DNA recombinant technology: Insulin production, gene therapy, safety issues
- 2. A series of procedures used to recombine DNA segments.. Under certain conditions, a recombinant DNA molecule can enter a cell and replicate. Definition of recombinant DNA technology(r DNA)
- 3. History of recombinant DNA technology Recombinant DNA technology is one of the recent advances in biotechnology, which was developed by two scientists named Boyer and Cohen in 1973.
- 4. Basic principle of recombinant DNA technology The DNA is inserted into another DNA molecule called ‘vector’ . The recombinant vector is then introduced into a host cell where it replicates itself, the gene is then produced
- 5. Basic principle of recombinant DNA technology
- 6. Applications of Recombinant DNA Technology Large-scale production of human proteins by genetically engineered bacteria. Such as : insulin, Growth hormone, Interferons and Blood clotting factors (VIII & IX)
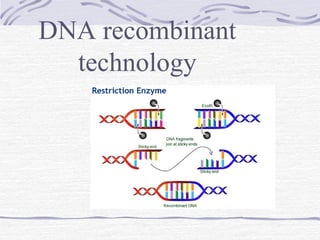
- 7. Production of Human Insulin 1) Obtaining the human insulin gene Human insulin gene can be obtained by making a complementary DNA (cDNA) copy of the messenger RNA (mRNA) for human insulin.
- 8. 2)Joining the human insulin gene into a plasmid vector The bacterial plasmids and the cDNA are mixed together. The human insulin gene (cDNA) is inserted into the plasmid through complementary base pairing at sticky ends.
- 9. 3)Introducing the recombinant DNA plasmids into bacteria The bacteria E.coli is used as the host cell. If E. coli and the recombinant plasmids are mixed together in a test-tube.
- 10. 4)Selecting the bacteria which have taken up the correct piece of DNA The bacteria are spread onto nutrient agar. The agar also contains substances such as an antibiotic which allows growth of only the transformed bacteria.
- 11. Vaccinedevelopment The surface antigen of Plasmodium falciparum, one of the 4 species of malaria has been transferred to E. coli to produce amounts large enough to develop a vaccine against this form of malaria. It works well enough for people who will visit a malarious region for a relatively short period of time
- 12. Hemophilia A and B The genes encoding factors 8 and 9 are on the X chromosome. Like other X-linked disorders, hemophilia A and B are found almost exclusively in males because they inherit just a single X chromosome, and if the gene for factor 8 (or 9) on it is defective, they will suffer from the disease. There are many different mutant versions of the genes for factors 8 and 9. Although some produce only a minor effect on the function of their protein, others fail to produce any functioning clotting factor.
- 13. Treating Hemophilia A and B Factor 8 and 9 can be extracted from donated blood, usually pooled from several thousand donors, and purified. Injections of this material can halt episodes of bleeding in hemophiliacs and have allowed countless young men to live relatively normal lives. However, blood contaminated with the human immunodeficiency virus (HIV) was unknowingly used to manufacture preparations of factors 8 and 9. Many have since died of AIDS.
- 14. The Future of Treating Hemophilia A and B •all donated blood is now tested to see if the donor has been infected with HIV (as well as hepatitis B and C); •plasma-derived preparations of factors 8 and 9 are now treated with heat and/or solvents to destroy any viruses that might be present; •recombinant factor 8 and recombinant factor 9 made by genetic engineering are now available. Now the treatment become more safety!
- 15. Still in the experimental stages, it may be possible to transfer the gene for normal adult hemoglobin into marrow stem cells of an individual with sickle-cell anemia. The goal is to promote the growth of enough cells to produce enough normal hemoglobin to alleviate the symptoms of sickle-cell anemia. One hundred percent (100%) is NOT required to attain the alleviation of symptoms. Gene therapy for genetic diseases
- 16. Safety Issues in relation to Recombinant DNA Technology As bacteria is commonly used in recombinant DNA work, there has always been a concern among scientists and a worry among people that there is a possibility that a clone of highly pathogenic recombinant bacteria were made by accident, then escaped from the laboratory and caused an epidemic for which no drugs were available. Recombinant DNA Advisory Committee (RAC) was established in 1974 in the United States, which responds to public concerns regarding the safety of manipulation of genetic material through the use of recombinant DNA techniques.
- 17. 2 types of control : physical containment and biological containment Effective biological safety programs were operated in a variety of laboratories, which include a set of standard practices generally used in microbiological laboratories, and special procedures, equipment and laboratory installations that provide physical barriers of varying degrees.
- 18. In considering biological containment, the vector (plasmid, organelle, or virus) for the recombinant DNA and the host (bacterial, plant, or animal cell) in which the vector is propagated in the laboratory will be considered together. (i) survival of the vector in its host outside the laboratory, and (ii) transmission of the vector from the propagation host to other non- laboratory hosts. Biological containment
- 19. Dangerous of DNA recombinant technology It is always possible that an antibiotic-resistant plasmid could be accidentally incorporated into a dangerous pathogen with serious medical consequences.
- 20. ADDRESS MAHESH MAHENDRAKAR Lecturer in Biotechnology Mahendrakar.mahesh@gmail.com
