Presenter Neal D. Shore, MD, FACS, Sia Daneshmand, MD, and Guru P. Sonpavde, MD, prepared useful Practice Aids pertaining to bladder cancer for this CME/MOC/AAPA activity titled “Pioneering Precision Medicine in Bladder Cancer: Multidisciplinary Perspectives on Personalizing Patient Care.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/AAPA information, and to apply for credit, please visit us at https://bit.ly/40qGkJH. CME/MOC/AAPA credit will be available until December 28, 2024.Pioneering Precision Medicine in Bladder Cancer: Multidisciplinary Perspectiv...

Pioneering Precision Medicine in Bladder Cancer: Multidisciplinary Perspectiv...PVI, PeerView Institute for Medical Education
Luciano J. Costa, MD, PhD, prepared useful Practice Aids pertaining to multiple myeloma for this CME activity titled "Exploring Clinical Decisions in the New Era of Myeloma Management: The Impact of Novel Platforms and Agent Classes Across the Spectrum of Care." For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2Dypn7b. CME credit will be available until March 12, 2020.Exploring Clinical Decisions in the New Era of Myeloma Management: The Impact...

Exploring Clinical Decisions in the New Era of Myeloma Management: The Impact...PVI, PeerView Institute for Medical Education
Co-Chairs Srdan Verstovsek, MD, PhD, and Ruben A. Mesa, MD, FACP, prepared useful Practice Aids pertaining to myelofibrosis for this CME activity titled “Understanding the Clinical Spectrum of Myelofibrosis: Expert Perspectives on Molecular Biology, JAK Inhibitors, and Emerging Therapeutics.” For the full presentation, downloadable Practice Aids, and complete CME information, and to apply for credit, please visit us at https://bit.ly/3wzK6zG. CME credit will be available until October 9, 2022.Understanding the Clinical Spectrum of Myelofibrosis: Expert Perspectives on ...

Understanding the Clinical Spectrum of Myelofibrosis: Expert Perspectives on ...PVI, PeerView Institute for Medical Education
Chair A. Michael Lincoff, MD, discusses obesity in this CME activity titled “Exploring the Evidence: Improving Cardiovascular Outcomes and the Role of Weight Loss Pharmacotherapy.” For the full presentation, downloadable Practice Aids, and complete CME information, and to apply for credit, please visit us at https://bit.ly/3KAO98K. CME credit will be available until April 25, 2025.Exploring the Evidence: Improving Cardiovascular Outcomes and the Role of Wei...

Exploring the Evidence: Improving Cardiovascular Outcomes and the Role of Wei...PVI, PeerView Institute for Medical Education
Co-Chairs and Planners Saakshi Khattri, MBBS, MD, FAAD, FACR, Marla Dubinsky, MD, Emma Guttman-Yassky, MD, PhD, and Alexis Ogdie, MD, MSCE, discuss immune-mediated inflammatory diseases in this CME/AAPA activity titled “Interdisciplinary Approaches to Management of Immune-Mediated Inflammatory Diseases: Addressing Shared Pathophysiology With JAK Inhibitors.” For the full presentation, downloadable Practice Aids, and complete CME/AAPA information, and to apply for credit, please visit us at https://bit.ly/3JhsIZ7. CME/AAPA credit will be available until April 24, 2025.Interdisciplinary Approaches to Management of Immune-Mediated Inflammatory Di...

Interdisciplinary Approaches to Management of Immune-Mediated Inflammatory Di...PVI, PeerView Institute for Medical Education
More Related Content
Similar to Hitting the BCMA Target in Multiple Myeloma: Insights on CAR-T Therapy and Innovative Antibody Options
Chair & Presenter, Robert Z. Orlowski, MD, PhD, Noa Biran, MD, and Ajay K. Nooka, MD, MPH, FACP, prepared useful Practice Aids pertaining to multiple myeloma for this CME/MOC/AAPA activity titled “The New ABCs of Myeloma Care: Enhancing Outcomes With CD38 Antibodies, BCMA Immunotherapy, and Unique Bispecific Platforms.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/AAPA information, and to apply for credit, please visit us at https://bit.ly/3MPq140. CME/MOC/AAPA credit will be available until July 3, 2024.The New ABCs of Myeloma Care: Enhancing Outcomes With CD38 Antibodies, BCMA I...

