Case record...Parasellar craniopharyngioma
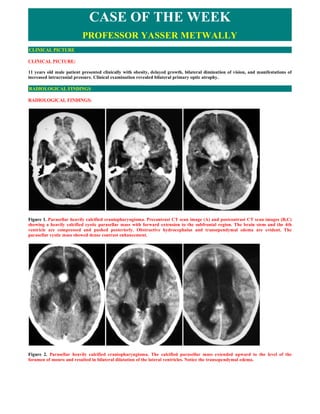
- 1. CASE OF THE WEEK PROFESSOR YASSER METWALLY CLINICAL PICTURE CLINICAL PICTURE: 11 years old male patient presented clinically with obesity, delayed growth, bilateral diminution of vision, and manifestations of increased intracranial pressure. Clinical examination revealed bilateral primary optic atrophy. RADIOLOGICAL FINDINGS RADIOLOGICAL FINDINGS: Figure 1. Parasellar heavily calcified craniopharyngioma. Precontrast CT scan image (A) and postcontrast CT scan images (B,C) showing a heavily calcified cystic parasellar mass with forward extension to the subfrontal region. The brain stem and the 4th ventricle are compressed and pushed posteriorly. Obstructive hydrocephalus and transependymal edema are evident. The parasellar cystic mass showed dense contrast enhancement. Figure 2. Parasellar heavily calcified craniopharyngioma. The calcified parasellar mass extended upward to the level of the foramen of monro and resulted in bilateral dilatation of the lateral ventricles. Notice the transependymal edema.
- 2. Figure 3. CT scan precontrast A, and postcontrast B,C,D. A heavily calcified suprasellar cystic craniopharyngioma, with subfrontal extension, the lesion also showed suprasellar extension to the foramen of monro causing obstructive hydrocephalus. Dense contrast enhancement occurred in some parts of the tumour. DIAGNOSIS: DIAGNOSIS: PARASELLAR CRANIOPHARYNGIOMA DISCUSSION DISCUSSION: Introduction Craniopharyngiomas represent 3% of all intracranial neoplasms. 17 They are common suprasellar and intrasellar tumors in childhood. They are most frequently seen in children between the ages of 5 and 10 years. A second age peak is encountered between the fifth and seventh decades. The tumors are mostly suprasellar in location, but the tumor also can originate within the sella and extend through the sella to the suprasellar region. These tumors originate from the squamous epithelial remnant of Rathke's pouch at the junction of the infundibulum and the pituitary gland. Figure 4. Suprasellar Adamantinomatous craniopharyngioma compressing the hypothalamus, and the brain stem Microscopically the tumour is composed of a solid and a cystic component. The solid parts of the tumour are composed of sheets of epithelial cells arranged in several layers; the periphery is formed by a basal layer of palisading cells beneath which several layers of stratified epithelial cells are found resulting in the formation of horny pearls composed of keratinized cells,among these epithelial areas small cysts lined by palisading cells may be seen as well as deposits of lamellar bone. These epithelial areas are separated by connective tissues rich in blood vessels ,microcysts, macrophage, cholesterol crystals and lymphocytic infiltrates. The cystic portions of the tumour are lined by stratified squamous epithelium. The cystic fluid is rich in cholesterol crystals and protein substance.
