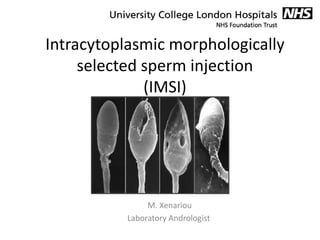
IMSI
- 1. Intracytoplasmic morphologically selected sperm injection (IMSI) M. Xenariou Laboratory Andrologist
- 2. Sperm morphology & IVF outcome • Head abnormalities mainly responsible for sperm fertilization deficiency • Flagellar abnormalities haven’t been shown to have any impact on fertilization (Berkovitz et al., 2006; Chemes et al., 2003) • Sperm nuclear DNA damage is closely related to male- derived repeated failed ICSI attempts (Tesarik et al., 2004; Tesarik 2005) • Presence of large nuclear vacuoles: negatively associated with natural male fertility potential (Bartoov et al., 1994; Mundi et al., 1994)
- 3. ICSI + MSOME = IMSI • ICSI (Palermo et al., 1992): inverted microscope magnification of x 400 • MSOME - motile sperm organellar morphology examination (Bartoov et al., 2002) The resulting magnification was based on 4 parameters: 1. Objective magnification x100 2. Magnification selector x1.5 3. Video coupler magnification x0.99 4. Calculated video magnification (ccd x monitor diagonal dimension) x 44.45 TOTAL MAGNIFICATION: 100 x 1.5 x 0.99 x 44.45 = x 6600 !!!
- 4. IMSI equipment IMSI can be achieved on a Nikon inverted microscope equipped with high resolution Nomarski optics (100x or 60x oil Plan Apo VC) enhanced by a videozoom and digital imaging. The final magnification of the system can be calculated as follows: (objective) X (magnification changer) X (videozoom) X (diagonal of image camera CCD chip diagonal)
- 5. ICSI vs IMSI
- 6. IMSI Sperm assessment (1) The MSOME criteria for the morphological normalcy of the sperm nucleus were defined as below: Smooth Symmetric Oval Configuration Homogeneity of the nuclear chromatin mass (no more than one vacuole / less than 4% of the nuclear area) The average length and width limits in 100 spermatozoa with a normally looking nucleus, are estimated as follow: Length: 4.75 ± 0.28 μm Width: 3.28 ± 0.20 μm
- 7. IMSI Sperm assessment (2)
- 8. Vacuoles (1) • Vacuole: concavity extending from the surface of the sperm head to the nucleus through the acrosome • Initially: considered as degenerative structures with no physiological significance – normal feature of sperm head (Fawcett, 1958; Chrzanowski 1966; Bedford 1967; Pedersen 1969)
- 9. Vacuoles (2) Post-IVF era: • Mundi et al., 1994: sperm nuclei in subfertile men contained more intranuclear vacuoles than fertile controls • ICSI with sperm with vacuoles = decreased PR, early miscarriage (Bartoov et al., 2001, 2002, 2003; Berkovitz et al., 2005, 2006; Franco et al., 2008; Antinori et al., 2008) Vacuoles are not just a polymorphism but pose a risk for an abnormality that is accompanied by DNA injury
- 10. Sperm grading: vacuoles • Vanderzwalmen et al., 2008: Grade I: absence of vacuoles Grade II: max. of 2 small vacuoles Grade III: >2 small vacuoles or at least 1 large Grade IV: abnormal head shapes or other abnormalities with or without vacuoles (identified even at x400 magnification)