The New ABCs of Myeloma Care: Enhancing Outcomes With CD38 Antibodies, BCMA I...PVI, PeerView Institute for Medical Education
Chair, Amit Singal, MD, MS, Anthony El-Khoueiry, MD, Ahmed Omar Kaseb, MD, CMQ, and Anjana Pillai, MD, prepared useful Practice Aids pertaining to hepatocellular carcinoma for this CME/MOC activity titled “It Takes a Team for HCC: Improving Outcomes Through Multidisciplinary Collaboration & Modern Therapeutics.” For the full presentation, downloadable Practice Aids, and complete CME/MOC information, and to apply for credit, please visit us at https://bit.ly/3AbNtC5. CME/MOC credit will be available until December 4, 2023.
It Takes a Team for HCC: Improving Outcomes Through Multidisciplinary Collabo...

It Takes a Team for HCC: Improving Outcomes Through Multidisciplinary Collabo...PVI, PeerView Institute for Medical Education
Dr. Cathy Eng delivers updates on current trials and studies impacting the treatment of colon and rectal cancer. Colorectal Cancer Research & Treatment News - recap from the May 2014 ASCO co...

Colorectal Cancer Research & Treatment News - recap from the May 2014 ASCO co...Fight Colorectal Cancer
Chair and Presenter Neal D. Shore, MD, FACS, Sia Daneshmand, MD, and Guru P. Sonpavde, MD, prepared useful Practice Aids pertaining to bladder cancer for this CME/MOC/AAPA activity titled “Pioneering Precision Medicine in Bladder Cancer: Multidisciplinary Perspectives on Personalizing Patient Care.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/AAPA information, and to apply for credit, please visit us at https://bit.ly/40qGkJH. CME/MOC/AAPA credit will be available until December 28, 2024.Pioneering Precision Medicine in Bladder Cancer: Multidisciplinary Perspectiv...

Pioneering Precision Medicine in Bladder Cancer: Multidisciplinary Perspectiv...PVI, PeerView Institute for Medical Education
Luciano J. Costa, MD, PhD, prepared useful Practice Aids pertaining to multiple myeloma for this CME activity titled "Exploring Clinical Decisions in the New Era of Myeloma Management: The Impact of Novel Platforms and Agent Classes Across the Spectrum of Care." For the full presentation, monograph, complete CME information, and to apply for credit, please visit us at http://bit.ly/2Dypn7b. CME credit will be available until March 12, 2020.Exploring Clinical Decisions in the New Era of Myeloma Management: The Impact...

Exploring Clinical Decisions in the New Era of Myeloma Management: The Impact...PVI, PeerView Institute for Medical Education
Co-Chairs Srdan Verstovsek, MD, PhD, and Ruben A. Mesa, MD, FACP, prepared useful Practice Aids pertaining to myelofibrosis for this CME activity titled “Understanding the Clinical Spectrum of Myelofibrosis: Expert Perspectives on Molecular Biology, JAK Inhibitors, and Emerging Therapeutics.” For the full presentation, downloadable Practice Aids, and complete CME information, and to apply for credit, please visit us at https://bit.ly/3wzK6zG. CME credit will be available until October 9, 2022.Understanding the Clinical Spectrum of Myelofibrosis: Expert Perspectives on ...

Understanding the Clinical Spectrum of Myelofibrosis: Expert Perspectives on ...PVI, PeerView Institute for Medical Education
Similar to Hitting the BCMA Target in Multiple Myeloma: Insights on CAR-T Therapy and Innovative Antibody Options (20)
The New ABCs of Myeloma Care: Enhancing Outcomes With CD38 Antibodies, BCMA I...

The New ABCs of Myeloma Care: Enhancing Outcomes With CD38 Antibodies, BCMA I...
Report Back from 2023 San Antonio Breast Cancer Symposium (SABCS)

Report Back from 2023 San Antonio Breast Cancer Symposium (SABCS)
It Takes a Team for HCC: Improving Outcomes Through Multidisciplinary Collabo...

It Takes a Team for HCC: Improving Outcomes Through Multidisciplinary Collabo...
Colorectal Cancer Research & Treatment News - recap from the May 2014 ASCO co...

Colorectal Cancer Research & Treatment News - recap from the May 2014 ASCO co...
Summary of PROTAC And Other Targeted Protein Degradation Technologies.pdf

Summary of PROTAC And Other Targeted Protein Degradation Technologies.pdf
New drug approvals & upcoming fda approvals 2021

New drug approvals & upcoming fda approvals 2021
Pioneering Precision Medicine in Bladder Cancer: Multidisciplinary Perspectiv...

