Principles of Combustion (GIKI)
- 1. Combustions
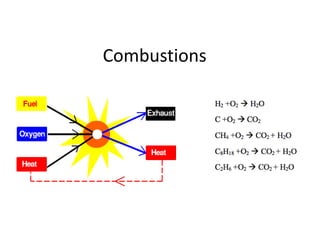
- 2. Principles of combustion • Combustion refers to the rapid oxidation of fuel accompanied by the production of heat, or heat and light. • Complete combustion (complete oxidation) of a fuel is possible only in the presence of an adequate supply of oxygen. (Air: 20.9% oxygen, 79% nitrogen and other gases). • Rapid fuel oxidation results in large amounts of heat. • Solid or liquid fuels must be changed to a gas before they will burn. • Fuel gases will burn in their normal state if enough air is present. • Nitrogen is considered to be a temperature reducing diluter. • Nitrogen reduces combustion efficiency by absorbing heat from the combustion of fuels and diluting the flue gases. • Nitrogen reduces the heat available for transfer through the heat exchanger surfaces. • It also increases the volume of combustion by-products, which then have to travel through the heat exchanger and up the stack faster to allow the introduction of additional fuel-air mixture. • Nitrogen also can combine with oxygen (particularly at high flame temperatures) to produce oxides of nitrogen (NOx), which are toxic pollutants. • Carbon may also combine with oxygen to form carbon monoxide, which results in the release of a smaller quantity of heat. Carbon burned to CO2 will produce more heat per unit of fuel than when CO or smoke are produced.
- 3. • The objective of good combustion is to release all of the heat in the fuel which is accomplished by controlling o Temperature which should be high enough to ignite and maintain ignition of the fuel o Turbulence or intimate mixing of the fuel and oxygen o Time should be sufficient for complete combustion. • Commonly used fuels like natural gas and propane generally consist of carbon and hydrogen so the flue gases mainly consist of CO2 and H2O. • Water vapor formed removes heat from the flue gases, which would otherwise be available for more heat transfer.
- 4. Stoichiometric calculation of air required • Stoichiometry is a branch of chemistry that deals with the relative quantities of reactants and products in chemical reactions. • For combustion air is needed. The amount of air needed can be calculated by: • The first step is to calculate the theoretical amount of air required for combustion. For example, for each kg of fuel burnt, 14.12 kg of air is required. • The second step is to calculate the theoretical CO2 content in the flue gases, for example 15.5%. • The next step is to measure the actual CO2 percentage in the flue gases, for example 10%. • This can then be used to calculate the % of excess air, for example 55%. • Once you know this you can calculate the weight of constituents in e.g. 100 kg of fuel with 55% excess air: CO2, H2O, SO2, O2, N2 • Finally you can convert the weight into volume of constituents. This could result in for example 10% CO2 and 7.5% O2.
- 5. Concept of Excess Air • For optimum combustion, the real amount of combustion air must be greater than that required theoretically. This additional amount of air is called “excess air”. • A certain amount of excess air is needed for complete combustion of fuel. • Too much excess air leads to heat losses and too little excess air leads to incomplete combustion. • You can tell if there is too little or too much excess air by measuring the CO2 in the flue gases. • The amount of excess air required depend on various factors like type of fuel used, type of firing system, size of fuel in case of solid fuel etc. Combustion analyzer
- 7. Residual oxygen (%) Excess air (%) This illustrates the relation between residual oxygen and excess air. The higher the percentage of oxygen measured in the flue gas, the more excess air there is. The typical range of O2 percentage that indicates combustion is good, e.g. 6-8%, depends on type of fuel used and the firing system.
- 8. Exercise A sample of coal has the following composition: Carbon = 80% Hydrogen = 20% Calculate the amount of theoretical air required for complete combustion.