- 11. Vacuoles & DNA fragmentation • Spermatozoa with large vacuoles should be avoided (Tanaka et al., 2012) • Spermatozoa with large vacuoles (>50% of the sperm head) & other potential abnormalities were described as having higher DNA fragmentation levels than normal spermatozoa (Franco et al., 2008) • Spermatozoa with multiples vacuoles (>4% of the head area) have higher DNA fragmentation levels than normal spermatozoa (Wilding et al., 2011) • Large vacuoles associated with DNA fragmentation (Oliveira et al., 2010; Hammoud et al., 2012) • No difference in DNA fragmentation between normal ones & morphometrically normal ones with a single, large vacuole (15%-25% of head area) (Watanabe et al., 2011; Boitrelle et al., 2011) • No difference in DNA fragmentation between spermatozoa with 1 large vacuole & non-selected ones (Perdrix et al., 2011)
- 12. Studies (1) • Prospective randomized trials with unselected pt - Antinori et al. , 2008 - Balaban et al., 2011 • Benefit of IMSI after ICSI implantation failure (without assessing influence of sperm abnormalities) - Berkovitz et al., 2006 - Bartoov et al., 2003 - Nadalini et al., 2009 - Oliveira et al., 2011 not significantly higher ongoing pregnancy, miscarriage & LBR after IMSI • Pt with isolated teratozoospermia - Knez et al., 2012 higher PR in IMSI group
- 13. Studies (2) • Comparable embryo development between IMSI & ICSI on Day 2 (Mauri et al., 2010; Oliveira et al., 2011) and Day 3 (Vanderzwalmen et al., 2008; Balaban et al., 2011; de Almeida Ferreira Braga et al., 2011; Knez et al., 2011)
- 14. Meta-analysis Setti et al., 2010 IMSI outcomes (% of top quality embryos, IR, PR) significantly improved as compared with ICSI cycles. Miscarriage rate significantly decreased as compared with ICSI. NO difference between ICSI & IMSI regarding the FR (late paternal effect??)
- 15. When to use IMSI • Severe oligo-terato-asthenospermia Antinori et al., 2008 (prospective randomized trial; 446 couples: ICSI (n=219) VS IMSI (n=227) higher IR & PR) Setti et al., 2011: Sperm selection under high magnification results in improve treatment outcomes in patients with OATS • Previous conventional ICSI failures with significant % of DNA fragmented spermatozoa in the ejaculate Hazout et al., 2006: - Improved pregnancy outcome in pt with repeated conventional ICSI failure - Subgroup of pt (n=72): assessment of DNA integrity (TUNEL)improvement in clinical outcomes (IR & LBR) in pt with >30% DNA fragmentation • Couples with 1-3 yrs of infertility & semen characteristics 1-20mill/ml Wilding et al., 2011: - Improved embryo morphology together with significantly increased PR & IR • De Vos et al., 2013: no benefit of IMSI in a non-selected population with fresh ejaculation sperm containing >1mil/ml • Klement et al., 2013: IMSI procedure is an option to be mentioned after the 1st unsuccessful ICSI cycle
- 16. Sperm preparation techniques • Both SUP & DGP efficient in recovering lower percentage of sperm cells with vacuolization (Monqaut et al., 2011; Borges et al., 2013) • POOR SPERM MOTILITY : temperature 37°C, no PVP and supplemented with 6% of human serum albumin • HIGH SPERM MOTILITY : temperature 20°C and concentration of PVP about 8%.