- 3. Figure 5. histopathological picture of a craniopharyngioma,notice the squamous epithelium,microcysts and cholesterol crystals Figure 6. A, Craniopharyngioma, compressing the optic chiasma, hypothalamus and extending upward into the lateral ventricle. The tumour is partially cystic with calcified material. B, A sagittal section of the brain shows a large craniopharyngioma below the cerebral ventricle. Note the stippled pattern of the tumor. Craniopharyngiomas are divided into two types based on clinical and microscopic features-ordinary or adamantinomatous and papillary. Both are midline tumors that arise in the region of the sella. Adamantinomatous craniopharyngiomas occur more caudally, often with intrasellar as well as suprasellar involvement, whereas papillary tumors are suprasellar and often extend to involve the third ventricle. 5,14 About half occur in children and adolescents, and half occur in adults. 4,7 The cells of origin of craniopharyngiomas are controversial. As the alternative name, adamantinoma, implies, some bear strong resemblance to the adamantinoma of the jaw and to the calcifying odontogenic cyst. This resemblance has prompted the consideration that craniopharyngiomas arise from rests of enamel organ epithelium. 2,10,12 Squamous epithelial rests of Rathke's pouch found in the pars tuberalis of the pituitary stalk are a more likely candidate. 1,9,10,13,14,15,17,18 The significant differences and the different location and clinical behavior raise the possibility of a different origin for the adamantinomatous and papillary variants-1. Adamantinomatous craniopharyngiomas are more common and predominately Adamantinomatous craniopharyngiomas have a cystic with frequent calcification. characteristic microscopic appearance. 6,13,17 A palisading layer of basaloid epithelium surrounds irregularly arranged cells that resemble the stellate reticulum of the epidermis. These nests may be solid but often form a complex trabecular network of microcysts. Bands of fibrous tissue weave between nests of epithelial cells and around cysts. The squamous epithelium of craniopharyngiomas demonstrates typical immunoreactivity for keratin and has no special immunohistochemical features. It usually does not undergo surface maturation with keratinization and desquamation of flattened epithelial cells, but this pattern may be present in the flattened epithelium of larger cysts, where it may mimic an epidermoid cyst. Instead, microscopic nodules of keratin debris form from the degeneration of plump squamous cells, the outlines of which impart a distinctive appearance: Although the cysts appear empty after tissue processing and sectioning, in vivo they contain cholesterol-rich oil that is highly irritating and produces striking fibroglial and foreign body reactions. These findings alone are sufficient for a presumptive diagnosis of craniopharyngioma in a limited biopsy specimen that does not demonstrate epithelial elements. Microcalcifications and necrotic foci often are present. Papillary craniopharyngiomas are something of a misnomer. Squamous epithelial nests that surround loose fibrovascular tissue rather than microcysts create a solid tumor with a pseudopapillary pattern. The peripheral epithelium lacks a basaloid appearance. Keratin nodules are not seen.
- 4. Figure 7.Classic adamantinoma-like appearance of a craniopharyngioma. The pink region corresponds to brain tissue (BT) at the interface with the tumor and is mainly composed of reactive astrocytes. The tumor itself is composed of epithelial cells arranged around cystic spaces. The cells proximal to the spaces are cuboidal in shape and arranged in palisades, above this cell layer the epithelium became squamous. Some cysts are empty but others contain keratin debris (K). B, Gross pathological specimen of a craniopharyngioma Figure 8. Epithelial lesion with peripheral palisading of basal squamous epithelium surrounding loosely arranged epithelial cells, the so-called quot;stellate reticulumquot; and nodules of keratin and variable calcification are typical histologic features of a craniopharyngioma. Craniopharyngiomas are slow-growing, indolent tumors and are classified as WHO grade I. Similar to chordomas, their classification by some as low-grade malignant tumors is more clinical than biologic because of the difficulty of surgical cure. The tumors spread locally along intracranial surfaces and do not metastasize. As they spread, they form strongly adherent fibroglial attachments to any structures they encounter, including brain, cranial nerves, and critical vascular structures. Complete surgical removal is the optimal treatment for a craniopharyngioma. 4,7 Their pattern of spread among the complex surfaces of the skull base, however, often makes complete removal impossible or, with involvement of the hypothalamus and third ventricle, clinically too costly. Consequently, postoperative radiation therapy is employed for local control of the tumor. 4,7,14 About 50% of patients remain free of disease, with therapeutic failure resulting in a slowly progressive course. 