Metals 1
•Als PPT, PDF herunterladen•
1 gefällt mir•1,034 views
This document provides information about the periodic table of elements. It discusses the key properties of metals, nonmetals, and metalloids based on their location on the periodic table. Metals are located on the left side and have properties like luster, malleability, conductivity, and corrosion. Nonmetals are on the right side and have properties such as being dull, brittle, low density, and poor conductivity. Metalloids are in between and share some properties of both metals and nonmetals.
Melden
Teilen
Melden
Teilen
Empfohlen
Weitere ähnliche Inhalte
Was ist angesagt?
Was ist angesagt? (19)
Andere mochten auch
Andere mochten auch (20)
Primero corporate presentation january 2015 update final

Primero corporate presentation january 2015 update final
Ähnlich wie Metals 1
Ähnlich wie Metals 1 (20)
PPT - Properties of Metals Non-Metals Metalloids.ppt

PPT - Properties of Metals Non-Metals Metalloids.ppt
Presentation - Properties of Metals Non-Metals Metalloids.ppt

Presentation - Properties of Metals Non-Metals Metalloids.ppt
PPT - Properties of Metals Non-Metals Metalloids (1).ppt

PPT - Properties of Metals Non-Metals Metalloids (1).ppt
PPT - Properties of Metals Non-Metals Metalloids.pptx

PPT - Properties of Metals Non-Metals Metalloids.pptx
M NM M Review and Practice metals nonmetals etc ppt.ppt

M NM M Review and Practice metals nonmetals etc ppt.ppt
Mehr von Sylvester Saragih
Mehr von Sylvester Saragih (20)
Presentation washing plant kel. 5 Pencucian batubara dengan jig, pencucian ba...

Presentation washing plant kel. 5 Pencucian batubara dengan jig, pencucian ba...
Pencucian batubara kel 4 Operasi pemisahan bak media berat dan operasi siklon...

Pencucian batubara kel 4 Operasi pemisahan bak media berat dan operasi siklon...
Kelompok 3 Teori Pengendapan partikel untuk konsentrasi operasi dan prinsip ...

Kelompok 3 Teori Pengendapan partikel untuk konsentrasi operasi dan prinsip ...
Bahan materi kuliah rekayasa bahan galian industri

Bahan materi kuliah rekayasa bahan galian industri
Tugas Kelompok II Operasi kominusi dan operasi pengayakan Dalam pencucian bat...

Tugas Kelompok II Operasi kominusi dan operasi pengayakan Dalam pencucian bat...
Uji Ketercucian dalam pencucian batubara (tugas kelompk I)

Uji Ketercucian dalam pencucian batubara (tugas kelompk I)
Tugas amdal uu no 32 tahun 2009 pplh terhadap lingkungan tambang

Tugas amdal uu no 32 tahun 2009 pplh terhadap lingkungan tambang
Bahan MK PERALATAN DAN PENGANGKUTAN TAMBANG BAWAH TANAH.Peralatan tambang baw...

Bahan MK PERALATAN DAN PENGANGKUTAN TAMBANG BAWAH TANAH.Peralatan tambang baw...
Tugas makalah teknik eksplorasi tambang peralatan yang digunakan alam eksplor...

Tugas makalah teknik eksplorasi tambang peralatan yang digunakan alam eksplor...
Tugas batubara ii lingkungan dan bentuk endapan batubara, kalsifikasi dan jen...

Tugas batubara ii lingkungan dan bentuk endapan batubara, kalsifikasi dan jen...
Tugas paper cekungan batubara pada pulau kalimantan

Tugas paper cekungan batubara pada pulau kalimantan
Kürzlich hochgeladen
VVVIP Call Girls In Greater Kailash ➡️ Delhi ➡️ 9999965857 🚀 No Advance 24HRS Live
Booking Contact Details :-
WhatsApp Chat :- [+91-9999965857 ]
The Best Call Girls Delhi At Your Service
Russian Call Girls Delhi Doing anything intimate with can be a wonderful way to unwind from life's stresses, while having some fun. These girls specialize in providing sexual pleasure that will satisfy your fetishes; from tease and seduce their clients to keeping it all confidential - these services are also available both install and outcall, making them great additions for parties or business events alike. Their expert sex skills include deep penetration, oral sex, cum eating and cum eating - always respecting your wishes as part of the experience
(07-May-2024(PSS)VVVIP Call Girls In Greater Kailash ➡️ Delhi ➡️ 9999965857 🚀 No Advance 24HRS...