- 17. Benefits • Berkovitz et al., 2007 Sperm nucleus morphological normalcy assessed at high magnification could decrease the prevalence of major fetal malformations in ICSI children
- 18. Concerns • Prolonged exposure to 37°C during IMSI could impair the morphological integrity of the sperm nuclei (Peer et al., 2007) • The No of spermatozoa without vacuoles in humans is extremely small and trying to find and then inject such cells is a very difficult & time-consuming process (Tanaka et al., 2012)
- 19. References • Antinori, M., Licata, E., Dani, G., et al., 2008. Intracytoplasmic morphologically selected sperm injection: a prospective randomized trial. Reprod. Biomed. Online 16, 835–841. • Bartoov, B., Berkovitz, A., Eltes, F., 2001. Selection of spermatozoa with normal nuclei to improve the pregnancy rate with intracytoplasmic sperm injection. N. Engl. J. Med. 345, 1067–1068. • Bartoov, B., Berkovitz, A., Eltes, F., et al., 2003. Pregnancy rates are higher with intracytoplasmic morphologically selected sperm injection than with conventional intracytoplasmic injection. Fertil. Steril. 80, 1413–1419. • Bartoov, B., Berkovitz, A., Eltes, F., et al., 2002. Real-time fine morphology of motile human sperm cells is associated with IVF-ICSI outcome. J. Androl. 23, 1–8. • Hazout, A., Dumont-Hassan, M., Junca, A.M., et al., 2006. High-magnification ICSI overcomes paternal effect resistant to conventional ICSI. Reprod. Biomed. Online 12, 19–25. • Vanderzwalmen, P., Hiemer, A., Rubner, P., et al., 2008. Blastocystmdevelopment after sperm selection at high magnification is associated with size and number of nuclear vacuoles. Reprod. Biomed. Online 17, 617–627. • Palermo, G., Joris, H., Devroey, P., et al., 1992. Pregnancies after intracytoplasmic injection of single spermatozoon into an oocyte. Lancet 340, 17–18. • Berkovitz, A., Eltes, F., Yaari, S., Katz, N., Barr, I., Fishman, A., Bartoov, B., 2005. The morphological normalcy of the sperm nucleus and pregnancy rate of intracytoplasmic injection with morphologically selected sperm. Hum. Reprod. 20, 185–190. • Berkovitz, A., Eltes, F., Lederman, H., Peer, S., Ellenbogen, A., Feldberg, B., Bartoov, B., 2006a. How to improve IVF- ICSI outcome by sperm selection. Reprod. Biomed. Online 12, 634–638. • Berkovitz, A., Eltes, F., Ellenbogen, A., Peer, S., Feldberg, D., Bartoov, B., 2006b. Does the presence of nuclear vacuoles in human sperm selected for ICSI affect pregnancy outcome? Hum. Reprod. 21, 1787–1790. • Knez, K., Zorn, B., Tomazevic, T., Vrtacnik-Bokal, E., Virant-Klun, I., 2011. The IMSI procedure improves poor embryo development in the same infertile couples with poor semen quality: a comparative prospective randomized study. Reprod. Biol. Endocrinol. 9, 123.
- 20. References • Franco Jr., J.G., Baruffi, R.L., Mauri, A.L., Petersen, C.G., Oliveira, J.B., Vagnini, L., 2008. Significance of large nuclear vacuoles in human spermatozoa: implications for ICSI. Reprod. Biomed. Online 17, 42–45. • Souza Setti A, Ferreira RC, Paes de Almeida Ferreira Braga D, de Cassia Savio Figueira R, Iaconelli A Jr, Borges E Jr. Intracytoplasmic sperm injection outcome versus intracytoplasmic morphologically selected sperm injection outcome: a meta-analysis. Reprod Biomed Online 2010;21:450–5. • Balaban B, Yakin K, Alatas C, Oktem O, Isiklar A, Urman B. Clinical outcome of intracytoplasmic injection of spermatozoa morphologically selected under high magnification: a prospective randomized study. Reprod Biomed Online 2011;15 [Epub ahead of print]. • Fawcett DW. The structure of the mammalian spermatozoon. Int Rev Cytol 1958;7:195–234. • Chrzanowski S. Ultrastructure of human spermatozoon. Pol Med J 1966;5: • 482–91. • Bedford JM. Observation on the fine structure of spermatozoa of the Bush baby (Galago senegalensis), the African green monkey (Cercopothecus aethiops) and man. Am J Anat 1967;121:443–60. • Pedersen H. Ultrastructure of the ejaculated human sperm. Z Zellforsch 1969;94:542–54. • Mundy AI, Ryder TA, Edmonds DK. A quantitative study of sperm head ultrastructure in subfertile males with excess sperm precursors. Fertil Steril 1994;61:751–4.