Pioneering Precision Medicine in Bladder Cancer: Multidisciplinary Perspectiv...
Summary of Approved HER2 ADCs on The Market & in Clinical Trials.pdf

Summary of Approved HER2 ADCs on The Market & in Clinical Trials.pdf
Exploring Clinical Decisions in the New Era of Myeloma Management: The Impact...

Exploring Clinical Decisions in the New Era of Myeloma Management: The Impact...
Understanding the Clinical Spectrum of Myelofibrosis: Expert Perspectives on ...

Understanding the Clinical Spectrum of Myelofibrosis: Expert Perspectives on ...
Non-Metastatic Castration-Resistant Prostate Cancer Treated with Androgen Rec...

Non-Metastatic Castration-Resistant Prostate Cancer Treated with Androgen Rec...
Non-Metastatic Castration-Resistant Prostate Cancer Treated with Androgen Rec...

Non-Metastatic Castration-Resistant Prostate Cancer Treated with Androgen Rec...
More from PVI, PeerView Institute for Medical Education
Chair A. Michael Lincoff, MD, discusses obesity in this CME activity titled “Exploring the Evidence: Improving Cardiovascular Outcomes and the Role of Weight Loss Pharmacotherapy.” For the full presentation, downloadable Practice Aids, and complete CME information, and to apply for credit, please visit us at https://bit.ly/3KAO98K. CME credit will be available until April 25, 2025.Exploring the Evidence: Improving Cardiovascular Outcomes and the Role of Wei...

Exploring the Evidence: Improving Cardiovascular Outcomes and the Role of Wei...PVI, PeerView Institute for Medical Education
Co-Chairs and Planners Saakshi Khattri, MBBS, MD, FAAD, FACR, Marla Dubinsky, MD, Emma Guttman-Yassky, MD, PhD, and Alexis Ogdie, MD, MSCE, discuss immune-mediated inflammatory diseases in this CME/AAPA activity titled “Interdisciplinary Approaches to Management of Immune-Mediated Inflammatory Diseases: Addressing Shared Pathophysiology With JAK Inhibitors.” For the full presentation, downloadable Practice Aids, and complete CME/AAPA information, and to apply for credit, please visit us at https://bit.ly/3JhsIZ7. CME/AAPA credit will be available until April 24, 2025.Interdisciplinary Approaches to Management of Immune-Mediated Inflammatory Di...

Interdisciplinary Approaches to Management of Immune-Mediated Inflammatory Di...PVI, PeerView Institute for Medical Education
Co-Chairs Alicia K. Morgans, MD, MPH, and Neal D. Shore, MD, FACS, discuss prostate cancer in this CME/MOC/NCPD/CPE/AAPA/IPCE activity titled “Treatment Advances and Individualized Therapeutic Strategies in Prostate Cancer: Expert Insights on Key Evidence, Practical Tips for Personalized Therapy, and Clinical Integration Approaches.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/CPE/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3SQrJ6G. CME/MOC/NCPD/CPE/AAPA/IPCE credit will be available until April 24, 2025.Treatment Advances and Individualized Therapeutic Strategies in Prostate Canc...

Treatment Advances and Individualized Therapeutic Strategies in Prostate Canc...PVI, PeerView Institute for Medical Education
Co-Chairs Prof. Nicolas Girard, MD, PhD, and Aaron Lisberg, MD, discuss NSCLC in this CME/MOC/NCPD/AAPA/IPCE activity titled “Charting a New Path to Better Outcomes With TROP2-Targeting ADCs in Lung Cancer: Unveiling Potential, Shaping Tomorrow.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3RmX3dU. CME/MOC/NCPD/AAPA/IPCE credit will be available until April 24, 2025.Charting a New Path to Better Outcomes With TROP2-Targeting ADCs in Lung Canc...

Charting a New Path to Better Outcomes With TROP2-Targeting ADCs in Lung Canc...PVI, PeerView Institute for Medical Education
Co-Chairs, Joseph K. Han, MD, and Seth J. Isaacs, MD, prepared useful Practice Aids pertaining to chronic rhinosinusitis with nasal polyps for this CME/MOC/CC/AAPA/IPCE activity titled “Biologics in CRSwNP: Putting a Paradigm Shift Into Practice.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/CC/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3Tq6n1G. CME/MOC/CC/AAPA/IPCE credit will be available until May 6, 2025.Biologics in CRSwNP: Putting a Paradigm Shift Into Practice