4,7 Papillary craniopharyngiomas have a slightly worse prognosis, probably more as a result of their location than biologic aggressiveness. Rarely, carcinomatous transformation occurs in recurrent tumors. 9 Figure 9. Papillary craniopharyngioma: In contrast to the adamantinomatous variant, papillary craniopharyngiomas do not show the complex heterogeneous architecture seen in the former but rather are composed of simple squamous epithelium and fibrovascular islands of connective tissue (hematoxylin-eosin, x40) (A). Papillary craniopharyngiomas: Under high power, only simple squamous epithelium is seen in papillary craniopharyngioma. The distinctive peripheral nuclear palisading, internal stellate reticulum, and nodules of quot;wetquot; keratin, that typify the adamantinomatous variant, are not seen in the papillary variant (hematoxylin-eosin, x100). (B)
- 5. Figure 10. Adamantinomatous craniopharyngiomas: Another distinctive feature of the adamantinomatous variant is scattered nodules of keratin. These nodules are referred to as quot;wetquot; keratin because of the plump appearance of the keratinocytes; this is in contrast to the flat, flaky keratin seen in epidermoid and dermoid cysts (hematoxylin-eosin, x100) (A). Adamantinomatous craniopharyngiomas: Nodules of quot;wetquot; keratin frequently calcify; in aggregate, this calcification can often be detected on CT scans, which is a recognized radiologic feature of craniopharyngiomas (hematoxylin-eosin, x100).(B) Figure 11. The adamantinomatous craniopharyngioma is a histologically complex epithelial lesion with several, very distinctive morphologic features. (hematoxylin-eosin, x40) (A). Adamantinomatous craniopharyngiomas: Peripheral palisading of the epithelium is a pronounced feature (hematoxylin-eosin, x100) (B). Adamantinomatous craniopharyngiomas: Frequently, the inner epithelium beneath the superficial palisade undergoes hydropic vacuolization and is referred to as the stellate reticulum (hematoxylin-eosin, x100) (C). The gross features and growth patterns of both types of craniopharyngioma are similar, even if their locations are not. Craniopharyngiomas attach to and spread along the surfaces of structures of the suprasellar region. Bony infiltration is less common than in chordomas. Their external surfaces are lobular and appear smooth. They have interdigitating interfaces with surrounding soft tissue, including brain, where a striking glial response is provoked. 8 The cut surfaces demonstrate a complex network of fibrous septa and nodules. Cysts are prominent in adamantinomatous tumors, where they are filled with cholesterol- rich fluid or soft necrotic debris. In some cases, a single large cyst predominates. 6,13 Papillary tumors are predominantly solid. Calcification is variably prominent, again more so in adamantinomatous tumors. Table 1. Differences between adamantinomatous and papillary craniopharyngiomas Adamantinomatous craniopharyngiomas Papillary craniopharyngiomas More common Less common Occur at a younger age Occur at an older age Commonly calcified Calcification is less common Commonly cystic and filled with cholesterol-rich fluid or soft Commonly solid necrotic debris A palisading layer of basaloid epithelium surrounds irregularly Squamous epithelial nests that surround loose fibrovascular arranged cells that resemble the stellate reticulum of the tissue rather than microcysts create a solid tumor with a epidermis. These nests may be solid but often form a complex pseudopapillary pattern. trabecular network of microcysts. Bands of fibrous tissue weave between nests of epithelial cells and around cysts. Keratin nodules are commonly seen. Keratin nodules are not seen. The differential diagnosis of craniopharyngioma is limited. The gross appearance is generally distinctive. Microscopically the principal considerations are epidermoid cyst, discussed earlier, and metastatic squamous carcinoma. Some authors believe that the distinction between craniopharyngioma and epidermoid cysts is artificial. 13,17 Infiltration of brain parenchyma induces an exuberant gliosis, often with Rosenthal fibers, that can resemble a hypothalamic pilocytic astrocytoma. Figure 12a. Rosenthal fibers in neuropil surrounding the craniopharyngioma. The brain parenchyma that surrounds both variants of craniopharyngioma is typically gliotic and often shows profuse numbers of eosinophilic Rosenthal fibers. The latter structures are composed of densely compacted bundles of glial filaments and are typically seen in astrocytic cell processes of neuropil that has been subjected to chronic compression from slowly expanding mass lesions. Rosenthal fibers are a characteristic feature of juvenile pilocytic astrocytomas (JPAs), which may also arise in the suprasellar/third ventricular region. Hence, a biopsy that samples only the surrounding neuropil of a craniopharyngioma may yield an erroneous diagnosis of JPA, if the pathologist is unaware of the close association of craniopharyngioma with Rosenthal fiber formation (hematoxylin-eosin, x100).