VVVIP Call Girls In Greater Kailash ➡️ Delhi ➡️ 9999965857 🚀 No Advance 24HRS...Call Girls In Delhi Whatsup 9873940964 Enjoy Unlimited Pleasure
Saudi Arabia [ Abortion pills) Jeddah/riaydh/dammam/++918133066128☎️] cytotec tablets uses abortion pills 💊💊 How effective is the abortion pill? 💊💊 +918133066128) "Abortion pills in Jeddah" how to get cytotec tablets in Riyadh " Abortion pills in dammam*💊💊 The abortion pill is very effective. If you’re taking mifepristone and misoprostol, it depends on how far along the pregnancy is, and how many doses of medicine you take:💊💊 +918133066128) how to buy cytotec pills
At 8 weeks pregnant or less, it works about 94-98% of the time. +918133066128[ 💊💊💊 At 8-9 weeks pregnant, it works about 94-96% of the time. +918133066128) At 9-10 weeks pregnant, it works about 91-93% of the time. +918133066128)💊💊 If you take an extra dose of misoprostol, it works about 99% of the time. At 10-11 weeks pregnant, it works about 87% of the time. +918133066128) If you take an extra dose of misoprostol, it works about 98% of the time. In general, taking both mifepristone and+918133066128 misoprostol works a bit better than taking misoprostol only. +918133066128 Taking misoprostol alone works to end the+918133066128 pregnancy about 85-95% of the time — depending on how far along the+918133066128 pregnancy is and how you take the medicine. +918133066128 The abortion pill usually works, but if it doesn’t, you can take more medicine or have an in-clinic abortion. +918133066128 When can I take the abortion pill?+918133066128 In general, you can have a medication abortion up to 77 days (11 weeks)+918133066128 after the first day of your last period. If it’s been 78 days or more since the first day of your last+918133066128 period, you can have an in-clinic abortion to end your pregnancy.+918133066128
Why do people choose the abortion pill? Which kind of abortion you choose all depends on your personal+918133066128 preference and situation. With+918133066128 medication+918133066128 abortion, some people like that you don’t need to have a procedure in a doctor’s office. You can have your medication abortion on your own+918133066128 schedule, at home or in another comfortable place that you choose.+918133066128 You get to decide who you want to be with during your abortion, or you can go it alone. Because+918133066128 medication abortion is similar to a miscarriage, many people feel like it’s more “natural” and less invasive. And some+918133066128 people may not have an in-clinic abortion provider close by, so abortion pills are more available to+918133066128 them. +918133066128 Your doctor, nurse, or health center staff can help you decide which kind of abortion is best for you. +918133066128 More questions from patients: Saudi Arabia+918133066128 CYTOTEC Misoprostol Tablets. Misoprostol is a medication that can prevent stomach ulcers if you also take NSAID medications. It reduces the amount of acid in your stomach, which protects your stomach lining. The brand name of this medication is Cytotec®.+918133066128) Unwanted Kit is a combination of two medicines, ounwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi![unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE AbudhabiAbortion pills in Kuwait Cytotec pills in Kuwait
Kürzlich hochgeladen (20)
Ensure the security of your HCL environment by applying the Zero Trust princi...

Ensure the security of your HCL environment by applying the Zero Trust princi...
VVVIP Call Girls In Greater Kailash ➡️ Delhi ➡️ 9999965857 🚀 No Advance 24HRS...

VVVIP Call Girls In Greater Kailash ➡️ Delhi ➡️ 9999965857 🚀 No Advance 24HRS...
B.COM Unit – 4 ( CORPORATE SOCIAL RESPONSIBILITY ( CSR ).pptx

B.COM Unit – 4 ( CORPORATE SOCIAL RESPONSIBILITY ( CSR ).pptx
Russian Call Girls In Gurgaon ❤️8448577510 ⊹Best Escorts Service In 24/7 Delh...

Russian Call Girls In Gurgaon ❤️8448577510 ⊹Best Escorts Service In 24/7 Delh...
Call Girls Pune Just Call 9907093804 Top Class Call Girl Service Available

Call Girls Pune Just Call 9907093804 Top Class Call Girl Service Available
FULL ENJOY Call Girls In Mahipalpur Delhi Contact Us 8377877756

FULL ENJOY Call Girls In Mahipalpur Delhi Contact Us 8377877756
Uneak White's Personal Brand Exploration Presentation

Uneak White's Personal Brand Exploration Presentation
Falcon's Invoice Discounting: Your Path to Prosperity

Falcon's Invoice Discounting: Your Path to Prosperity
FULL ENJOY Call Girls In Majnu Ka Tilla, Delhi Contact Us 8377877756

FULL ENJOY Call Girls In Majnu Ka Tilla, Delhi Contact Us 8377877756
Quick Doctor In Kuwait +2773`7758`557 Kuwait Doha Qatar Dubai Abu Dhabi Sharj...