Biologics in CRSwNP: Putting a Paradigm Shift Into PracticePVI, PeerView Institute for Medical Education
Co-Chairs, Joseph K. Han, MD, and Seth J. Isaacs, MD, discuss chronic rhinosinusitis with nasal polyps in this CME/MOC/CC/AAPA/IPCE activity titled “Biologics in CRSwNP: Putting a Paradigm Shift Into Practice.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/CC/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3Tq6n1G. CME/MOC/CC/AAPA/IPCE credit will be available until May 6, 2025.Biologics in CRSwNP: Putting a Paradigm Shift Into Practice

Biologics in CRSwNP: Putting a Paradigm Shift Into PracticePVI, PeerView Institute for Medical Education
Co-Chairs, R. Donald Harvey, PharmD, BCOP, FCCP, FHOPA, FASCO, Zahra Mahmoudjafari, PharmD, MBA, BCOP, FHOPA, and James Davis, PharmD, BCOP, discuss multiple myeloma in this CME/CPE/IPCE activity titled “Prescriptions for Successful Myeloma Care: Pharmacy Strategies for Delivering Effective Therapy With Antibody Platforms.” For the full presentation, downloadable Practice Aids, and complete CME/CPE/IPCE information, and to apply for credit, please visit us at https://bit.ly/4aa0iMX. CME/CPE/IPCE credit will be available until May 2, 2025.Prescriptions for Successful Myeloma Care: Pharmacy Strategies for Delivering...

Prescriptions for Successful Myeloma Care: Pharmacy Strategies for Delivering...PVI, PeerView Institute for Medical Education
Co-Chairs, Carlos G. Romo, MD, and Aimee Sato, MD, discuss Neurofibromatosis in this CME/MOC activity titled “Precision & Progress Against NF1: Solutions for Better Outcomes With MEKi & Multimodal Care for NF1 pNF and Other Tumors.” For the full presentation, downloadable Practice Aids, and complete CME/MOC information, and to apply for credit, please visit us at https://bit.ly/3SZRz8p. CME/MOC credit will be available until May 2, 2025.Precision & Progress Against NF1: Solutions for Better Outcomes With MEKi & M...

Precision & Progress Against NF1: Solutions for Better Outcomes With MEKi & M...PVI, PeerView Institute for Medical Education
Chair and Presenters Kathleen N. Moore, MD, MS, Floor J. Backes, MD, and Bhavana Pothuri, MD, MS, prepared useful Practice Aids pertaining to endometrial cancer for this CME/MOC/NCPD/AAPA/IPCE activity titled “Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Potential of Immunotherapy, ADCs, PARP Inhibitors, and Other Emerging Treatment Strategies.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3SjJyuH. CME/MOC/NCPD/AAPA/IPCE credit will be available until April 17, 2025.Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...

Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...PVI, PeerView Institute for Medical Education
Chair and Presenters Kathleen N. Moore, MD, MS, Floor J. Backes, MD, and Bhavana Pothuri, MD, MS, discuss endometrial cancer in this CME/MOC/NCPD/AAPA/IPCE activity titled “Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Potential of Immunotherapy, ADCs, PARP Inhibitors, and Other Emerging Treatment Strategies.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3SjJyuH. CME/MOC/NCPD/AAPA/IPCE credit will be available until April 17, 2025.Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...

Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...PVI, PeerView Institute for Medical Education
Chair and Presenters Bradley J. Monk, MD, FACS, FACOG, Kathleen N. Moore, MD, MS, and Ana Oaknin, MD, PhD, discuss gynecologic cancers in this CME/MOC/NCPD/AAPA/IPCE activity titled “Advancing ADCs in Gynecologic Cancers: Expert Insights on Recent Evidence, Implementation Strategies, and Patient Care.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/4a56tly. CME/MOC/NCPD/AAPA/IPCE credit will be available until April 16, 2025.Advancing ADCs in Gynecologic Cancers: Expert Insights on Recent Evidence, Im...

Advancing ADCs in Gynecologic Cancers: Expert Insights on Recent Evidence, Im...PVI, PeerView Institute for Medical Education
Chair Lecia V. Sequist, MD, MPH, and Patrick Nana-Sinkam, MD, FCCP, prepared useful Practice Aids pertaining to lung cancer for this CME/MOC/AAPA/IPCE activity titled “Screening and Early Intervention as the Keys to Success in Lung Cancer: A Practical Approach to Implementing Lung Cancer Screening for High-Risk Individuals.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/46VvwoP. CME/MOC/AAPA/IPCE credit will be available until April 16, 2025.Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...

Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...PVI, PeerView Institute for Medical Education
Chair Lecia V. Sequist, MD, MPH, and Patrick Nana-Sinkam, MD, FCCP, discuss lung cancer screening in this CME/MOC/AAPA/IPCE activity titled “Screening and Early Intervention as the Keys to Success in Lung Cancer: A Practical Approach to Implementing Lung Cancer Screening for High-Risk Individuals.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/46VvwoP. CME/MOC/AAPA/IPCE credit will be available until April 16, 2025.Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...

Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...PVI, PeerView Institute for Medical Education
Chair and Presenter, Olalekan Oluwole, MBBS, MPH, Veronika Bachanova, MD, PhD, and David L. Porter, MD, prepared useful Practice Aids pertaining to CAR-T therapy for this CME/NCPD activity titled “Democratizing the CAR-T Experience: The Principles and Practice of Outpatient Cellular Therapy.” For the full presentation, downloadable Practice Aids, and complete CME/NCPD information, and to apply for credit, please visit us at https://bit.ly/3TfIABM. CME/NCPD credit will be available until April 15, 2025.Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...

Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...PVI, PeerView Institute for Medical Education
Chair and Presenter, Olalekan Oluwole, MBBS, MPH, Veronika Bachanova, MD, PhD, and David L. Porter, MD, discuss CAR-T therapy in this CME/NCPD activity titled “Democratizing the CAR-T Experience: The Principles and Practice of Outpatient Cellular Therapy.” For the full presentation, downloadable Practice Aids, and complete CME/NCPD information, and to apply for credit, please visit us at https://bit.ly/3TfIABM. CME/NCPD credit will be available until April 15, 2025.Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...

Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...PVI, PeerView Institute for Medical Education
Co-Chairs Lipika Goyal, MD, MPhil, and Riad Salem, MD, MBA, discuss HCC in this CME activity titled “The Convergence of Interventional Radiologists and Oncologists in HCC: Shared Decision-Making and Care Coordination at the Center of Personalized Care Across the Disease Continuum.” For the full presentation, downloadable Practice Aids, and complete CME information, and to apply for credit, please visit us at https://bit.ly/48BAasz. CME credit will be available until April 26, 2025.The Convergence of Interventional Radiologists and Oncologists in HCC: Shared...

The Convergence of Interventional Radiologists and Oncologists in HCC: Shared...PVI, PeerView Institute for Medical Education
Chair, Richard K. Bogan, MD, FCCP, FAASM, discusses sleep disorders in this CME/MOC activity titled “Navigating Narcolepsy in Family Practice: Patient-Centered Strategies to Optimize the Experience and Outcomes of Treatment.” For the full presentation, downloadable Practice Aids, and complete CME/MOC information, and to apply for credit, please visit us at https://bit.ly/48QOONd. CME/MOC credit will be available until April 9, 2025.
Navigating Narcolepsy in Family Practice: Patient-Centered Strategies to Opti...

Navigating Narcolepsy in Family Practice: Patient-Centered Strategies to Opti...PVI, PeerView Institute for Medical Education
Chair and Presenter, Jennifer Wargo, MD, MMSc, Charlotte E. Ariyan, MD, PhD, and Hussein Tawbi, MD, PhD, discuss melanoma in this CME/MOC/AAPA/IPCE activity titled “New Chapters in the Immunotherapy Story for Melanoma: Collaborative Care and Next Steps With Adjuvant and Neoadjuvant Therapy.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3T70bfi. CME/MOC/AAPA/IPCE credit will be available until April 21, 2025.New Chapters in the Immunotherapy Story for Melanoma: Collaborative Care and ...

New Chapters in the Immunotherapy Story for Melanoma: Collaborative Care and ...PVI, PeerView Institute for Medical Education
Chair and Presenters Laura S. Dominici, MD, FACS, Albert Henry Diehl, III, MD, FACS, and Jane L. Meisel, MD, discuss breast cancer in this CME/MOC/CC activity titled “Unraveling the Complex Choices in Early Breast Cancer: A Roadmap to Informed Multidisciplinary Decisions About Assessment and Treatment.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/CC information, and to apply for credit, please visit us at https://bit.ly/42BZBZE. CME/MOC/CC credit will be available until April 21, 2025.Unraveling the Complex Choices in Early Breast Cancer: A Roadmap to Informed ...

Unraveling the Complex Choices in Early Breast Cancer: A Roadmap to Informed ...PVI, PeerView Institute for Medical Education
Chair, Meghan C. Thompson, MD, discusses chronic lymphocytic leukemia in this CME/MOC/NCPD/CPE/AAPA/IPCE activity titled “From Resistance to Resilience in R/R CLL: Sequencing Strategies for Achieving Effective Continuous Care.” For the full presentation, downloadable Practice Aids, and complete CME/MOC/NCPD/CPE/AAPA/IPCE information, and to apply for credit, please visit us at https://bit.ly/3uoWOWG. CME/MOC/NCPD/CPE/AAPA/IPCE credit will be available until April 4, 2025.From Resistance to Resilience in R/R CLL: Sequencing Strategies for Achieving...