- 6. Figure 12b. A, Craniopharyngiomas account for about 3% of all intraranial tumors. They are most frequently seen in children and adolescents. These tumors are suprasellar in location and it is thought that they arise from epithelial remnants of Rathke's pouch that are trapped in the stalk of the pituitary gland. Grossly, the tumor appears partially solid and partially cystic and compresses the optic chiasm in front, the pituitary below, and the third ventricle above. B, Craniopharyngiomas are composed of sheets of squamous epithelial cells and keratin set in a loose connective tissue stroma. Note in this image that sheets of epithelial cells (E) surround cysts, some of which contain keratin (K). CT scan imaging of craniopharyngiomas Craniopharyngiomas occur almost exclusively in the region of the sella turcica and third ventricle and account for 1.2% to 3% of intracranial tumors. 24 They are tumors of epithelial origin and can be divided into adamantinomatous craniopharyngiomas and squamous-papillary craniopharyngiomas based on histopathology. 24 Mixed tumors, having histologic features of both types, occur 15% of the time. The clinical and imaging features of mixed tumors are the same as those of the adamantinomatous type. 24 As a group, craniopharyngiomas have a bimodal age distribution. Most are encountered in children and adolescents, with a peak incidence between 6 and 10 years of age. 20 A second, smaller peak occurs in the 50s. Half of all suprasellar tumors in children are craniopharyngiomas. 25 Figure 13. A, This photograph of a lateral X-ray of the skull shows the expansion of the sella turcica and calcification in mass (look between arrows). B, Suprasellar cystic craniopharyngioma with peripheral enhancement Figure 14. Suprasellar heavily calcified solid craniopharyngioma with intrasellar extension,notice ballooning of the sella
- 7. Craniopharyngioma, which develops from remnants of Rathke's pouch, may arise within the sella but is most characteristically centered above the dorsum sellae. Suprasellar calcification on plain x-rays is evident in over 90 per cent of childhood tumours but in under 50 per cent of adult craniopharyngiomas. The tip of the dorsum sellae may be truncated by the longstanding suprasellar mass; occasionally the outlet of the sella may be widened with the calcification extending down into it, this is called an open sella. Figure 15. A solid heavily calcified craniopharyngioma,with the characteristic open sella type The pathologic features of craniopharyngiomas may be quite varied. This is reflected in the diversity of CT findings. The characteristic type of craniopharyngiomas consists of a cystic lesion. In this case, CT shows a hypodense round lesion with a solid calcification or a peripheral rim of punctate or continuous rim of calcification. The hypodense lesion is frequently demonstrated to be lower attenuation than CSF, reflecting the presence of thick proteinaceous material. In other cases, the CT evidence of calcification may be absent. There may be thin peripheral or variable thickness rim of enhancement in craniopharyngioma. The finding of a hypodense lesion of a craniopharyngioma may be similar to that seen with subarachnoid cysts, epidermoid cysts, or cystic pituitary adenomas. Figure 16. Parasellar craniopharyngioma: A, Precontrast CT scan (left) and postcontrast CT scan (right), B, postcontrast MRI T1 image (left) and precontrast MRI T1 image (right). Notice the calcification, the mild postcontrast enhancement, the MRI signal heterogeneity and the associated hydrocephalus. Craniopharyngiomas that are predominantly solid appear as irregularly marginated heterogeneous hyperdense lesions with irregular and complex enhancement; however, in other cases, there may be homogeneous dense sharply marginated enhancement. In rare cases, craniopharyngiomas may be isodense without evidence of calcification. However complete absence of calcified elements is very rare in children, but over 20 per cent of tumours presenting in adults are uncalcified. If this lesion does not enhance, it may only be delineated by the finding of anterior third ventricular distortion and effacement of the suprasellar cistern. In these isodense craniopharyngiomas, metrizamide CT cisternography or MRI may be necessary to demonstrate the presence of the tumor.
