A MasterClass on New Avenues in Asthma Management: Finding the Right Patients for Targeted Therapies
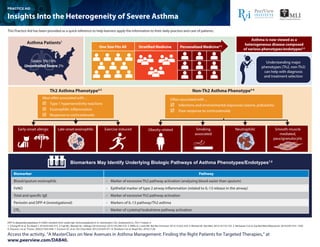
- 1. DPP-4: dipeptidyl peptidase-4; FeNO: exhaled nitric oxide; IgE: immunoglobulin E; IL: interleukin; LTE4: leukotriene E4; Th2: T helper 2. 1. Chung KF et al. Eur Respir J. 2014;43:343-373. 2. Fajt ML, Wenzel SE. J Allergy Clin Immunol. 2015;135:299-310. 3. Willis JC, Lord GM. Nat Rev Immunol. 2015;15:323-329. 4. Wenzel SE. Nat Med. 2012;18:716-725. 5. Mohanan S et al. Exp Biol Med (Maywood). 2014;239:1531-1540. 6. Douwes J et al. Thorax. 2002;57:643-648. 7. Erzurum SC et al. Clin Chest Med. 2012;33:459‐471. 8. Shiobara S et al. Respir Res. 2016;17:28. Access the activity, “A MasterClass on New Avenues in Asthma Management: Finding the Right Patients for Targeted Therapies,” at www.peerview.com/DAB40. Th2 Asthma Phenotype4,5 Non-Th2 Asthma Phenotype4-6 Smoking associated Obesity relatedEarly-onset allergic Late-onset eosinophilic Exercise induced Neutrophilic Smooth-muscle mediated, paucigranulocytic Biomarkers May Identify Underlying Biologic Pathways of Asthma Phenotypes/Endotypes7,8 Asthma Patients1 One Size Fits All Stratified Medicine Personalized Medicine2,3 Most often associated with ... Type 1 hypersensitivity reactions Eosinophilic inflammation Response to corticosteroids Often associated with ... Infections and environmental exposures (ozone, pollutants) Poor response to corticosteroids Severe 5%-10% UncontrolledSevere 2% Asthma is now viewed as a heterogeneous disease composed of various phenotypes/endotypes2,4 Understanding major phenotypes (Th2, non-Th2) can help with diagnosis and treatment selection Biomarker Pathway Blood/sputum eosinophils • Marker of excessive Th2 pathway activation (analyzing blood easier than sputum) • Epithelial marker of type 2 airway inflammation (related to IL-13 release in the airway) • Marker of excessive Th2 pathway activation • Markers of IL-13 pathway/Th2 asthma • Marker of cysteinyl leukotriene pathway activation FeNO Total and specific IgE Periostin and DPP-4 (investigational) LTE4 Insights Into the Heterogeneity of Severe Asthma PRACTICE AID This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients.
- 2. CS: corticosteroid; FEV1: forced expiratory volume in 1 second; IgE: immunoglobulin E; IL: interleukin; SBM: subepithelial basement membrane; Th2: T helper 2; Th17: T helper 17. 1. Wenzel SE. Nat Med. 2012;18:716-725. 2. Mohanan S et al. Exp Biol Med (Maywood). 2014;239:1531-1540. 3. Douwes J et al. Thorax. 2002;57:643-648. Access the activity, “A MasterClass on New Avenues in Asthma Management: Finding the Right Patients for Targeted Therapies,” at www.peerview.com/DAB40. Early-onset allergic Late-onset eosinophilic Exercise induced Smoking associated Obesity related Smooth-muscle mediated, paucigranulocytic Neutrophilic Non-Th2 Asthma Phenotype1-3 Th2 Asthma Phenotype1,2 Genetics 17q12; Th2-related genes Response to Therapy CS-responsive; Th2-targeted Responsive to anti–IL-5 therapy and cysteinyl leukotriene modifiers; CS-refractory Responsive to cysteinyl leukotriene modifiers, β agonists, and antibody to IL-9 Responsive to weight loss, antioxidants, and possibly hormonal therapy Possibly responsive to macrolide antibiotics Pathobiology and Biomarkers Specific IgE; Th2 cytokines; thick SBM CS-refractory eosinophilia; IL-5 Mast-cell activation; Th2 cytokines; cysteinyl leukotrienes Lack of Th2 biomarkers; oxidative stress No Th2 inflammation Sputum neutrophilia; Th17 pathways; IL-8 Clinical and Physiological Features Allergic symptoms and other diseases Possible asthma- COPD overlap syndrome Sinusitis; less allergic Mild; intermittent with exercise Low FEV1 ; more air trapping Primarily affects women; very symptomatic; airway hyper-responsiveness less clear Natural History Early onset (~3-10 y); mild to severe Adult onset; often severe Adult onset This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. Features of Th2 and Non-Th2 Asthma Phenotypes PRACTICE AID