From Resistance to Resilience in R/R CLL: Sequencing Strategies for Achieving...PVI, PeerView Institute for Medical Education
More from PVI, PeerView Institute for Medical Education (20)
Exploring the Evidence: Improving Cardiovascular Outcomes and the Role of Wei...

Exploring the Evidence: Improving Cardiovascular Outcomes and the Role of Wei...
Interdisciplinary Approaches to Management of Immune-Mediated Inflammatory Di...

Interdisciplinary Approaches to Management of Immune-Mediated Inflammatory Di...
Treatment Advances and Individualized Therapeutic Strategies in Prostate Canc...

Treatment Advances and Individualized Therapeutic Strategies in Prostate Canc...
Charting a New Path to Better Outcomes With TROP2-Targeting ADCs in Lung Canc...

Charting a New Path to Better Outcomes With TROP2-Targeting ADCs in Lung Canc...
Biologics in CRSwNP: Putting a Paradigm Shift Into Practice

Biologics in CRSwNP: Putting a Paradigm Shift Into Practice
Biologics in CRSwNP: Putting a Paradigm Shift Into Practice

Biologics in CRSwNP: Putting a Paradigm Shift Into Practice
Prescriptions for Successful Myeloma Care: Pharmacy Strategies for Delivering...

Prescriptions for Successful Myeloma Care: Pharmacy Strategies for Delivering...
Precision & Progress Against NF1: Solutions for Better Outcomes With MEKi & M...

Precision & Progress Against NF1: Solutions for Better Outcomes With MEKi & M...
Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...

Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...
Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...

Redefining Endometrial and Ovarian Carcinoma Care: Maximizing the Clinical Po...
Advancing ADCs in Gynecologic Cancers: Expert Insights on Recent Evidence, Im...

Advancing ADCs in Gynecologic Cancers: Expert Insights on Recent Evidence, Im...
Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...

Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...
Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...

Screening and Early Intervention as the Keys to Success in Lung Cancer: A Pra...
Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...

Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...
Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...

Democratizing the CAR-T Experience: The Principles and Practice of Outpatient...
The Convergence of Interventional Radiologists and Oncologists in HCC: Shared...

The Convergence of Interventional Radiologists and Oncologists in HCC: Shared...
Navigating Narcolepsy in Family Practice: Patient-Centered Strategies to Opti...

Navigating Narcolepsy in Family Practice: Patient-Centered Strategies to Opti...
New Chapters in the Immunotherapy Story for Melanoma: Collaborative Care and ...

New Chapters in the Immunotherapy Story for Melanoma: Collaborative Care and ...
Unraveling the Complex Choices in Early Breast Cancer: A Roadmap to Informed ...

Unraveling the Complex Choices in Early Breast Cancer: A Roadmap to Informed ...
From Resistance to Resilience in R/R CLL: Sequencing Strategies for Achieving...

From Resistance to Resilience in R/R CLL: Sequencing Strategies for Achieving...
Recently uploaded
Book Paid Powai Call Girls Mumbai 𖠋 9930245274 𖠋Low Budget Full Independent High Profile Call Girl 24×7
Booking Contact Details
WhatsApp Chat: +91-9930245274
Mumbai Escort Service includes providing maximum physical satisfaction to their clients as well as engaging conversation that keeps your time enjoyable and entertaining. Plus they look fabulously elegant; making an impressionable.
Independent Escorts Mumbai understands the value of confidentiality and discretion - they will go the extra mile to meet your needs. Simply contact them via text messaging or through their online profiles; they'd be more than delighted to accommodate any request or arrange a romantic date or fun-filled night together.
We provide -
Flexibility
Choices and options
Lists of many beauty fantasies
Turn your dream into reality
Perfect companionship
Cheap and convenient
In-call and Out-call services
And many more.
29-04-24 (Smt)Book Paid Powai Call Girls Mumbai 𖠋 9930245274 𖠋Low Budget Full Independent H...

Book Paid Powai Call Girls Mumbai 𖠋 9930245274 𖠋Low Budget Full Independent H...Call Girls in Nagpur High Profile
Recently uploaded (20)
Call Girls Tirupati Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Tirupati Just Call 8250077686 Top Class Call Girl Service Available
Call Girls Bangalore Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Bangalore Just Call 8250077686 Top Class Call Girl Service Available
All Time Service Available Call Girls Marine Drive 📳 9820252231 For 18+ VIP C...