- 8. Figure 17. Craniopharyngioma intraoperatively The tumour may be large, displacing the adjacent brain substance and extending through the region of the third ventricle to the foramina of Monro causing hydrocephalus. The relationship of the tumour to the optic chiasm and third ventricle is well shown on sagittal reformatted sections. Angiography is the only additional study which is likely to be required before radical surgery, mainly to demonstrate the relationship of the Posterior communicating artery to the mass and to estimate the relative importance of the contributions of this vessel and the basilar artery to the blood supply to the tumour. MR imaging of craniopharyngiomas On MR imaging, craniopharyngiomas exhibit variable appearance, depending on the solid and cystic components of the mass. The solid lesions appear hypointense on Tl- weighted images and hyperintense on T2- weighted images, with areas of dark signal intensity contributed by calcifications. A unique MR imaging appearance of craniopharyngioma is produced by machine oil content of the cyst, in which the cyst appears hyperintense both in precontrast Tl-weighted and in T2- weighted images. The hyperintensity of the cyst within craniopharyngioma in Tl- weighted images has been attributed to a high concentration of protein and cholesterol. Figure 18. Suprasellar craniopharyngioma, precontrast MRI T1 images (A,C) and postcontrast MRI T1 image (B). Notice the partial tumour enhancement, the T1 hyperintensity in the precontrast images is either due to increased lipid content of the sac or previous haemorrhage, also notice the associated hydrocephalus
- 9. Figure 19. Craniopharyngioma. A 6-year-old boy presenting with nausea, vomiting, and conjugate gaze disturbance. A, CT image reveals a mass, isodense to the brain, compressing the cerebral peduncles and third ventricle. There is resulting hydrocephalus. B, The mass has high signal intensity on a Tl-weighted sagittal image. It is entirely suprasellar in location and effaces the third ventricle. C, The lesion has homogeneously high signal intensity on the T2-weighted image. Again seen is mass effect on the pons and evidence of hydrocephalus. Adamantinomatous craniopharyngiomas are the more common variety. These tumors frequently present as suprasellar masses causing symptoms of increased intracranial pressure: headache, nausea, and vomiting. The lesions are lobulated masses and almost always have a cystic component. The cystic component is generally hyperintense on T2-weighted images but can also be hypointense depending on the cyst contents. 24 It contains proteinaceous fluid that typically causes Tl shortening and hyperintense signal on Tl-weighted images. The solid component of the lesion demonstrates intense enhancement with contrast administration. Calcification is common and is better seen on CT. Encasement of adjacent vessels is a feature of adamantinomatous craniopharyngiomas that is not seen in the squamous papillary type. Adamantinomatous craniopharyngiomas must be distinguished from other parasellar lesions demonstrating increased signal on T1-weighted images. These include Rathke's cleft cysts, lipomas, dermoids, aneurysms, and cases of an ectopic posterior pituitary bright spot. Squamous-papillary craniopharyngiomas are less likely to have calcifications than adamantinomatous craniopharyngiomas and are primarily solid rather than cystic lesions. They often are rounded in shape as opposed to lobulated. 24 On MR imaging, their appearance is nonspecific. The solid portion of these tumors is isointense to gray matter on Tl-weighted images and hyperintense on T2-weighted images. These tumors demonstrate marked contrast enhancement. They are more common in adult patients and often originate in the third ventricle. In contrast to adamantinomatous craniopharyngiomas, these lesions rarely recur after excision . 24 Figure 20. Suprasellar cystic craniopharyngioma with intrasellar extension, notice the cystic hypointensity with compression of the optic chiasma superiorly, MRI T1 postcontrast Figure 21. Suprasellar cystic craniopharyngioma with intrasellar extension, notice the cystic hyperintensity which is attributable to increased lipid or protein content of the cyst, MRI T1 precontrast An ectopic posterior pituitary bright spot can be seen in patients with pituitary gland hypoplasia as well as after trauma and surgery. 22,23 The ectopic posterior pituitary bright spot is a relatively small rounded lesion. Although this abnormality has