Quick Doctor In Kuwait +2773`7758`557 Kuwait Doha Qatar Dubai Abu Dhabi Sharj...
Call Girls Service In Old Town Dubai ((0551707352)) Old Town Dubai Call Girl ...

Call Girls Service In Old Town Dubai ((0551707352)) Old Town Dubai Call Girl ...
Chandigarh Escorts Service 📞8868886958📞 Just📲 Call Nihal Chandigarh Call Girl...

Chandigarh Escorts Service 📞8868886958📞 Just📲 Call Nihal Chandigarh Call Girl...
Call Girls Ludhiana Just Call 98765-12871 Top Class Call Girl Service Available

Call Girls Ludhiana Just Call 98765-12871 Top Class Call Girl Service Available
Call Girls in Delhi, Escort Service Available 24x7 in Delhi 959961-/-3876

Call Girls in Delhi, Escort Service Available 24x7 in Delhi 959961-/-3876
RSA Conference Exhibitor List 2024 - Exhibitors Data

RSA Conference Exhibitor List 2024 - Exhibitors Data
unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi![unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
unwanted pregnancy Kit [+918133066128] Abortion Pills IN Dubai UAE Abudhabi
Metals 1
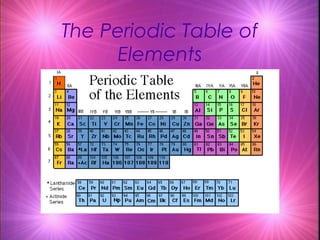
- 1. The Periodic Table of Elements
- 2. Elements Science has come along way since Aristotle’s theory of Air, Water, Fire, and Earth. Scientists have identified 92 Natural elements, and created about 28 others.
- 3. Elements The elements, alone or in combinations, make up our bodies, our world, our sun, and in fact, the entire universe.
- 4. Periodic Table A great deal of information about an element can be gathered from its position on the period table. Understanding the organization and plan of the periodic table will help you obtain basic information about each of the 118 known elements.
- 6. Metals
- 7. Properties of Metals Metals appear to the leftof the dark ziz-zag line on the periodic table. Most metals are solidat room temperature.
- 8. Properties of Metals Metals have luster. This means they are shiny
- 9. Properties of Metals Ductile metals can be drawn into wire.
- 10. Properties of Metals Malleable metals can be hammered into sheets
- 11. Properties of Metals Metals have a high melting point. They are also very dense.
- 12. Properties of Metals Conductors Metals are good conductors of electricity and heat
- 13. Properties of Metals A chemical property of metal is its reaction with water and oxygen. This results in corrosion and rust.
- 14. Nonmetals
- 15. Properties of Nonmetals Nonmetals occur to the right of the dark zig-zag on the periodic table. Although Hydro gen is in family 1, it is also a nonmetal. Many nonmetals are gases at room temperature.
- 16. Properties of Nonmetals Nonmetals do not have luster; they are dull.
- 17. Properties of Nonmetals Brittle Nonmetals are brittle so they break easily. This means nonmetals ARE NOT ductileor malleable.
- 18. Properties of Nonmetals Nonmetals have low density.
- 19. Properties of Nonmetals They also have a low melting point. This is why they are poor conductors of heat and electricity.
- 20. Metalloids
- 21. Propertiesof Metalloids Metalloids can be found clustered around the dark zig-zag line that separates metals and nonmetals.
- 22. Propertiesof Metalloids Metalloids (metal- like) have properties of both metals and nonmetals.
- 23. Propertiesof Metalloids Metalloids are solids that can be shiny or dull.
- 24. Propertiesof Metalloids They conduct electricity and heat better than nonmetals but not as well as metals.
- 25. Propertiesof Metalloids Metalloids are malleable and ductile
- 28. Families Families in the periodic table share chemical properties because all elements in a family have the same number of valence electrons This means that all elements in a family bond with other atoms in a similar way.
- 29. Properties of Metals Most metals have 3 or less valence electrons and therefore are likely to lose these electrons in chemical bonds.
- 30. Properties of Nonmetals Nonmetals have 5 or more valence electrons and therefore usually gain electrons in chemical bonds.