All Time Service Available Call Girls Marine Drive 📳 9820252231 For 18+ VIP C...
Call Girls Service Jaipur {9521753030} ❤️VVIP RIDDHI Call Girl in Jaipur Raja...

Call Girls Service Jaipur {9521753030} ❤️VVIP RIDDHI Call Girl in Jaipur Raja...
(Low Rate RASHMI ) Rate Of Call Girls Jaipur ❣ 8445551418 ❣ Elite Models & Ce...

(Low Rate RASHMI ) Rate Of Call Girls Jaipur ❣ 8445551418 ❣ Elite Models & Ce...
(👑VVIP ISHAAN ) Russian Call Girls Service Navi Mumbai🖕9920874524🖕Independent...

(👑VVIP ISHAAN ) Russian Call Girls Service Navi Mumbai🖕9920874524🖕Independent...
Top Rated Bangalore Call Girls Mg Road ⟟ 9332606886 ⟟ Call Me For Genuine S...

Top Rated Bangalore Call Girls Mg Road ⟟ 9332606886 ⟟ Call Me For Genuine S...
Top Rated Bangalore Call Girls Ramamurthy Nagar ⟟ 9332606886 ⟟ Call Me For G...

Top Rated Bangalore Call Girls Ramamurthy Nagar ⟟ 9332606886 ⟟ Call Me For G...
O898O367676 Call Girls In Ahmedabad Escort Service Available 24×7 In Ahmedabad

O898O367676 Call Girls In Ahmedabad Escort Service Available 24×7 In Ahmedabad
Call Girls in Delhi Triveni Complex Escort Service(🔝))/WhatsApp 97111⇛47426

Call Girls in Delhi Triveni Complex Escort Service(🔝))/WhatsApp 97111⇛47426
Best Rate (Hyderabad) Call Girls Jahanuma ⟟ 8250192130 ⟟ High Class Call Girl...

Best Rate (Hyderabad) Call Girls Jahanuma ⟟ 8250192130 ⟟ High Class Call Girl...
Call Girls Bhubaneswar Just Call 9907093804 Top Class Call Girl Service Avail...

Call Girls Bhubaneswar Just Call 9907093804 Top Class Call Girl Service Avail...
Call Girls Agra Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Agra Just Call 8250077686 Top Class Call Girl Service Available
Call Girls Visakhapatnam Just Call 9907093804 Top Class Call Girl Service Ava...

Call Girls Visakhapatnam Just Call 9907093804 Top Class Call Girl Service Ava...
VIP Service Call Girls Sindhi Colony 📳 7877925207 For 18+ VIP Call Girl At Th...

VIP Service Call Girls Sindhi Colony 📳 7877925207 For 18+ VIP Call Girl At Th...
The Most Attractive Hyderabad Call Girls Kothapet 𖠋 9332606886 𖠋 Will You Mis...

The Most Attractive Hyderabad Call Girls Kothapet 𖠋 9332606886 𖠋 Will You Mis...
Top Rated Bangalore Call Girls Richmond Circle ⟟ 9332606886 ⟟ Call Me For Ge...

Top Rated Bangalore Call Girls Richmond Circle ⟟ 9332606886 ⟟ Call Me For Ge...
Call Girls Guntur Just Call 8250077686 Top Class Call Girl Service Available

Call Girls Guntur Just Call 8250077686 Top Class Call Girl Service Available
Book Paid Powai Call Girls Mumbai 𖠋 9930245274 𖠋Low Budget Full Independent H...

Book Paid Powai Call Girls Mumbai 𖠋 9930245274 𖠋Low Budget Full Independent H...
Russian Call Girls Service Jaipur {8445551418} ❤️PALLAVI VIP Jaipur Call Gir...