- 10. increased signal on T1-weighted images, signal on T2- weighted images is not as intense as that seen in a craniopharyngioma. Partially thrombosed aneurysms may have regions of increased Tl and T2 signal. These lesions, however, typically demonstrate signal misregistration artifact in the phase encoding direction because of pulsatile flow as well as a characteristic flow void. 19,21 SUMMARY SUMMARY Craniopharyngioma is a slow-growing, extra-axial, epithelial-squamous, calcified, cystic tumor arising from remnants of the craniopharyngeal duct and/or Rathke cleft and occupying the (supra)sellar region. Two main hypotheses explain the origin of craniopharyngioma—embryogenetic and metaplastic; they complement each other and explain the craniopharyngioma spectrum.26 The embryogenetic theory26 The embryogenetic theory relates to the development of the adenohypophysis and transformation of the remnant ectoblastic cells of the craniopharyngeal duct and the involuted Rathke pouch. Both the Rathke pouch and the infundibulum develop during the fourth week of gestation and together form the hypophysis. Both elongate and come in contact during the second month. The infundibulum is a downward invagination of diencephalons; Rathke pouch is an upward invagination of the primitive oral cavity (stomodeum). The craniopharyngeal duct is the neck of the pouch, connecting to the stomodeum, which narrows, closes, and separates the pouch from the primitive oral cavity by the end of the second month. Thus, the pouch becomes a vesicle, which flattens and surrounds the anterior and lateral surfaces of the infundibulum. Walls of this vesicle form different structures of the hypophysis. Finally, this vesicle involutes into a mere cleft and may disappear completely. Rathke cleft, together with remnants of the craniopharyngeal duct, can be the site of origin of craniopharyngiomas. The metaplastic theory 26 The metaplastic theory relates to the residual squamous epithelium (derived from stomodeum and normally part of the adenohypophysis), which may undergo metaplasia. The dual theory explains the craniopharyngioma spectrum attributing the adamantinous type (most prevalent in childhood) to embryonic remnants and the adult type (squamous papillary) to metaplastic foci derived from mature cells of the anterior hypophysis (prevalence of the adult type increases with each decade of life and is almost never found in children). Other cystic lesions may originate from remnants of the stomodeum and pharyngohypophyseal duct as well, such as Rathke cleft cysts, epithelial cysts, epidermoid cysts, and dermoid cysts. 26 Addendum A new version of this PDF file (with a new case) is uploaded in my web site every week (every Saturday and remains available till Friday.) To download the current version follow the link quot;http://pdf.yassermetwally.com/case.pdfquot;. You can also download the current version from my web site at quot;http://yassermetwally.comquot;. To download the software version of the publication (crow.exe) follow the link: http://neurology.yassermetwally.com/crow.zip The case is also presented as a short case in PDF format, to download the short case follow the link: http://pdf.yassermetwally.com/short.pdf At the end of each year, all the publications are compiled on a single CD-ROM, please contact the author to know more details. Screen resolution is better set at 1024*768 pixel screen area for optimum display. For an archive of the previously reported cases go to www.yassermetwally.net, then under pages in the right panel, scroll down and click on the text entry quot;downloadable case records in PDF formatquot; REFERENCES