- 3. CRTh2: chemoattractant receptor-homologous molecule expressed on T-helper 2 cells; IgE: immunoglobulin E; IL-#R: interleukin-# receptor; TSLP: thymic stromal lymphopoietin. 1. Xolair (omalizumab) Prescribing Information. https://www.gene.com/download/pdf/xolair_prescribing.pdf. Accessed September 21, 2018. 2. Nucala (mepolizumab) Prescribing Information. http://www.accessdata.fda.gov/drugsatfda_docs/label/2015/125526Orig1s000Lbl.pdf. Accessed September 21, 2018. 3. Cinqair (reslizumab) Prescribing Information. http://cinqair.com/pdf/ PrescribingInformation.pdf. Accessed September 21, 2018. 4. Fasenra (benralizumab) Prescribing Information. https://www.azpicentral.com/fasenra/fasenra_pi.pdf#page=1. Accessed September 21, 2018. 5. Dupixent (dupilumab) Prescribing Information. https://www.regeneron.com/sites/default/files/Dupixent_FPI.pdf. Accessed October 22, 2018. 6. https://www.prnewswire.com/news-releases/ tezepelumab-granted-breakthrough-therapy-designation-by-us-fda-for-the-treatment-of-patients-with-severe-asthma-without-an-eosinophilic-phenotype-300708680.html. Accessed September 21, 2018. 7. https://clinicaltrials.gov/ct2/show/NCT03347279. Accessed September 21, 2018. 8. https://clinicaltrials.gov/ct2/show/NCT03406078. Accessed September 21, 2018. 9. Corren J et al. N Engl J Med. 2017;377:936-946. 10. https://clinicaltrials.gov/ct2/show/NCT02563067. Accessed September 21, 2018. 11. https://clinicaltrials.gov/ct2/show/NCT03052517. Accessed September 21, 2018. 12. https://clinicaltrials.gov/ct2/show/NCT02555683. Accessed September 21, 2018. Access the activity, “A MasterClass on New Avenues in Asthma Management: Finding the Right Patients for Targeted Therapies,” at www.peerview.com/DAB40. Agent/Target Indication/Current Status Route/Dosing Patients ≥6 y with moderate to severe persistent allergic asthma inadequately controlled with inhaled corticosteroids Subcutaneous 75 mg to 375 mg every 2-4 weeks (dosage based on IgE level and body weight) Add-on maintenance treatment of patients ≥12 y with severe asthma and an eosinophilic phenotype Subcutaneous 100 mg every 4 weeks Add-on maintenance treatment of patients ≥18 y with severe asthma and an eosinophilic phenotype Intravenous infusion 3 mg/kg every 4 weeks over 20-50 minutes Add-on maintenance treatment of patients ≥12 y with severe asthma and an eosinophilic phenotype Subcutaneous 30 mg every 4 weeks for first 3 doses, then every 8 weeks Add-on maintenance treatment of patients ≥12 years with moderate to severe asthma with an eosinophilic phenotype or with oral corticosteroid-dependent asthma Subcutaneous Initial dose of 400 mg followed by 200 mg every 2 weeks or initial dose of 600 mg followed by 300 mg every 2 weeks Phase 3 trials; breakthrough designation for noneosinophilic phenotype Subcutaneous 70 mg and 210 mg every 4 weeks and 280 mg every 2 weeks tested in phase 2 trials Phase 3 trials Oral Dupilumab5 IL-4Rα (IL-4/IL-13) Tezepelumab6-9 TSLP Fevipiprant10-12 CRTh2 Investigational Omalizumab1 IgE Mepolizumab2 IL-5 Reslizumab3 IL-5 Benralizumab4 IL-5Rα/βc Approved This Practice Aid has been provided as a quick reference to help learners apply the information to their daily practice and care of patients. A Guide to Biologic Therapies for Severe, Uncontrolled Asthma PRACTICE AID