Russian Call Girls Service Jaipur {8445551418} ❤️PALLAVI VIP Jaipur Call Gir...
Hitting the BCMA Target in Multiple Myeloma: Insights on CAR-T Therapy and Innovative Antibody Options
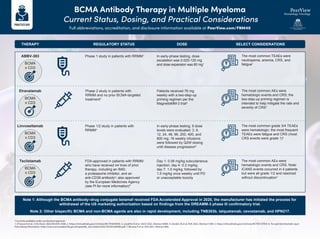
- 1. BCMA Antibody Therapy in Multiple Myeloma Current Status, Dosing, and Practical Considerations Full abbreviations, accreditation, and disclosure information available at PeerView.com/FNM40 a Currently available under accelerated approval. 1. D'Souza A et al. J Clin Oncol. 2022;40:3576-3586. 2. https://clinicaltrials.gov/ct2/show/NCT04649359. 3. Lesokhin A et al. ASCO 2022. Abstract 8006. 4. Zonder JA et al. EHA 2022. Abstract S189. 5. https://clinicaltrials.gov/ct2/show/NCT05137054. 6. Tecvayli (teclistamab-cqyv) Prescribing Information. https://www.accessdata.fda.gov/drugsatfda_docs/label/2022/761291s000lbl.pdf. 7. Moreau P et al. ASH 2021. Abstract 896. REGULATORY STATUS Phase 2 study in patients with RRMM and no prior BCMA-targeted treatment2 Phase 1/2 study in patients with RRMM4 FDA-approved in patients with RRMM who have received ≥4 lines of prior therapy, including an IMiD, a proteasome inhibitor, and an anti-CD38 antibodya ; also approved by the European Medicines Agency (see PI for more information)6 Phase 1 study in patients with RRMM1 DOSE Patients received 76 mg weekly with a two-step-up priming regimen per the MagnetisMM-3 trial3 In early-phase testing, 9 dose levels were evaluated: 3, 6, 12, 24, 48, 96, 200, 400, and 800 mg; 16 weekly infusions were followed by Q2W dosing until disease progression5 Day 1: 0.06 mg/kg subcutaneous injection; day 4: 0.3 mg/kg; day 7: 1.5 mg/kg; followed by 1.5 mg/kg once weekly until PD or unacceptable toxicity In early-phase testing, dose escalation was 0.025-120 mg and dose expansion was 60 mg1 SELECT CONSIDERATIONS The most common AEs were hematologic events and CRS; the two-step-up priming regimen is intended to help mitigate the rate and severity of CRS3 The most common grade 3/4 TEAEs were hematologic; the most frequent TEAEs were fatigue and CRS (most CRS events were grade 1)4 The most common AEs were hematologic events and CRS. Note: ICANS events occurred in 4 patients but were all grade 1/2 and resolved without discontinuation7 The most common TEAEs were neutropenia, anemia, CRS, and fatigue1 THERAPY Elranatamab BCMA x CD3 Linvoseltamab BCMA x CD3 Teclistamab BCMA x CD3 ABBV-383 BCMA x CD3 Note 1: Although the BCMA antibody–drug conjugate belamaf received FDA Accelerated Approval in 2020, the manufacturer has initiated the process for withdrawal of the US marketing authorization based on findings from the DREAMM-3 phase III confirmatory trial. Note 2: Other bispecific BCMA and non-BCMA agents are also in rapid development, including TNB383b, talquetamab, cevostamab, and HPN217.
- 2. CAR-T Cell Therapy in Multiple Myeloma Current Status, Dosing, and Practical Considerations Full abbreviations, accreditation, and disclosure information available at PeerView.com/FNM40 1. Carvykti (ciltacabtagene autoleucel) Prescribing Information. https://www.fda.gov/media/156560/download. 2. Abecma (idecabtagene vicleucel) Prescribing Information. https://www.fda.gov/media/147055/download. REMS Cilta-Cel1 Approved in RRMM after ≥4 prior therapies, including an anti-CD38 mAb, a proteasome inhibitor, and an IMiD Recommended Dose Range 0.5-1.0 × 106 CAR-positive viable T cells (maximum dose of 1.0 × 108 CAR-positive viable T cells per single-dose infusion) Recommended Dose Range 300-460 × 106 CAR-positive viable T cells Ide-Cel2 Approved in RRMM after ≥4 prior therapies, including an anti-CD38 mAb, a proteasome inhibitor, and an IMiD General Principles for CAR-T Therapy Referral to a certified healthcare facility is required for collection of patient’s cells and administration of CAR-T therapy Avoid prophylactic use of dexamethasone or other systemic corticosteroids Premedicate with acetaminophen and an H1 antihistamine Monitor for CRS and ICANS and confirm tocilizumab availability before infusion Ide-cel and cilta-cel are available only through a restricted program under a Risk Evaluation and Mitigation Strategy (REMS) Monitor for neurologic events, hemophagocytic lymphohistiocytosis/ macrophage activation syndrome, and cytopenias Administer a lymphodepleting chemotherapy regimen of cyclophosphamide and fludarabine before CAR-T infusion Do not use a leukodepleting filter when administering