- 11. References 1. Asa SL, Kovacs K, Bilbao JM, et al: Immunohistochemical localization of keratin craniopharyngiomas and squamous cell nests of the human pituitary. Acta Neuropathol 54:257-260, 1981 2. Badger KV, Gardner DG: The relationship of adamantinomatous craniopharyngioma to ghost cell ameloblastoma of the jaws: A histopathologic and immunohistochemical study. J Oral Pathol Med 26:349-355,1997 3. Banjeree AK, Sharma BS, Kak VK, et al: Gliosarcoma with cartilage formation. Cancer 63:518-523, 1989 9. Banna M: Craniopharyngioma in adults. Surg Neurol 1:202-204,1973 4. Baskin DS, Wilson CB: Surgical management of craniopharyngiomas. J Neurosurg 65:22-27, 1986 5. Crotty TB, Scheithauer BW, Young WF Jr, et al: Papillary craniopharyngioma: A clinicopathological study of 48 cases. J Neurosurg 83:206-214, 1995 6. Goldberg GM, Eshbaugh DE: Squamous cell nests of the pituitary gland as related to the origin of craniopharyngiomas. Arch Pathol 70:293-299,1960 7. Hoff JT, Patterson RH Jr: Craniopharyngioma in children and adults. J Neurosurg 36:299-302,1972 8. Kasai H, Hirano A, Liena JF, et al: A histopathological study of craniopharyngioma with special reference to its stroma and surrounding tissue. Brain Tumor Pathol 14:41-45, 1997 9. Nelson GA, Bastian FO, Schfitt M, et al: Malignant transformation in craniopharyngioma. Neurosurgery 22:427-429,1988 10. Oka H, Kawano N, Yagishita S, et al: Ciliated craniopharyngioma indicates histogenetic relationship to Rathke cleft epithelium. Clin Neuropathol 16:103- 106,1997 11. Paulus W, Stockel C, Drauss J, et al: Odontogenic classification of craniopharyngiomas: A clinicopathological study of 54 cases. Histopathology 30:172- 176,1997 12. Petito CK: Craniopharyngioma: Prognostic importance of histologic features [comments]. AJNR Am j Neuroradiol 17:1427- 1439, 1996 13. Petito CK, DeGirolami U, Earle KM: Craniopharyngiomas: A clinical and pathological review. Cancer 37:1944-1952,1976 14. Pollack IF, Lunsford MD, Slamovits TL, et al: Stereotaxic intracavitary irradiation for cystic craniopharyngiomas. J Neurosurg 68:227-233,1988 14. Pusey E, Kortman KE, Flannigan BD, et al: MR of craniopharyngioma. Neurosurgery 422:427-429, 1988 16. Sartoretti-Schefer S, Wichmann W, Aguzzi A, et al: MR differentiation of adamantinomatous and squamous- papillary craniopharyngiomas. AJNR Am j Neuroradiol 18:77-87, 1977 17. Russell DS , Rn Lj: Pathology of Tumors of the Nervous System, ed 5. Baltimore, Williams & Wilkins, 1989 18. Boggan JE, Davis RL, Zorman G, et al: Intrasellar epidermoid cyst: Case report. I Neurosurg 58:411-415, 1983 19. Biondi A, Scialfa G, Scotti G: Intracranial aneurysms: MR imaging. Neuroradiology 30:214-218,1988 20. Carmel PW, Antunes JL, Chang CH: Craniopharyngiomas in children. Neurosurgery 11:382-389,1982 21. Donovan JL, Nesbit GM: Distinction of masses involving the sella and suprasellar space: Specificity of imaging features. AJR Am J Roentgenol 167:597-603, 1996 22. Fujisawa 1, Kikuchi K, Nishimura K, et al: Transection of the pituitary stalk: Development of an ectopic posterior lobe assessed with MR imaging. Radiology 165:487-489, 1987 23. Kelly WM, Kucharczyk W, Kucharczyk J, et al: Posterior pituitary ectopia: An MR feature of pituitary dwarfism. AJNR Am j Neuroradiol 9:453-460,1988 24. Sartoretti-Schefer S, Wichmann W, Aguzzi A: MR differentiation of adamantinomatous and squamous-papillary craniopharyngiomas. AJNR Am j Neuroradiol 18:77-87,1997 25. Osborn AG: Brain tumors and tumorlike masses: Classification and differential diagnosis. In Diagnostic Neuroradiology. St Louis, Mosby, 1994 26. Metwally, MYM: Textbook of neuroimaging, A CD-ROM publication, (Metwally, MYM editor) WEB-CD agency for electronic publication, version 9.2a April 2008
